Abstract
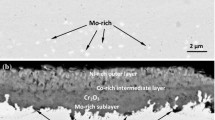
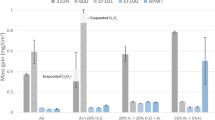
The mixed oxidation of nickel in gases containing both oxygen and chlorine has been investigated using the techniques of thermogravimetric analysis and examination of the corrosion products by scanning electron microscopy. Two regimes of corrosion behavior have been detected: one in which a protective oxide film is present and the rate of corrosion is controlled by the transport of the volatile products of the reaction between the gas phase and the oxide, and one in which the protective film is penetrated and corrosion occurs rapidly enough to result in the ignition of the specimen. The oxide corrosion products on the specimens after corrosion have characteristic morphologies, with the products of volatilization type corrosion consisting of large grains and the products of ignition type attack consisting of fine spherical particles. Ignition is a serious hazard in environments containing 0.25 pct or more Cl2 and 4 pct or more O2 by volume at temperatures between 1000 K and 1100 K, but is not produced by exposures at higher temperatures. A model based on the stability of the protective film in steady state corrosion is presented which can be used to predict the transition from volatilization to ignition type attack.
Similar content being viewed by others
References
G. H. Meier, N. Birks, F. S. Pettit, and C. S. Giggins:High Tem-perature Corrosion, R. A. Rapp, ed., National Association of Corro-sion Engineers, Houston, TX, 1983, pp. 327–35.
P. D. Miller, H. H. Krause, D. A. Vaughan, and W. K. Boyd:Corrosion, 1972, vol. 28, pp. 274–81.
M. J. McNallan, C. T. Kang, and W. W. Liang:Metall. Trans. A, 1984, vol. 15A, pp. 403–05.
M. J. Maloney and M. J. McNallan:Metall. Trans B, 1985, vol. 16B, pp. 751–61.
N. S. Jacobson, M. J. McNallan, and Y. Y. Lee:Metall. Trans. A, 1986, vol. 17A, pp. 1223–28.
P. Elliott and G. Marsh:Corrosion Science, 1984, vol. 24, pp. 397–409.
Y. Ihara, H. Ohgame, K. Sakiyama, and K. Hashimoto:Trans. Japan Inst. Metals, 1982, vol. 23, pp. 682–92.
R. P. Viswanath, D. Rein, and K. Hauffe:Werkstoffe u. Korros., 1980, vol. 31, pp. 778–82.
J. M. Oh, M. J. McNallan, G. Y. Lai, and M. F. Rothman:Metall. Trans. A, 1986, vol. 17A, pp. 1087–94.
M. J. McNallan and W. W. Liang:J. Amer. Ceram. Soc, 1981, vol. 64, pp. 302–07.
C. S. Tedmon:J. Electrochem. Soc, 1966, vol. 113, pp. 766–68.
R. A. Rapp:Metall. Trans. B, 1984, vol. 15B, pp. 195–212.
A. H. P. Skelland:Diffusional Mass Transfer, John Wiley and Sons, New York, NY, 1974, p. 117.
J. O. Hirschfelder, C. F. Curtiss, and R. B. Bird:Molecular Theory of Gases and Liquids, John Wiley and Sons, New York, NY, 1954, pp. 529–39.
R. J. Fruehan:Metall. Trans., 1972, vol. 3, pp. 2585–92.
K. Hauffe and J. Hinrichs:Werkstoffe u. Korrosion, 1970, pp. 954-65.
M. J. McNallan, W. W. Liang, J. M. Oh, and C. T. Kang:Oxid. Metals, 1982, vol. 17, pp. 371–89.
JANAF Thermochemical Tables, 1975 supplement,J. Phys. Chem. Ref. Data, 1975, vol. 4, pp. 79–80.
F. A. Elrefaie, A. Manolescu, and W. W. Smeltzer:J. Electrochem. Soc, 1985, vol. 132, pp. 2489–93.
A. Atkinson and R. I. Taylor:J. Materials Science, 1978, vol. 13, pp. 427–32.
A. A. Moosa, S. J. Rothman, and L. J. Nowicki:Oxid. Metals, 1985, vol. 24, pp. 115–32.
A. Atkinson and R. I. Taylor:Phil. Mag., 1981, vol. A43, pp. 979–98.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, Y.Y., McNallan, M.J. Ignition of nickel in environments containing oxygen and chlorine. Metall Trans A 18, 1099–1107 (1987). https://doi.org/10.1007/BF02668559
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02668559