Abstract
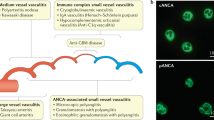
Clinical observations, including a report of neonatal vasculitis occurring in a child born from a mother with anti-neutrophil cytoplasmic antibody directed to myeloperoxidase (MPO-ANCA)-associated vasculitis, suggest a pathogenic role for ANCA. Such a role is supported by in vitro experimental data showing that ANCA can activate primed neutrophils to the production of reactive oxygen species and lytic enzymes resulting in lysis of endothelial cells. An interplay between neutrophils, the alternative pathway of complement, and MPO-ANCA resulting in systemic vasculitis including necrotizing glomerulonephritis has clearly been demonstrated in animal models. An in vivo pathogenic role of ANCA directed to proteinase 3 (PR3-ANCA) has, however, not been substantiated. In PR3-ANCA-associated vasculitis, granulomatous inflammation points to involvement of cell-mediated immunity. In vitro studies, indeed, suggest that PR3-specific Th17 CD4-positive lymphocytes are operative in lesion development. The triggering role of microbial factors is becoming more clear. In particular Staphylococcus aureus carriage and infection with Gram-negative bacteria could contribute to induction and persistence of ANCA-associated vasculitis (AAV). Insight into the pathogenic pathways involved in AAV have opened and will further open new ways to targeted treatment.


Similar content being viewed by others
References
Chen M, Kallenberg CGM (2010) ANCA-associated vasculitides—advances in pathogenesis and treatment. Nat Rev Rheumatol 6:653–664
Kallenberg CGM (2005) Churg–Strauss syndrome: just one disease entity? (Editorial). Arthritis Rheum 52:2589–2593
van der Woude FJ, Rasmussen N, Lobatto S et al (1985) Autoantibodies to neutrophils and monocytes: a new tool for diagnosis and a marker of disease activity in Wegener’s Granulomatosis. Lancet ii:425–429
Cohen Tervaert JW, van der Woude FJ, Fauci AS et al (1989) Association between active Wegener's granulomatosis and anticytoplasmic antibodies. Arch Intern Med 149:2461–2465
Cohen Tervaert JW, Huitema MG, Hené RJ et al (1990) Prevention of relapses in Wegener's granulomatosis by treatment based on antineutrophil cytoplasmic antibody titre. Lancet 336:709–711
Boomsma MM, Stegeman CA, van der Leij MJ et al (2000) Prediction of relapses in Wegener's granulomatosis by measurement of antineutrophil cytoplasmic antibody levels: a prospective study. Arthritis Rheum 43:2025–2033
Finkielman JD, Merkel PA, Schroeder D et al (2007) Antiproteinase 3 antineutrophil cytoplasmic antibodies and disease activity in Wegener granulomatosis. Ann Intern Med 147:611–619
Stegeman CA, Cohen Tervaert JW, Sluiter WJ et al (1994) Association of nasal carriage of Staphylococcus aureus and higher relapse in Wegener's granulomatosis. Ann Intern Med 120:12–17
Sanders JS, Huitema MG, Kallenberg CG, Stegeman CA (2006) Prediction of relapses in PR3-ANCA-associated vasculitis by assessing responses of ANCA titres to treatment. Rheumatology 45:724–729
Stone JH, Merkel PA, Spiera R et al (2010) Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N Engl J Med 363:221–232
Jones RB, Cohen Tervaert JW, Hauser T et al (2010) Rituximab versus cyclophosphamide in ANCA-associated renal vasculitis. N Engl J Med 363:211–220
Cartin-Ceba R, Golbin J, Keogh KA et al (2010) Ritixumab for remission induction and maintenance in ANCA-associated vasculitis: a single-center ten-year experience in 108 patients. Arthritis Rheum 62(S10):S283–S284
Bansal PJ, Tobin MC (2004) Neonatal microscopic polyangiitis secondary to transfer of maternal myeloperoxidase-antineutrophil cytoplasmic antibody resulting in neonatal pulmonary hemorrhage and renal involvement. Ann Allergy Asthma Immunol 93:398–401
Falk RJ, Terrell RS, Charles LA, Jennette JC (1990) Anti-neutrophil cytoplasmic autoantibodies induce neutrophils to degranulate and produce oxygen radicals in vitro. Proc Natl Acad Sci USA 87:4115–4119
