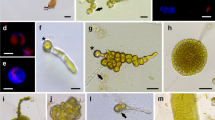
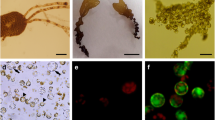
Abstract
High yields of protoplasts have been obtained from vegetative thalli of three species ofEnteromorpha by enzymatic degradation of the cell wall. Several commercial and crude enzymes prepared from the digestive system and hepatopancrease of abalone and top-shell were tested at different concentrations and combinations to evaluate the yield. Commercial enzymes in combination with either abalone or top-shell crude enzymes, consistently produced a high yield of protoplasts from all three species. High regeneration rate (85–95%) occurred in the protoplasts cultured at a density greater than 1.72 × 103 cells cm−2 at 20 and 25°C. Light intensities tested in the present study did not affect protoplast wall formation and regeneration. Protoplasts, after regenerating the cell wall, followed different types of developmental patterns under identical culture conditions. In one type some cells underwent repeated cell divisions and formed a round and oval shaped hollow thallus with a single layer of cells. In the second type many cells underwent one or two cell divisions (occasionally no division) and soon matured and discharged many motile spores, which on germination grew into normal plantlets. In the third type some cells divided irregularly to form a mass of callus-like cells (exceptE. prolifera). Culture medium supplemented with either mannitol, sorbitol, dextrose, saccharose or NaCl at higher concentrations (> 0.4 M) inhibited cell division and further differentiation in all species.
Similar content being viewed by others
References
Boyen C, Kloareg B, Polne-Fuller M, Gibor A (1990) Preparation of alginate lyases from marine molluscs for protoplast isolation in brown algae. Phycologia 29: 173–181.
Butler DM, Evans LV, Kloareg B (1990) Isolation of protoplasts from marine algae. In: Akatsuka I (ed.), An Introduction to Applied Phycology. SPB Academic Publisher, The Netherlands, 647–668.
Cheney DP (1986) Genetic engineering in seaweeds: applications and current status. Beih. Nova Hedwigia 81: 22–29.
Cronshaw J, Meyers A, Preston RD (1958) A chemical and physical investigation of the cell walls of some marine algae. Biochem. Biophys. Acta. 27: 89–103.
Dodson JR, Jr, Aronson JM (1978) Cell wall composition ofEnteromorpha intestinalis. Bot. mar. 21: 241–246.
Fujimura T, Kajiwara T (1990) Production of bioflavor by regeneration from protoplasts ofUlva pertusa (Ulvales, Chlorophyta). Hydrobiologia 204/205: 143–149.
Fujita Y, Migita S (1985) Isolation and culture of protoplasts from some seaweeds. Bull. Fac. Fish. Nagasaki Univ. 57: 39–45.
Fujita Y, Migita S (1987) Fusion of protoplasts from thalli of two different color types inPorphyra yezoensis Ueda and development of fusion products. Jap. J. Phycol. 35: 201–208.
Fujita Y, Saito M (1990) Protoplast isolation and fusion inPorphyra (Bangiales, Rhodophyta). Hydrobiologia 204/205: 161–166.
Gibor A, Polne-Fuller M, Biniaminov M, Neushul M (1980) Exploration studies of vegetative propagation of marine algae: procedure for obtaining axenic tissue. Proc. Int. Seaweed Symp. 10: 587–593.
Kajiwara T, Fujimura T, Hatanaka A (1989) Production of seaweed flavor by using plant cell culture from protoplasts of a marine green alga,Ulva pertusa. In Miyachi S, Karube I, Ishida Y (eds), Current Topics in Marine Biotechnology. The Japanese Society for Marine Biotechnology, Tokyo 239–242.
Kawashima Y, Matsumoto M, Tokuda H (1989) Protoplast isolation and regeneration of a green alga.Enteromorpha prolifera. In: Miyachi S, Karube I, Ishida Y (eds), Current Topics in Marine Biotechnology. The Japanese Society for Marine Biotechnology, Tokyo 231–234.
Liu WS, Tang YL, Liu XW, Fang TC (1984) Studies on the preparation and on the properties of sea snail enzymes. Hydrobiologia 116/117: 319–320.
Mariani P, Tolomio C, Baldan B, Braghetta P (1990) Cell wall ultrastructure and cation localization in some benthic marine algae. Phycologia 29: 253–262.
McArthur DM, Moss BC (1977) Ultrastructure of cell walls inEnteromorpha intestinalis (L.) Link. Br. phycol. J. 12: 359–368.
Millner PA, Callow ME, Evans LV (1979) Preparation of protoplasts from the green algaEnteromorpha intestinalis (L.) Link. Planta (Berlin) 147: 174–177.
Nagata T, Takebe I (1971) Plating of isolated tobacco mesophyll protoplasts on agar medium. Planta 99: 12–20.
Nicolai E, Preston RD (1952) Cell wall studies in the chlorophyceae I. A general survey of submicroscopic structure in filamentous species. Proc. R. Soc. London Ser. B 140: 244–274.
Pearce RS, Cocking EC (1973) Behaviour in culture of isolated protoplasts from ‘Paul's Scarlet’ rose suspension culture cells. Protoplasma 77: 165–180.
Polne-Fuller M, Gibor A (1984) Developmental studies inPorphyra. I. Blade differentiation inPorphyra perforata as expressed by morphology, enzymatic digestion and protoplast regeneration. J. Phycol. 20: 609–616.
Polne-Fuller M, Gibor A (1987) Tissue culture of seaweeds. In: Bird KT, Benson PH (eds), Seaweed Cultivation for Renewable Resources. Elsevier, Amsterdam, 219–239.
Polne-Fuller M, Gibor A (1990) Developmental studies inPorphyra (Rhodophyceae) III. Effect of culture conditions on wall regeneration and differentiation of protoplasts. J. Phycol. 26: 674–682.
Reddy CRK, Migita S, Fujita Y (1989) Protoplast isolation and regeneration of three species ofUlva in axenic culture. Bot. mar. 32: 483–490.
Saga N (1984) Isolation of protoplasts from edible seaweeds. Bot. Mag. Tokyo 97: 423–427.
Saga N, Polne-Fuller M, Gibor A (1986) Protoplasts from seaweeds: production and fusion. Nova Hedwigia 81: 37–43.
Waaland SA, Watson BA (1980) Isolation of a cell fusion hormone fromGriffithsia pacifica Kylin, a red alga. Planta 149: 493–497.
Yamaguchi K, Araki T, Aoki T, Tseng C, Kitamikado M (1989) Algal cell wall degrading enzymes from viscera of marine animals. Nippon Suisan Gakkaishi, 55: 105–110.
Author information
Authors and Affiliations
Additional information
author for correspondence
Rights and permissions
About this article
Cite this article
Reddy, C.R.K., Fujita, Y. Regeneration of plantlets fromEnteromorpha (Ulvales, Chlorophyta) protoplasts in axenic culture. J Appl Phycol 3, 265–275 (1991). https://doi.org/10.1007/BF00003585
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00003585