Abstract
Objective
To determine the factors associated with adherence and persistence to bisphosphonate therapy in osteoporosis.
Design
Cross-sectional survey.
Setting
National survey in the UK.
Participants
Participants were recruited through the National Osteoporosis Society and advertisements in the press and on the radio and included 533 women over age 50 with osteoporosis who were currently taking or had taken bisphosphonate therapy within the previous 12 months.
Main outcome measures
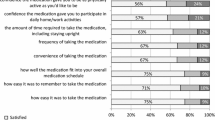
Self-reported factors influencing adherence and persistence to bisphosphonate therapy in osteoporosis: fracture history, pain, practical difficulties taking medication (frequency of dosing, dealing with comedications, impact on daily routine), perceptions of therapy, and concerns about bisphosphonate therapy.
Results
Adherence to bisphosphonate therapy was 48% and was associated with previous fracture [odds ratio (OR) 1.62, 95% confidence interval (CI) 1.14–3.02], concerns about medication (OR 1.49, 95% CI 1.01–2.20), and less dissatisfaction with medication (OR 0.65, 95% CI 0.44–0.97). Nonpersistence was associated with dissatisfaction with medication (hazard ratio (HR) 1.83, 95% CI 1.38–2.43), side effects (HR 3.69, 95% CI 2.74–4.97), and concerns about bisphosphonate therapy (HR 2.21, 95% CI 1.48–3.30). For both daily (HR 1.53, 95% CI 1.1–2.33) and weekly bisphosphonates (HR 1.90, 95% CI 1.17–3.07), practical difficulties taking bisphosphonate medication—in particular, too frequent dosing—were associated with nonpersistence.
Conclusions
Self-reported nonadherence to daily and weekly bisphosphonates is independent of the decision to stop taking treatment (nonpersistence). Nonpersistence is associated with side effects and other factors that could be modified in clinical practice through education, information, and concordant partnerships.
Similar content being viewed by others
References
Caro JJ, Ishak KJ, Huybrechts KF, Raggio G, Naujoks C (2004) The impact of compliance with osteoporosis therapy on fracture rates in actual practice. Osteoporosis Int 15:1003–1008
Tosteson AN, Grove MR, Hammond CS et al (2003) Early discontinuation of treatment for osteoporosis. Am J Med 115:209–216
Miller NH (1997) Compliance with treatment regimens in chronic asymptomatic diseases. Am J Med 102:43–49
Simon JA, Lewiecki EM, Smith ME et al (2002) Patient preference for once-weekly aledronate 70 mg versus once daily aledronate 10 mg: a multicenter, randomised, open label, cross-over study. Clin Ther 24:1871–1886
Donovan JL, Blake DR (1992) Patient noncompliance: deviance or reasoned decision-making? Soc Sci Med 34:507–513
Donovan JL, Blake DR, Fleming WG (1989) The patient is not a blank sheet: lay beliefs and their relevance to patient education. Br J Rheumatol 28:58–61
Horne R (1999) Patients’ beliefs about treatment: the hidden determinant of treatment outcome? J Psychosom Res 47:491–495
Carr AJ, Hughes RA, Vincent K, Carr M, Thwaites C (2004) The impact and implications of new treatments in arthritis: validation of a questionnaire to measure the ‘real life’ effectiveness of medication. Rheumatology 43(suppl):ii83
O’Neill TW, Cooper C, Cannata JB, Diaz Lopez, Hoszowski K et al (1994) Reproducibility of a questionnaire on risk factors for osteoporosis in a multicentre prevalence survey: The European Vertebral Osteoporosis Study. Int J Epidemiology 23(3):559–565
Fletcher RH (1989) Patient compliance with therapeutic advice: a modern view. The Mount Sinai Journal of Medicine 56:453–458
Kaplan RM, Simon HJ (1990) Compliance in medical care: reconsideration of self-predictions. Annals of Behavioural Medicine 12:66–71
Turbí C, Herrero-Beaumont G, Acebes JC, Torrijos A, Graña J, Miguélez R, Sacristán JA, Marín F (2004) Compliance and satisfaction with raloxifene versus alendronate for the treatment of postmenopausal osteoporosis in clinical practice: an open-label, prospective, nonrandomized, observational study. Clin Ther 26:245–256
Cramer J, Amonkar M, Hebborn A, Suppapanya N (2004) Does dosing regimen impact persistence with bisphosphonate therapy among postmenopausal osteoporotic women? J Bone Miner Res 19(Suppl 1):S448 (Abstract M434)
Unson CG, Siccion E, Gaztambide J, Gaztambide S, Mahoney Trella P, Prestwood K (2003) Nonadherence and osteoporosis treatment preferences of older women: a qualitative study. J Women’s Health 12:1037–1045
Kendler D, Kung AWC, El-Hajj Fuleihan G, González JGG, Gaines KA, Verbruggen N, Melton ME (2004) Patients with osteoporosis prefer once weekly to once daily dosing with alendronate. Maturitas 48; 243–251
Simon JA, Lewiecki EM, Smith ME, Petruschke RA, Wang L, Palmisano JJ (2002) Patient preference for once-weekly alendronate 70 mg versus once-daily alendronate 10 mg: a multicenter, randomized, open-label, crossover study. Clin Ther 24:1871–1886
Boccuzzi SJ, Folz SH, Omar MA, Kahler KH (2005) Assessment of adherence and persistence with daily and weekly dosing regimens of oral bisphosphonates. Osteoporosis Int 16(Suppl 4):S35–S36 (Abstract P7.10)
Ettinger MP, Gallagher R, Amonkar M, Smith JC, MacCosbe PE (2004) Medication persistence is improved with less frequent dosing of bisphosphonates, but remains inadequate. Arthritis Rheum 15(Suppl.):S513 (Abstract 1325)
Funding
This study was funded by Roche Products and GlaxoSmithKline. The funders had no role in the day-to-day running of the study, the data analysis or interpretation, the drafting of the paper, or the decision to publish. The study was run by an independent clinical research company, Clinimatrix, in association with C.C. and P.W.T. Data were collected and analysed by Clinimatrix and reviewed and interpreted by C.C. and P.W.T. The authors were responsible for determining the content of the paper, writing the paper, and deciding when and where to publish. The funders had no role in these processes.
Declaration of competing interests
P.T. and C.C. have received consulting fees from Roche Products, Merck Sharp & Dohme, and Procter & Gamble, all manufacturers of competing bisphosphonates. A.C. has shares in Clinimatrix, the independent clinical research company that obtained funding for this study from Roche Products and GlaxoSmithKline.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carr, A.J., Thompson, P.W. & Cooper, C. Factors associated with adherence and persistence to bisphosphonate therapy in osteoporosis: a cross-sectional survey. Osteoporos Int 17, 1638–1644 (2006). https://doi.org/10.1007/s00198-006-0166-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-006-0166-2