Charles LA, Caldas ML, Falk RJ, Terrell RS, Jennette JC (1991) Antibodies against granule proteins activate neutrophils in vitro. J Leukoc Biol 50:539–546
Mulder AHL, Heeringa P, Brouwer E, Limburg PC, Kallenberg CGM (1994) Activation of granulocytes by anti-neutrophil cytoplasmic antibodies (ANCA): a FcγRII-dependent process. Clin Exp Immunol 98:270–278
Kocher M, Edberg JC, Fleit HB, Kimberly RP (1998) Antineutrophil cytoplasmic antibodies preferentially engage FcγRIIIb on human neutrophils. J Immunol 161:6909–6914
Reumaux D, Vossebeld PJ, Roos D, Verhoeven AJ (1995) Effect of tumor necrosis factor-induced integrin activation on Fc gamma receptor II-mediated signal transduction: relevance for activation of neutrophils by anti-proteinase 3 or anti-myeloperoxidase antibodies. Blood 86:3189–3195
Radford DJ, Savage CO, Nash GB (2000) Treatment of rolling neutrophils with antineutrophil cytoplasmic antibodies causes conversion to firm integrin-mediated adhesion. Arthritis Rheum 43:1337–1345
Ewert BH, Jennette JC, Falk RJ (1992) Anti-myeloperoxidase antibodies stimulate neutrophils to damage human endothelial cells. Kidney Int 41:375–383
Williams JM, Pettitt TR, Powell W, Grove J, Savage CO, Wakelam MJ (2007) Antineutrophil cytoplasm antibody-stimulated neutrophil adhesion depends on diacylglycerol kinase-catalyzed phosphatidic acid formation. J Am Soc Nephrol 18:1112–1120
Xiao H, Schreiber A, Heeringa P, Falk RJ, Jennette JC (2007) Alternative complement pathway in the pathogenesis of disease mediated by anti-neutrophil cytoplasmic autoantibodies. Am J Pathol 170:52–64
Xing CQ, Chen M, Liu G et al (2009) Complement activation is involved in renal damage in human antineutrophil cytoplasmic autoantibody associated pauci-immune vasculitis. J Clin Immunol 29:282–291
Brouwer E, Huitema MG, Klok PA, Cohen Tervaert JW, Weening JJ, Kallenberg CGM (1993) Anti-myeloperoxidase associated proliferative glomerulonephritis: an animal model. J Exp Med 177:905–914
Heeringa P, Brouwer E, Klok PA, Huitema MG, van den Born J, Weening JJ, Kallenberg CGM (1996) Autoantibodies to myeloperoxidase aggravate mild anti-glomerular-basement-membrane-mediated glomerular injury in the rat. Am J Pathol 149:1695–1706
Xiao H, Heeringa P, Hu P et al (2002) Antineutrophil cytoplasmic autoantibodies specific for myeloperoxidase cause glomerulonephritis and vasculitis in mice. J Clin Invest 110:955–963
Huugen D, Xiao H, van Esch A et al (2005) Aggravation of anti-myeloperoxidase antibody-induced glomerulonephritis by bacterial lipopolysaccharide: role of tumor necrosis factor-alpha. Am J Pathol 167:47–58
Xiao H, Heeringa P, Liu Z et al (2005) The role of neutrophils in the induction of glomerulonephritis by anti-myeloperoxidase antibodies. Am J Pathol 167:39–45
Schreiber A, Xiao H, Falk RJ, Jennette JC (2006) Bone marrow-derived cells are sufficient and necessary targets to mediate glomerulonephritis and vasculitis induced by anti-myeloperoxidase antibodies. J Am Soc Nephrol 17:3355–3364
Xiao H, Schreiber A, Heeringa P et al (2007) Alternative complement pathway in the pathogenesis of disease mediated by anti-neutrophil cytoplasmic autoantibodies. Am J Pathol 170:52–64
Huugen D, van Esch A, Xiao H et al (2007) Inhibition of complement factor C5 protects against anti-myeloperoxidase antibody-mediated glomerulonephritis in mice. Kidney Int 71:646–654
van Timmeren MM, van der Veen BS, Stegeman CA et al (2010) IgG glycan hydrolysis attenuates ANCA-mediated glomerulonephritis. J Am Soc Nephrol 21:1103–1114
Little MA, Smyth CL, Yadav R et al (2005) Antineutrophil cytoplasm antibodies directed against myeloperoxidase augment leukocyte-microvascular interactions in vivo. Blood 106:2050–2058
