Abstract
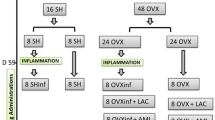
Epidemiological studies have reported an association between arterial calcification and bone loss after menopause. However, the underlying mechanism of the association remains unclear. Therefore, to explore the possible mechanisms of the association, we tried to develop a new combined model rat of ovariectomy (OVX, an animal model of osteoporosis) and vitamin D3 plus nicotine (VDN rat, an animal model of arterial calcification). We tested them by using sham-operated control rats (SC), OVX control rats (OC), and OVX plus VDN-treated rats (OVN). Dissections were performed twice at 4 (4SC, 4OC, and 4OVN) and 8 (8SC, 8OC, and 8OVN) weeks after treatment. 8OVN showed bone loss and arterial calcification, although 8OC showed only bone loss. Moreover, arterial calcium content was associated with indexes of bone loss at 8 weeks. Thus, the OVN rat is considered a good model to examine the relationship of the two disorders after menopause. Additionally, the arterial endothelin-1 (ET-1, a potent regulator of arterial calcification) levels increased in both 4OVN and 8OVN, and the level was associated with arterial calcium content at 8 weeks. Furthermore, the arterial endothelial nitric oxide synthase (eNOS) protein, which is an enzyme that produces nitric oxide (an antiatherosclerotic substance), was significantly reduced in only 8OVN. Estrogens affect the alterations of the eNOS and ET-1 proteins. Therefore, we suggest that impairment of the ET-1- and NO-producing system in arterial tissue during periods of rapid bone loss by estrogen deficiency might be a mechanism of the relationship between the two disorders seen in postmenopausal women.





Similar content being viewed by others
References
Farhat GN, Strotmeyer ES, Newman AB, Sutton-Tyrrell K, Bauer DC, Harris T, Johnson KC, Taaffe DR, Cauley JA (2006) Volumetric and areal bone mineral density measures are associated with cardiovascular disease in older men and women: the Health, Aging, and Body Composition Study. Calcif Tissue Int 79:102–111
Shaffer JR, Kammerer CM, Rainwater DL, O’Leary DH, Bruder JM, Bauer RL, Mitchell BD (2007) Decreased bone mineral density is correlated with increased subclinical atherosclerosis in older, but not younger, Mexican American women and men: the San Antonio Family Osteoporosis Study. Calcif Tissue Int 81:430–441
Mosca L, Manson JE, Sutherland SE, Langer RD, Manolio T, Barrett-Connor E (1997) Cardiovascular disease in women: a statement for healthcare professionals from the American Heart Association. Writing Group. Circulation 96:2468–2482
Kuh D, Langenberg C, Hardy R, Kok H, Cooper R, Butterworth S, Wadsworth ME (2005) Cardiovascular risk at age 53 years in relation to the menopause transition and use of hormone replacement therapy: a prospective British birth cohort study. BJOG 112:476–485
Nordin BE, Polley KJ (1987) Metabolic consequences of the menopause. A cross-sectional, longitudinal, and intervention study on 557 normal postmenopausal women. Calcif Tissue Int 41:S1–S59
Fischer M, Raue F (1999) Measurements of bone mineral density. Mineral density in metabolic bone disease. Q J Nucl Med 43:233–240
Tankò LB, Bagger YZ, Christiansen C (2003) Low bone mineral density in the hip as a marker of advanced atherosclerosis in elderly women. Calcif Tissue Int 73:15–20
Garcia-Perez MA, Moreno-Mercer J, Tarin JJ, Cano A (2006) Similar efficacy of low and standard doses of transdermal estradiol in controlling bone turnover in postmenopausal women. Gynecol Endocrinol 22:179–184
Hirose K, Tomiyama H, Okazaki R, Arai T, Koji Y, Zaydun G, Hori S, Yamashina A (2003) Increased pulse wave velocity associated with reduced calcaneal quantitative osteo-sono index: possible relationship between atherosclerosis and osteopenia. J Clin Endocrinol Metab 88:2573–2578
Hyder JA, Allison MA, Criqui MH, Wright CM (2007) Association between systemic calcified atherosclerosis and bone density. Calcif Tissue Int 80:301–306
