Abstract
Objective
Platinum nanoparticles (nano-Pt) have been reported to possess anti-oxidant and anti-tumor activities. However, the biological activity and mechanism of action of nano-Pt in inflammation are still unknown. The present study was designed to determine the in-vitro anti-inflammatory effects of nano-Pt on lipopolysaccharide (LPS)-stimulated RAW 264.7 cells.
Methods
RAW 264.7 macrophages were used for the study. The LPS-induced production of reactive oxygen species (ROS) was determined by flow cytometry. The prostaglandin E2 (PGE2) concentration was measured using a PGE2 assay kit. The protein levels and mRNA expression of the pro-inflammatory cytokines [tumor necrosis factor-α, interleukin (IL)-1β and IL-6], along with cyclooxygenase (COX-2) and inducible nitric oxide synthase, were analyzed by Western blotting and reverse transcription–polymerase chain reaction analysis. The phosphorylation of extracellular signal regulated kinase (ERK1/2) and Akt, and the phosphorylation and degradation of inhibitory kappa B-alpha (IκB-α) was determined by Western blot analysis.
Results
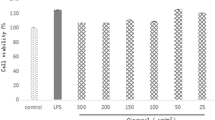
Nano-Pt significantly reduced the LPS-induced production of intracellular ROS and inflammatory mediators. In addition, nano-Pt suppressed the phosphorylation of ERK1/2 and Akt, and significantly inhibited the phosphorylation/degradation of IκB-α as well as nuclear factor kappa-B (NFκB) transcriptional activity.
Conclusion
These results suggest that the anti-inflammatory properties of nano-Pt may be attributed to their downregulation of the NFκB signaling pathway in macrophages, thus supporting the use of nano-Pt as an anti-inflammatory agent.








Similar content being viewed by others
Abbreviations
- Nano-Pt:
-
Platinum nanoparticles
- RT:
-
Reverse transcription
- PGE2 :
-
Prostaglandin E2
- iNOS:
-
Inducible nitric oxide synthase
- MAPK:
-
Mitogen activated protein kinases
References
Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539–45.
Lonkar P, Dedon PC. Reactive species and DNA damage in chronic inflammation. Int J Cancer. 2011;128:1999–2009.
Zaidi SF, Ahmed K, Yamamoto T, Kondo T, Usmanghani K, Kadowaki M, Sugiyama T. Effect of resveratrol on Helicobacter pylori-induced interleukin-8 secretion, reactive oxygen species generation and morphological changes in human gastric epithelial cells. Bio Pharm Bull. 2009;32:1931–5.
Lin WW, Karin M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J Clin Invest. 2007;117:1175–83.
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420:860–7.
Lawrence T, Willoughby DA, Gilroy DW. Anti-inflammatory lipid mediators and insights into the resolution of inflammation. Nat Rev Immunol. 2002;2:787–95.
Zaidi SF, Yamamoto T, Refaat A, Ahmed K, Sakurai H, Saiki I, Kondo T, Usmanghani K, Kadowaki M. Sugiyama T modulation of activation-induced cytidine deaminase by curcumin in Helicobacter pylori-infected gastric epithelial cells. Helicobacter. 2009;14:588–95.
Lee YM, Seon MR, Cho HJ, Kim JS, Park JH. Benzyl isothiocyanate exhibits anti-inflammatory effects in murine macrophages and in mouse skin. J Mol Med. 2009;87:1251–61.
Chaturvedi MM, Sung B, Yadav VR, Kannappan R, Aggarwal BB. NF-κB addiction and its role in cancer: ‘one size does not fit all’. Oncogene. 2011;30:1615–30.
Hoffmann A, Xia Y, Verma IM. Inflammatory tales of liver cancer. Cancer Cell. 2007;11:99–101.
Kajita M, Hikosaka K, Iitsuka M, Kanayama A, Toshima N, Miyamoto Y. Platinum nanoparticle is a useful scavenger of superoxide anion and hydrogen peroxide. Free Radic Res. 2007;41:615–26.
Bhattacharya R, Mukherjee P. Biological properties of “naked” metal nanoparticles. Adv Drug Deliv Rev. 2008;60:1289–306.
Yoshihisa Y, Zhao QL, Hassan MA, Wei ZL, Furuichi M, Miyamoto Y, Kondo T, Shimizu T. SOD/catalase mimetic platinum nanoparticles inhibit heat-induced apoptosis in human lymphoma U937 and HH cells. Free Radic Res. 2011;45:326–35.