Pfister H, Ollert M, Frohlich LF et al (2004) Antineutrophil cytoplasmic autoantibodies against the murine homologe of proteinase 3 (Wegener autoantigen) are pathogenic in vivo. Blood 104:1411–1418
van der Geld YM, Hellmark T, Selga D et al (2007) Rats and mice immunized with chimeric human/mouse proteinase 3 produce autoantibodies to mouse Pr3 and rat granulocytes. Ann Rheum Dis 66:679–682
Franssen CFM, Stegeman CA, Kallenberg CGM et al (2000) Antiproteinase 3- and antimyeloperoxidase-associated vasculitis. Kidney Int 57:2195–2206
Abdulahad WH, van der Geld YM, Stegeman CA, Kallenberg CG (2006) Persistent expansion of CD4+ effector memory T cells in Wegener's granulomatosis. Kidney Int 70:938–947
Abdulahad WH, Kallenberg CG, Limburg PC, Stegeman CA (2009) Urinary CD4+ effector memory T cells reflect renal disease activity in antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum 60:2830–2838
Dolff S, Abdulahad WH, van Dijk MC, Limburg PC, Kallenberg CG, Bijl M (2010) Urinary T cells in active lupus nephritis show an effector memory phenotype. Ann Rheum Dis 69:2034–2041
Abdulahad WH, Stegeman CA, Limburg PC, Kallenberg CG (2008) Skewed distribution of Th17 lymphocytes in patients with Wegener’s granulomatosis in remission. Arthritis Rheum 58:2196–2205
Hemdan NY, Birkenmeier G, Wichmann G et al (2010) Interleukin-17-producing T helper cells in autoimmunity. Autoimmun Rev 9:785–792
Laan M, Cui ZH, Hoshino H et al (1999) Neutrophil recruitment by human IL-17 via C-X-C chemokine release in the airways. J Immunol 162:2347–2352
Nogueira E, Hamour S, Sawant D et al (2010) Serum IL-17 and IL-23 levels and autoantigen-specific Th17 cells are elevated in patients with ANCA-associated vasculitis. Nephrol Dial Transplant 25:2209–2217
Abdulahad WH, Stegeman CA, van der Geld YM, Doornbos-van der Meer B, Limburg PC, Kallenberg CG (2007) Functional defect of circulating regulatory CD4+ T cells in patients with Wegener's granulomatosis in remission. Arthritis Rheum 56:2080–2091
Abdulahad WH, Boots AM, Kallenberg CG (2010) FoxP3+ CD4+ T cells in systemic autoimmune diseases: the delicate balance between true regulatory T cells and effector Th-17 cells. Rheumatology (Oxford) doi:10.1093/rheumatology/keq328
Stegeman CA, Cohen Tervaert JW, de Jong PE, Kallenberg CGM (1996) Trimethoprim-sulfamethoxazole (co-trimoxazole) for the prevention of relapses of Wegener's granulomatosis. N Engl J Med 335:16–20
Popa ER, Stegeman CA, Bos NA, Kallenberg CGM, Tervaert JWC (2003) Staphylococcal superantigens and T cell expansions in Wegener's granulomatosis. Clin Exp Immunol 132:496–504
Popa ER, Stegeman CA, Abdulahad WH et al (2007) Staphylococcal toxic-shock-syndrome-toxin-1 as a risk factor for disease relapse in Wegener's granulomatosis. Rheumatol Oxf 46:1029–1033
Voswinkel J, Mueller A, Kraemer JA et al (2006) B lymphocyte maturation in Wegener's granulomatosis: a comparative analysis of VH genes from endonasal lesions. Ann Rheum Dis 65:859–864
Voswinkel J, Assmann G, Held G et al (2008) Single cell analysis of B lymphocytes from Wegener's granulomatosis: B cell receptors display affinity maturation within the granulomatous lesions. Clin Exp Immunol 154:339–345
Tadema H, Abdulahad WH, Lepse N, Stegeman CA, Kallenberg CG, Heeringa P (2010) Bacterial DNA motifs trigger ANCA production in ANCA-associated vasculitis in remission. Rheumatology (Oxford) doi:10.1093/rheumatology/keq375
Kain R, Exner M, Brandes R et al (2008) Molecular mimicry in pauci-immune focal necrotizing glomerulonephritis. Nat Med 14:1088–1096
Abdulahad WH, Stegeman CA, Kallenberg CG (2009) Review article: the role of CD4(+) T cells in ANCA-associated vasculitis. Nephrology 14:26–32
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kallenberg, C.G.M. Pathogenesis of ANCA-Associated Vasculitis, an Update. Clinic Rev Allerg Immunol 41, 224–231 (2011). https://doi.org/10.1007/s12016-011-8258-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-011-8258-y