Omi N, Ezawa I (1995) The effect of ovariectomy on bone metabolism in rats. Bone 17:163S–168S
Kalu DN, Liu CC, Hardin RR, Hollis BW (1989) The aged rat model of ovarian hormone deficiency bone loss. Endocrinology 124:7–16
Tatchum-Talom R, Martel C, Marette A (2002) Influence of estrogen on aortic stiffness and endothelial function in female rats. Am J Physiol Heart Circ Physiol 282:H491–H498
Park JH, Omi N, Nosaka T, Kitajima A, Ezawa I (2008) Estrogen deficiency and low calcium diet increased bone loss and urinary calcium excretion, but did not alter arterial stiffness in young female rats. J Bone Miner Metab 26:218–225
Fleckenstein A, Frey M, Zorn J, Fleckensteingreon G (1990) Calcium, a neglected key factor in hypertension and arteriosclerosis. In: Laragh JH, Brenner BM (eds) Hypertension: pathophysiology, diagnosis and management. Raven, New York, pp 471–509
Niederhoffer N, Bobryshev YV, Lartaud-Idjouadiene I, Giummelly P, Atkinson J (1997) Aortic calcification produced by vitamin D3 plus nicotine. J Vasc Res 34:386–398
Lartaud-Idjouadiene I, Lompre AM, Kieffer P, Colas T, Atkinson J (1999) Cardiac consequences of prolonged exposure to an isolated increase in aortic stiffness. Hypertension 34:63–69
Palmer RMJ, Ferrige AG, Moncada S (1987) Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 327:524–526
Moncada S, Palmer RM, Higgs EA (1991) Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev 43:109–142
Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, Yazaki Y, Goto K, Masaki T (1988) A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332:411–415
Rizvi MA, Myers PR (1997) Nitric oxide modulates basal and endothelin-induced coronary artery vascular smooth muscle cell proliferation and collagen levels. J Mol Cell Cardiol 29:1779–1789
Wu SY, Zhang BH, Pan CS, Jiang HF, Pang YZ, Tang CS, Qi YF (2003) Endothelin-1 is a potent regulator in vivo in vascular calcification and in vitro in calcification of vascular smooth muscle cells. Peptides 24:1149–1156
Bakker EN, van der Linden PJ, Sipkema P (1997) Endothelin-1-induced constriction inhibits nitric-oxide-mediated dilation in isolated rat resistance arteries. J Vasc Res 34:418–424
Ramzy D, Rao V, Tumiati LC, Xu N, Sheshgiri R, Miriuka S, Delgado DH, Ross HJ (2006) Elevated endothelin-1 levels impair nitric oxide homeostasis through a PKC-dependent pathway. Circulation 114:I319–I326
Potenza MA, Marasciulo FL, Chieppa DM, Brigiani GS, Formoso G, Quon MJ, Montagnani M (2005) Insulin resistance in spontaneously hypertensive rats is associated with endothelial dysfunction characterized by imbalance between NO and ET-1 production. Am J Physiol Heart Circ Physiol 289:H813–H822
Spina M, Garbin G (1976) Age-related chemical changes in human elastins from non-atherosclerotic areas of thoracic aorta. Atherosclerosis 24:267–279
Cattell MA, Anderson JC, Hasleton PS (1996) Age-related changes in amounts and concentrations of collagen and elastin in normotensive human thoracic aorta. Clin Chim 245:73–84
Cola C, Almeida M, Li D, Romeo F, Mehta JL (2004) Regulatory role of endothelium in the expression of genes affecting arterial calcification. Biochem Biophys Res Commun 320:424–427
Guo X, Razandi M, Pedram A, Kassab G, Levin ER (2005) Estrogen induces vascular wall dilation: mediation through kinase signaling to nitric oxide and estrogen receptors alpha and beta. J Biol Chem 280:19704–19710
Simoncini T, Hafezi-Moghadam A, Brazil DP, Ley K, Chin WW, Liao JK (2000) Interaction of oestrogen receptor with the regulatory subunit of phosphatidylinositol-3-OH kinase. Nature 407:538–541
Nosaka T, Tanaka H, Watanabe I, Sato M, Matsuda M (2003) Influence of regular exercise on age-related changes in arterial elasticity: mechanistic insights from wall compositions in rat aorta. Can J Appl Physiol 28:204–212
Qi YF, Shi YR, Bu DF, Pang YZ, Tang CS (2003) Changes of adrenomedullin and receptor activity modifying protein 2 (RAMP2) in myocardium and aorta in rats with isoproterenol-induced myocardial ischemia. Peptides 24:463–468
