Abstract
Introduction
The impact of the number of lymph node (LN) evaluated pathologically on accurate staging is unknown. Our primary aim was to determine a minimum number of evaluated LN needed to provide accurate staging of pancreatic cancer.
Methods
Four hundred ninety-nine patients underwent a curative pancreatectomy for pancreatic adenocarcinoma cancer from 1981–2007. The probability of understaging a patient as N0 was estimated based on the number of LN evaluated. The prognostic value of LN ratio (LNR) was assessed.
Results
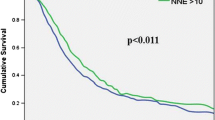
Survival for node-negative (pN0) patients with <11 LN examined was worse than for pN0 patients with ≥11 LNs with a hazard ratio (95 % CI) of 1.33 (1.1–1.7, p = 0.01) with 3-year survivals of 32 vs. 50%, respectively. Three-year survival for pN1 patients with <11 nodes evaluated was similar to pN1 patients with ≥11 nodes (25 vs. 30%). LNR ≥ 0.17 predicted worse survival with hazard ratio of 1.76 (1.3–2.4, p = 0.001) than LNR < 0.17; 3-year survivals were 37 vs. 19%.
Conclusion
Patients with “N0” disease with <11 LN evaluated pathologically have worse survival, suggesting that metastatic nodes were missed by evaluating too few nodes. For pN1 patients, LNR stratifies survival of patient cohorts more accurately. Adequate staging of pancreatic cancer requires pathologic evaluation of ≥11 LNs.



Similar content being viewed by others
References
Huang CM, Lin JX, Zheng CH, et al. Prognostic impact of metastatic lymph node ratio on gastric cancer after curative distal gastrectomy. World J Gastroenterol. 2010; 16: 2055–60.
Schiffman SC, McMasters KM, Scoggins CR, Martin RC, Chagpar AB. Lymph node ratio: a proposed refinement of current axillary staging in breast cancer patients. Journal of the American College of Surgeons. 2011; 213: 45–52.
Greenberg R, Itah R, Ghinea R, Sacham-Shmueli E, Inbar R, Avital S. Metastatic lymph node ratio (LNR) as a prognostic variable in colorectal cancer patients undergoing laparoscopic resection. Techniques in Coloproctology. 2011; 15: 273–9.
Japanese Gastric Cancer Society. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 1998; 1: 10–24.
Isaji S, Kawarada Y, Uemoto S. Classification of pancreatic cancer: comparison of Japanese and UICC classifications. Pancreas. 2004; 28: 231–4.
Pawlik TM, Abdalla EK, Barnett CC, et al. Feasibility of a randomized trial of extended lymphadenectomy for pancreatic cancer. Arch Surg. 2005; 140: 584–9; discussion 9–91.
Yeo CJ, Cameron JL, Lillemoe KD, et al. Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma, part 2: randomized controlled trial evaluating survival, morbidity, and mortality. Annals of Surgery. 2002; 236: 355–66; discussion 66–8.
Schwarz RE, Smith DD. Extent of lymph node retrieval and pancreatic cancer survival: information from a large US population database. Annals of Surgical Oncology. 2006; 13: 1189–200.
Tomlinson JS, Jain S, Bentrem DJ, et al. Accuracy of staging node-negative pancreas cancer: a potential quality measure. Arch Surg. 2007; 142: 767–23; discussion 73–4.
Schoenleber SJ, Schnelldorfer T, Wood CM, Qin R, Sarr MG, Donohue JH. Factors influencing lymph node recovery from the operative specimen after gastrectomy for gastric adenocarcinoma. J Gastrointest Surg. 2009; 13: 1233–7.
Berger AC, Watson JC, Ross EA, Hoffman JP. The metastatic/examined lymph node ratio is an important prognostic factor after pancreaticoduodenectomy for pancreatic adenocarcinoma. The American surgeon. 2004; 70: 235–40; discussion 40.
House MG, Gonen M, Jarnagin WR, et al. Prognostic significance of pathologic nodal status in patients with resected pancreatic cancer. J Gastrointest Surg. 2007; 11: 1549–55.
Pawlik TM, Gleisner AL, Cameron JL, et al. Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer. Surgery. 2007; 141: 610–8.
Riediger H, Keck T, Wellner U, et al. The lymph node ratio is the strongest prognostic factor after resection of pancreatic cancer. J Gastrointest Surg. 2009; 13: 1337–44.
Sierzega M, Popiela T, Kulig J, Nowak K. The ratio of metastatic/resected lymph nodes is an independent prognostic factor in patients with node-positive pancreatic head cancer. Pancreas. 2006; 33: 240–5.
Khan S, Sclabas G, Reid-Lombardo K, et al. Does body mass index/morbid obesity influence outcome in patients who undergo pancreatoduodenectomy for pancreatic adenocarcinoma? J Gastrointest Surg. 14: 1820–5.
Schnelldorfer T, Ware AL, Sarr MG, et al. Long-term survival after pancreatoduodenectomy for pancreatic adenocarcinoma: is cure possible? Annals of surgery. 2008; 247: 456–62.
Nitecki SS, Sarr MG, Colby TV, van Heerden JA. Long-term survival after resection for ductal adenocarcinoma of the pancreas. Is it really improving? Annals of surgery. 1995; 221: 59–66.
Gonen M, Schrag D, Weiser MR. Nodal staging score: a tool to assess adequate staging of node-negative colon cancer. J Clin Oncol. 2009; 27: 6166–71.
Altman DG, Lausen B, Sauerbrei W, Schumacher M. Dangers of using “optimal” cutpoints in the evaluation of prognostic factors. Journal of the National Cancer Institute. 1994; 86: 829–35.
Contal C, O'Quigley J. An application of changepoint methods in studying the effect of age on survival in breast cancer. Computational Statistics and Data Analysis. 1999; 30: 253–70.
R Development Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing 2009.
Vinh-Hung V, Nguyen NP, Cserni G, et al. Prognostic value of nodal ratios in node-positive breast cancer: a compiled update. Future oncology (London, UK). 2009; 5: 1585–603.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huebner, M., Kendrick, M., Reid-Lombardo, K.M. et al. Number of Lymph Nodes Evaluated: Prognostic Value in Pancreatic Adenocarcinoma. J Gastrointest Surg 16, 920–926 (2012). https://doi.org/10.1007/s11605-012-1853-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-012-1853-2