Abstract
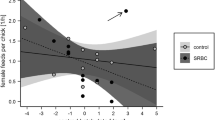
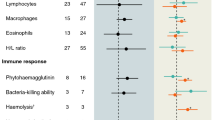
Hatching asynchrony in avian species leads to age and size differences between nestlings within a brood, handicapping last-hatched chicks in the sibling rivalry. Starvation due to this competitive disadvantage has been regarded as the primary cause of an increase in mortality with hatching order. However, for gulls it has also been suggested that disease is the cause of mortality for last-hatched chicks, possibly through reduced immunocompetence and thereby an enhanced susceptibility to infection. In addition, the male-biased mortality reported for several gull species may be related to a higher vulnerability to diseases in males compared to females. To determine the potential influence of the immune system on these mortality patterns, we investigated the T-cell-mediated immunity (CMI) of black-headed gull chicks in relation to hatching order and sex. We found a significant decrease in the CMI with hatching order. This result may be causally related to systematic changes in maternal yolk steroids and carotenoids within the laying sequence. For second-laid eggs, male CMI was significantly lower than female CMI. This is possibly linked to higher plasma levels of testosterone in male embryos which might have an immunosuppressive effect. If so, this effect is masked in eggs of either high (first egg) or low (last egg) quality. Chicks with low CMI showed enhanced mortality rates. Thus the differences in immune response are likely to contribute to the observed mortality patterns. However, hatching order significantly affected mortality independently of CMI, suggesting that competitive disadvantage due to hatching asynchrony is also important.


Similar content being viewed by others
References
Alonso-Alvarez C, Tella JL (2001) Effects of experimental food restriction and body-mass changes on avian T-cell mediated immune response. Can J Zool 79:101–105
Apanius V (1998) Ontogeny of immune function. In: Starck JM, Ricklefs RE (eds): Avian growth and development: evolution within the altricial-precocial. Oxford University Press, Oxford, pp. 203–222
Blount JD, Surai PF, Nager RG, Houston DC, Møller AP, Trewby ML, Kennedy M (2002) Carotenoids and egg quality in the lesser black-backed gull Larus fuscus: a supplemental feeding study of maternal effects. Proc R Soc Lond B 269:29–36
Blount, JD, Metcalfe, NB, Birkhead, TR, Surai, PF (2003) Carotenoid modulation of immune function and sexual attractiveness in zebra finches. Science 300:125–127
Bolton M (1991) Determinants of chick survival in the lesser black-backed gull: relative contributions of egg size and parental quality. J Anim Ecol 61:949–960
Bowry TR (1984) Immunology. Oxford University Press, Oxford
Clark AB, Wilson DS (1981) Avian breeding adaptations: hatching asynchrony, brood reduction and nest failure. Q Rev Biol 56:253–277
Cramp S, Simmons KEL (eds.) (1983) The birds of the western Palearctic, vol 3. Oxford University Press, Oxford
Christe P, Møller AP, de Lope F (1998) Immunocompetence and nestling survival in the house martin: the tasty chick hypothesis. Oikos 83:175–179
Dijkstra C, Daan S, Pen I (1998) Fledgling sex ratios in relation to brood size in size-dimorphic altricial birds. Behav Ecol 9: 287–296
Duffy DL, Bentley GE, Drazen DL, Ball GF (2000) Effects of testosterone on cell-mediated and humoral immunity in non-breeding adult European starlings. Behav Ecol 11:654–662
Eising CM, Eikenaar C, Schwabl H, TGG Groothuis (2001) Maternal androgens in black-headed gull (Larus ridibundus) eggs: consequences for chick development. Proc R Soc Lond B 268:839–846
Fargallo AJ, Laaksonen T, Pöyri V, Korpimäki E (2002) Inter-sexual differences in the immune response of Eurasian kestrel nestlings under food shortage. Ecology Lett 5: 95–101
Folstad I, Carter AJ (1992) Parasites, bright males, and the immunocompetence handicap. Am Nat 139:603–622
Glutz von Blotzheim UN, Bauer KM (1982) Handbuch der Vögel Mitteleuropas, band 8. Akademische Verlagsgesellschaft, Wiesbaden
Goldstein H (1995) Multilevel statistical approach. Arnold, London
Griffiths R (1992) Sex-biased mortality in the lesser black-backed gull Larus fuscus during the nestling stage. Ibis 134:237–244
Griffiths R, Double MC, Orr K, Dawson RJG (1998) A DNA test to sex most birds. Mol Ecol 7:1071–1075
Groothuis TGG, Schwabl H (2002) The influence of laying sequence and habitat characteristics on maternal yolk hormone levels. Funct Ecol 16:281–289
Hario M, Rudbäck E (1996) Effects of supplementary feeding on the C-egg constituents in the nominate lesser black-backed gull Larus f. fuscus. Ornis Fenn 74:167–177
Hario M, Rudbäck E (1999) Dying in the midst of plenty – the third chick fate in nominate lesser black-backed gulls Larus f. fuscus. Ornis Fenn 76:71–77
Hasselquist D, Marsh JA, Sherman PW, Wingfield, JC (1999) Is avian humoral immunocompetence suppressed by testosterone? Behav Ecol Sociobiol 45:167–175
Hõrak P, Tegelmann L, Ots I, Møller AP (1999) Immune function and survival of great tit nestlings in relation to growth conditions. Oecologia 121:316–322
