Abstract
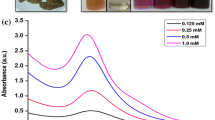
Gold nanoparticles (AuNPs) were synthesized by sonication using ethanolic leaf extract of Andrographis paniculata. We investigated the optimum parameters for AuNP synthesis and functionalization with polycaprolactone-gelatin (PCL-GL) composites. The AuNPs were characterized with various biophysical techniques such as TEM, XRD, FT-IR and EDX spectroscopy. TEM images showed that nanoparticles were spherical in shape with a size range from 5 to 75 nm. EDX analysis revealed the presence of molecular oxygen and carbon on the surface of AuNPs. The synthesized AuNPs were tested for their effect on HeLa (human cervical cancer) and MCF-7 (human breast cancer) cell lines and found to be nontoxic and biocompatible, which are potential carriers for hydrophobic drugs.
Similar content being viewed by others
References
Giljohann D A, Seferos D S, Daniel WL, et al. Gold nanoparticles for biology and medicine. Angewandte Chemie International Edition, 2010, 49(19): 3280–3294
Ghosh P, Han G, De M, et al. Gold nanoparticles in delivery applications. Advanced Drug Delivery Reviews, 2008, 60(11): 1307–1315
Lewis L N. Chemical catalysis by colloids and clusters. Chemical Reviews, 1993, 93(8): 2693–2730
Lee K-B, Kim E-Y, Mirkin C A, et al. The use of nanoarrays for highly sensitive and selective detection of human immunodeficiency virus type 1 in plasma. Nano Letters, 2004, 4(10): 1869–1872
Bhattacharya R, Mukherjee P. Biological properties of “naked” metal nanoparticles. Advanced Drug Delivery Reviews, 2008, 60(11): 1289–1306
Uboldi C, Bonacchi D, Lorenzi G, et al. Gold nanoparticles induce cytotoxicity in the alveolar type-II cell lines A549 and NCIH441. Particle and Fibre Toxicology, 2009, 6: 18 (12 pages)
Narayanan K B, Sakthivel N. Green synthesis of biogenic metal nanoparticles by terrestrial and aquatic phototrophic and heterotrophic eukaryotes and biocompatible agents. Advances in Colloid and Interface Science, 2011, 169(2): 59–79
Siripong P, Kongkathip B, Preechanukool K, et al. Cytotoxic diterpenoid constituents from Andrographis paniculata Nees. leaves. Journal of the Science Society of Thailand, 1992, 18(4): 187–194
Singha P K, Roy S, Dey S. Antimicrobial activity of Andrographis paniculata. Fitoterapia, 2003, 74(7–8): 692–694
Dua V K, Ojha V P, Roy R, et al. Anti-malarial activity of some xanthones isolated from the roots of Andrographis paniculata. Journal of Ethnopharmacology, 2004, 95(2–3): 247–251
Thisoda P, Rangkadilok N, Pholphana N, et al. Inhibitory effect of Andrographis paniculata extract and its active diterpenoids on platelet aggregation. European Journal of Pharmacology, 2006, 553(1–3): 39–45
Yang L, Wu D F, Luo K W, et al. Andrographolide enhances 5-fluorouracil-induced apoptosis via caspase-8-dependent mitochondrial pathway involving p53 participation in hepatocellular carcinoma (SMMC-7721) cells. Cancer Letters, 2009, 276(2): 180–188
Puri A, Saxena R, Saxena R P, et al. Immunostimulant agents from Andrographis paniculata. Journal of Natural Products, 1993, 56(7): 995–999
Sheeja K, Kuttan G. Effect of Andrographis paniculata as an adjuvant in combined chemo-radio and whole body hyperthermia treatment — a preliminary study. Immunopharmacology and Immunotoxicology, 2008, 30(1): 181–194
Madav S, Tandan S K, Lal J, et al. Anti-inflammatory activity of andrographolide. Fitoterapia, 1996, 67(5): 452–458
Chang R S, Yeung H W. Inhibition of growth of human immunodeficiency virus in vitro by crude extracts of Chinese medicinal herbs. Antiviral Research, 1988, 9(3): 163–175
Ahmad M, Asmawi M Z. Some pharmacological effects of aqueous extract of Andrographis paniculata Nees. In: Gan E K, ed. The International Conference on the Use of’ Traditional Medicine & Other Natural Products in Health Care (Abstract). Penang, Malaysia: School of Pharmaceutical Sciences, University of Science Malaysia, 1993