Kaito K, Urayama H, Watanabe G (2003) Doxycycline treatment in a model of early abdominal aortic aneurysm. Surg Today 33:426–433
Jegger D, da Silva R, Jeanrenaud X, Nasratullah M, Tevaearai H, von Segesser LK, Segers P, Gaillard V, Atkinson J, Lartaud I, Stergiopulo N (2006) Ventricular–arterial coupling in a rat model of reduced arterial compliance provoked by hypervitaminosis D and nicotine. Am J Physiol Heart Circ Physiol 291:H1942–H1951
Omi N, Morikawa N, Ezawa I (1994) The effect of voluntary exercise on bone mineral density and skeletal muscles in the rat model at ovariectomized and sham stages. Bone Miner 24:211–222
Myburgh KH, Noakes TD, Roodt M, Hough FS (1989) Effect of exercise on the development of osteoporosis in adult rats. J Appl Physiol 66:14–19
Sakai S, Miyauchi T, Sakurai T, Kasuya Y, Ihara M, Yamaguchi I, Goto K, Sugishita Y (1996) Endogenous endothelin-1 participates in the maintenance of cardiac function in rats with congestive heart failure. Marked increase in endothelin-1 production in the failing heart. Circulation 93:1214–1222
Iemitsu M, Miyauchi T, Maeda S, Matsuda M, Goto K, Yamaguchi I (2004) Time course alteration of endothelin-1 gene expression in the heart during exercise and recovery from post-exercise periods in rats. J Cardiovasc Pharmacol 44:S447–S450
Iemitsu M, Miyauchi T, Maeda S, Yuki K, Kobayashi T, Kumagai Y, Shimojo N, Yamaguchi I, Matsuda M (2000) Intense exercise causes decrease in expression of both endothelial NO synthase and tissue NOx level in hearts. Am J Physiol Regul Integr Comp Physiol 279:R951–R959
Faggiotto A (1988) Smooth muscle cell proliferation in atherosclerosis. Agents Actions 26:201–221
Partridge SM, Keeley FW (1974) Age related and atherosclerotic changes in aortic elastin. Adv Exp Med Biol 43:173–191
Mackey RH, Venkitachalam L, Sutton-Tyrrell K (2007) Calcifications, arterial stiffness and atherosclerosis. Adv Cardiol 44:234–244
Henrion D, Chillon JM, Godeau G, Muller F, Capdeville-Atkinson C, Hoffman M, Atkinson J (1991) The consequences of aortic calcium overload following vitamin D3 plus nicotine treatment in young rats. J Hypertens 9:919–926
Kieffer P, Robert A, Capdeville-Atkinson C, Atkinson J, Lartaud-Idjouadiene I (2000) Age-related arterial calcification in rats. Life Sci 66:2371–2381
Srivastava AK, Bhattacharyya S, Castillo G, Wergedal J, Mohan S, Baylink DJ (2000) Development and application of a serum C-telopeptide and osteocalcin assay to measure bone turnover in an ovariectomized rat model. Calcif Tissue Int 66:435–442
Nordin BE, Wishart JM, Clifton PM, McArthur R, Scopacasa F, Need AG, Morris HA, O’Loughlin PD, Horowitz M (2004) A longitudinal study of bone-related biochemical changes at the menopause. Clin Endocrinol 61:123–130
Nordin BE, Need AG, Morris HA, Horowitz M, Robertson WG (1991) Evidence for a renal calcium leak in postmenopausal women. J Clin Endocrinol Metab 72:401–407
Hiruma Y, Inoue A, Shiohama A, Otsuka E, Hirose S, Yamaguchi A, Hagiwara H (1998) Endothelins inhibit the mineralization of osteoblastic MC3T3–E1 cells through the A-type endothelin receptor. Am J Physiol Regul Integr Comp Physiol 275:R1099–R1105
Christodoulakos GE, Lambrinoudaki IV, Botsis DC (2006) The cardiovascular effects of selective estrogen receptor modulators. Ann N Y Acad Sci 1092:374–384
Stump AL, Kelley KW, Wensel TM (2007) Bazedoxifene: a third-generation selective estrogen receptor modulator for treatment of postmenopausal osteoporosis. Ann Pharmacother 41:833–839
Acknowledgments
The authors are grateful to Evan Thomas for help in the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, JH., Omi, N., Iemitsu, M. et al. Relationship Between Arterial Calcification and Bone Loss in a New Combined Model Rat by Ovariectomy and Vitamin D3 Plus Nicotine. Calcif Tissue Int 83, 192–201 (2008). https://doi.org/10.1007/s00223-008-9162-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-008-9162-1