Park YM, Won JH, Yun KJ, Ryu JH, Han YN, Choi SK, Lee KT. Preventive effect of Ginkgo biloba extract (GBB) on the lipopolysaccharide-induced expressions of inducible nitric oxide synthase and cyclooxygenase-2 via suppression of nuclear factor-kappaB in RAW 264.7 cells. Biol Pharm Bull. 2006;29:985–90.
Korhonen R, Lahti A, Kankaanranta H, Moilanen E. Nitric oxide production and signaling in inflammation. Cur Drug Targets Inflamm Allergy. 2005;4:471–9.
Hancock JT, Desikan R, Neill SJ. Role of reactive oxygen species in cell signaling pathways. Biochem Soc Trans. 2001;29:345–50.
Choi SY, Hwang JH, Ko HC, Park JG, Kim SJ. Nobiletin from citrus fruit peel inhibits the DNA-binding activity of NFκB and ROS production in LPS-activated RAW 264.7 cells. J Ethnopharmacol. 2007;113:149–55.
Yoshihisa Y, Honda A, Zhao QL, Makino T, Abe R, Matsui K, Shimizu H, Miyamoto Y, Kondo T, Shimizu T. Protective effects of platinum nanoparticles against UV-light-induced epidermal inflammation. Exp Dermatol. 2010;19:1000–6.
Nomura M, Yoshimura Y, Kikuiri T, Hasegawa T, Taniguchi Y, Deyama Y, Koshiro K, Sano H, Suzuki K, Inoue N. Platinum nanoparticles suppress osteoclastogenesis through scavenging of reactive oxygen species produced in RAW264.7 cells. J Pharmacol Sci. 2011;117:243–52.
Mazza J, Rossi A, Weinberg JM. Innovative uses of tumor necrosis factor alpha inhibitors. Dermatol Clin. 2010;28:559–75.
Jamieson ER, Lippard SJ. Structure, recognition, and processing of cisplatin–DNA adducts. Chem Rev. 1999;99:2467–98.
Asharani PV, Xinyi N, Hande MP, Valiyaveettil S. DNA damage and p53-mediated growth arrest in human cells treated with platinum nanoparticles. Nanomedicine. 2010;5:51–64.
Gehrke H, Pelka J, Hartinger CG, Blank H, Bleimund F, Schneider R, Gerthsen D, Bräse S, Crone M, Türk M, Marko D. Platinum nanoparticles and their cellular uptake and DNA platination at non-cytotoxic concentrations. Arch Toxicol. 2011;85:799–812.
Ma JS, Kim WJ, Kim JJ, Kim TJ, Ye SK, Song MD, Kang H, Kim DW, Moon WK, Lee KH. Gold nanoparticles attenuate LPS-induced NO production through the inhibition of NF-kappaB and IFN-beta/STAT1 pathways in RAW264.7 cells. Nitric Oxide. 2010;23:214–9.
Cargnello M, Roux PP. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev. 2011;75:50–83.
Turjanski AG, Vaque JP, Gutkind JS. MAP kinases and the control of nuclear events. Oncogene. 2007;26:3240–53.
Nakano H, Shindo M, Sakon S, Nishinaka S, Mihara M, Yagita H, Okumura K. Differential regulation of I kappa B kinase alpha and beta by two upstream kinases, NF-kappaB-inducing kinase and mitogen-activated protein kinase/ERK kinase kinase. Proc Natl Acad Sci USA. 1998;95:3537–42.
Seo HJ, Huh JE, Han JH, Jeong SJ, Jang J, Lee EO, Lee HJ, Lee HJ, Ahn KS, Kim SH. Polygoni Rhizoma inhibits inflammatory response through inactivation of nuclear factor-kappaB and mitogen activated protein kinase signaling pathways in RAW264.7 mouse macrophage cells. Phytother Res. 2012;26:239–45.
Wan F, Lenardo MJ. The nuclear signaling of NF-kappaB: current knowledge, new insights, and future perspectives. Cell Res. 2010;20:24–33.
Acknowledgments
This research was supported by a Grant-in-Aid for Scientific Research (No. 20591337) from the Japan Society for the Promotion of Science.
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Graham Wallace.
Rights and permissions
About this article
Cite this article
Rehman, M.U., Yoshihisa, Y., Miyamoto, Y. et al. The anti-inflammatory effects of platinum nanoparticles on the lipopolysaccharide-induced inflammatory response in RAW 264.7 macrophages. Inflamm. Res. 61, 1177–1185 (2012). https://doi.org/10.1007/s00011-012-0512-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-012-0512-0