Ketterson ED, Nolan V (1999) Adaptation, exaptation, and constraint: a hormonal perspective. Am Nat 154:4-25
Lessells CM, Boag PT (1987) Unrepeatable repeatabilities: a common mistake. Auk 104:116–121
Lipar JL, ED Ketterson (2000) Maternally derived yolk testosterone enhances the development of the hatching muscle in the red-winged blackbird Aegelius phoeniceus. Proc R Soc Lond B 267: 2005–2010
Magrath RD (1989) Hatching asynchrony and reproductive success in the blackbird. Nature 339:536–538
Martin, LB, Scheuerlein, A, Wikelski, M. (2002) Immune activity elevates energy expenditure of house sparrows: a link between direct and indirect costs. Proc R Soc Lond B 270:153–158
Martinez J, Potti J, Davila JA, Blanco G (2001) Sex, immune response and hatching asynchrony in Mediterranean kestrels (Falco tinnunculus). Abstr 4th Eurasian Congr Raptors, Seville 2001
Merino S, Møller AP, de Lope F (2000) Seasonal changes in cell-mediated immunocompetence and mass gain in nestling barn swallows: a parasite-mediated effect. Oikos 90:327–332
Müller W, Eising C, Dijkstra C, Groothuis TGG (2002) Sex differences in yolk hormones depend on maternal social status in Leghorn chickens (Gallus gallus domesticus). Proc R Soc Lond B 269:2249–2256
Nager RG, Monaghan P, Houston DC, Genovart M (2000a) Parental condition, brood sex ratio and differential young survival: an experimental study in gulls (Larus fuscus). Behav Ecol Sociobiol 48:452–457
Nager RG, Monaghan P, Houston DC (2000b) Within-clutch trade-offs between the number and quality of eggs; experimental manipulations in gulls. Ecology 81: 1339–1350
Norris K, Evans MR (2000) Ecological immunology: life history trade-offs and immune defense in birds. Behav Ecol 11:19–26
Parsons J (1975a) Asynchronous hatching and chick mortality in the herring gull Larus argentatus. Ibis 117:517–520
Parsons, J (1975b) Relationship between egg size and post-hatching chick mortality in the herring gull (Larus argentatus). Nature 228:1221–1222
Petrie M, Schwabl H, Brande-Lavridsen N, Burke T (2001) Sex differences in avian yolk hormone levels. Nature 412:498
Rasbash J, Browne W, Healy M, Cameron B, Charlton C (2000) Multilevel models project. University of London, London
Reid, WV (1987) Constraints on clutch size in the glaucous-winged gull. Stud Avian Biol 10:8-25
Royle NJ (2000) Overproduction in the lesser black-backed gull: can marginal chicks overcome the initial handicap of hatching asynchrony? J Avian Biol 31:335–344
Royle NJ, Surai PF, McCartney RJ, Speake BK (1999) Parental investment and egg yolk lipid composition in gulls. Funct Ecol 13:298–306
Royle NJ, Surai PF, Hartley IR (2001) Maternal derived androgens and antioxidants in bird eggs: complementary but opposing effects? Behav Ecol 12:381–385
Schantz T von, Bensch S, Grahn M, Hasselquist D, Wittzell H (1999) Good genes, oxidative stress and condition-dependent sexual signals. Proc R Soc Lond B: 266:1-12
Sayce JR, Hunt GL (1987) Sex ratios of prefledging western gulls. Auk 104:33–37
Schwabl H (1993) Yolk is a source of maternal testosterone for developing birds. Proc Natl Acad Sci USA 90:11446–11450
Schwabl H (1996) Maternal testosterone in the egg enhances postnatal growth. Comp Biochem Physiol114:271–276
Slagsvold T (1986) Hatching asynchrony: interspecific comparisons of altricial birds. Am Nat 128:120–125
Smits JE, Bortolotti GR, Tella JL (1999) Simplifying the phytohaemagglutinin skin-testing technique in studies of avian immunocompetence. Funct Ecol 13:567–572
Stockland JN, Amundsen, T (1988) Initial size hierarchy in broods of the shag: relative significance of egg size and hatching asynchrony. Auk 107:359–366
Surai PF (1999) Vitamin E in avian reproduction. Poult Avian Biol Rev 10:1-60
Tella JL, Bortolotti GR, Forero MG, Dawson RD (2000) Environmental and genetic variation in T-cell-mediated immune response of fledging American kestrels. Oecologia 123:453–459
Walsh PS, Metzger DA, Higuchi R (1991) Chelex-100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. Biotechniques 10:506–513
Williams JD (1994) Intraspecific variation in egg size and egg composition in birds: effects on offspring fitness. Biol Rev 68:35–69
Woods JE, Simpson RM, Moore PL (1975) Plasma testosterone levels in the chick embryo. Gen Comp Endocrinol 27:543–547
Acknowledgments
We thank Jenny de Vries and Durk Venema for their help in the field. We would also like to thank Akzo Nobel Delfzijl (particularly Harry Rodenhuis) and It Fryske Gea for granting us permission to work on their properties. We also thank the referees, Corine Eising, Serge Daan and Tim Fawcett for valuable comments on the manuscript. This project was approved by the animal experimentation committee of the University of Groningen (licences DEC 2687 and DEC 2843).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Webster
Rights and permissions
About this article
Cite this article
Müller, W., Dijkstra, C. & Groothuis, T.G.G. Inter-sexual differences in T-cell-mediated immunity of black-headed gull chicks (Larus ridibundus) depend on the hatching order. Behav Ecol Sociobiol 55, 80–86 (2003). https://doi.org/10.1007/s00265-003-0681-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-003-0681-5