Jarukamjorn K, Nemoto N. Pharmacological aspects of Andrographis paniculata on health and its major diterpenoid constituent Andrographolide. Journal of Health Science, 2008, 54(4): 370–381
Kirtikar K R, Basu B D. Indian medicinal plants. Periodical Experts, 1975, 3: 1884–1886
Nazimudeen S K, Ramaswamy S, Kameswaran L. Effect of Andrographis paniculata on snake venom induced death and its mechanism. Indian Journal of Pharmaceutical Sciences, 1978, 40(4): 132–133
Suslick K S. Sonochemistry. Science, 1990, 247(4949): 1439–1445
Maisonhaute E, Prado C, White P C, et al. Surface acoustic cavitation understood via nanosecond electrochemistry. Part III: Shear stress in ultrasonic cleaning. Ultrasonics Sonochemistry, 2002, 9(6): 297–303
Mason T J, Cobley A J, Graves J E, et al. New evidence for the inverse dependence of mechanical and chemical effects on the frequency of ultrasound. Ultrasonics Sonochemistry, 2011, 18(1): 226–230
Nune S K, Chanda N, Shukla R, et al. Green nanotechnology from tea: phytochemicals in tea as building blocks for production of biocompatible gold nanoparticles. Journal of Materials Chemistry, 2009, 19(19): 2912–2920
Mody V V, Siwale R, Singh A, et al. Introduction to metallic nanoparticles. Journal of Pharmacy and Bioallied Sciences, 2010, 2(4): 282–289
Babu P J, Sharma P, Borthakur B B, et al. Synthesis of gold nanoparticles using Mentha arvensis leaf extract. International Journal of Green Nanotechnology: Physics and Chemistry, 2010, 2(2): 62–68
Shankar S S, Rai A, Ahmad A, et al. Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. Journal of Colloid and Interface Science, 2004, 275(2): 496–502
Kannan P, Abraham John S. Synthesis of mercaptothiadiazolefunctionalized gold nanoparticles and their self-assembly on Au substrates. Nanotechnology, 2008, 19(8): 085602
Hiremath J G, Devi V K. Preparation and in vitro characterization of poly(epsilon-caprolactone)-based tamoxifen citrate-loaded cylindrical subdermal implant for breast cancer. Asian Journal of Pharmaceutics, 2011, 5(1): 9–14
Nagahama H, Maeda H, Kashiki T, et al. Preparation and characterization of novel chitosan/gelatin membranes using chitosan hydrogel. Carbohydrate Polymers, 2009, 76(2): 255–260
Babu P J, Sharma P, Kalita M C, et al. Green synthesis of biocompatible gold nanoparticles using Fagopyrum esculentum leaf extract. Frontiers of Materials Science, 2011, 5(4): 379–387
Shankar S S, Rai A, Ankamwar B, et al. Biological synthesis of triangular gold nanoprisms. Nature Materials, 2004, 3(7): 482–488
Song J Y, Kim B S. Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess and Biosystems Engineering, 2009, 32(1): 79–84
Shah Mohammadi M, Ahmed I, Marelli B, et al. Modulation of polycaprolactone composite properties through incorporation of mixed phosphate glass formulations. Acta Biomaterialia, 2010, 6(8): 3157–3168
Ghasemi-Mobarakeh L, Prabhakaran M P, Morshed M, et al. Electrospun poly(ɛ-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials, 2008, 29(34): 4532–4539
Ki C S, Baek D H, Gang K D, et al. Characterization of gelatin nanofiber prepared from gelatin-formic acid solution. Polymer, 2005, 46(14): 5094–5102
Krimm S, Bandekar J. Vibrational spectroscopy and conformation of peptides, polypeptides, and proteins. Advances in Protein Chemistry, 1986, 38: 181–364
Sahoo R, Sahoo S, Sahoo S, et al. Synthesis and characterization of polycaprolactone-gelatin nanocomposites for control release anticancer drug paclitaxel. European Journal of Scientific Research, 2011, 48(3): 527–537
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Babu, P.J., Saranya, S., Sharma, P. et al. Gold nanoparticles: sonocatalytic synthesis using ethanolic extract of Andrographis paniculata and functionalization with polycaprolactone-gelatin composites. Front. Mater. Sci. 6, 236–249 (2012). https://doi.org/10.1007/s11706-012-0175-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11706-012-0175-3