Abstract
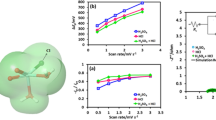
The present work was performed in order to evaluate sulfamic acid as the supporting electrolyte for VO2+/VO2 + redox couple in vanadium redox flow battery. The oxidation process of VO2+ has similar electrochemical kinetics compared with the reduction process of VO2 +. The exchange current density and standard rate constant of VO2+/VO2 + redox reaction on a graphite electrode in sulfamic acid are determined as 7.6 × 10−4 A cm−2 and 7.9 × 10−5 cm s−1, respectively. The energy efficiency of the cell employing sulfamic acid as supporting electrolyte in the positive side can reach 75.87 %, which is adequate for redox flow battery applied in energy storage. The addition of NH4 + to the positive electrolyte can enhance the electrochemical performance of the cell, with larger discharge capacity and energy efficiency. The preliminary exploration shows that the vanadium sulfamate electrolyte is promising for vanadium redox flow battery and is worthy of further study.








Similar content being viewed by others
References
Li LY, Kim S, Wang W, Vijayakumar M, Nie ZM, Chen BW, Zhang JL, Xia GG, Hu JZ, Graff G, Liu J, Yang ZG (2011) Adv Energy Mater 1:394
Xiong F, Zhou D, Xie Z, Chen Y (2012) Appl Energy 99:291
Suzuki Y, Koyanagi A, Kobayashi M, Shimada R (2005) Energy 30:2128
Hartikainen T, Mikkonen R, Lehtonen J (2007) Appl Energy 84:29
Cedzynska K (1995) Electrochim Acta 40:971
Oriji G, Katayama Y, Miura T (2004) Electrochim Acta 49:3091
Xia X, Liu HT, Liu Y (2002) J Electrochem Soc 149:A426
Wen YH, Zhang HM, Qian P, Zhou HT, Zhao P, Yi BL, Yang YS (2006) J Electrochem Soc 153:A929
Xue FQ, Wang YL, Wang WH, Wang XD (2008) Electrochim Acta 53:6636
Tang C, Zhou DB (2012) Electrochim Acta 65:179
Rahman F, Skyllas-Kazacos M (2009) J Power Sources 189:1212
Sun B, Skyllas-Kazacos M (1992) Electrochim Acta 37:1253
Kim S, Yan J, Schwenzer B, Zhang J, Li L, Liu J, Yang Z, Hickner MA (2010) Electrochem Commun 12:1650
Skyllas-Kazacos M, Rychcik M, Robins RG, Fane AG, Green MA (1986) J Electrochem Soc 133:1057
Wei Y, Fang B, Arai T, Kumagai M (2005) J Appl Electrochem 35:561
Leung PK, Ponce de León C, Low CTJ, Walsh FC (2011) Electrochim Acta 56:2145
Peng S, Wang NF, Wu XJ, Liu SQ, Fang D, Liu YN, Huang KL (2012) Int J Electrochem Sci 7:643
Kim S, Vijayakumar M, Wang W, Zhang J, Chen B, Nie Z, Chen F, Hu J, Li L, Yang Z (2011) Phys Chem Chem Phys 13:18186
Bober P, Trchová M, Prokeš J, Varga M, Stejskal J (2011) Electrochim Acta 56:3580
Ameen S, Ali V, Zulfequar M, Mazharul Haq M, Husain M (2007) Curr Appl Phys 7:215
Yadav JS, Purushothama Rao P, Sreenu D, Rao RS, Naveen Kumar V, Nagaiah K, Prasad AR (2005) Tetrahedron Lett 46:7249
Rostami A, Tavakoli A (2011) Chin Chem Lett 22:1317
Kanda FA, King AJ (1951) J Am Chem Soc 73:2315
Heravi MM, Alinejhad H, Bakhtiari K, Oskooie HA (2010) Mol Divers 14:621
Heravi MM, Baghernejad B, Oskooie HA (2009) Curr Org Chem 13:1002
Kazacos M, Cheng M, Skyllas-Kazacos M (1990) J Appl Electrochem 20:463
Zhong S, Skyllas-Kazacos M (1992) J Power Sources 39:1
Wu X, Wang J, Liu S, Wu X, Li S (2011) Electrochim Acta 56:10197
Xie Z, Zhou D, Xiong F, Zhang S, Huang K (2011) J Rare Earth 29:567
Wu X, Liu S, Wang N, Peng S, He Z (2012) Electrochim Acta 78:475
Fang B, Iwasa S, Wei Y, Arai T, Kumagai M (2002) Electrochim Acta 47:3971
Bard AJ, Faulkner LR (2001) Electrochemical methods: fundamental and applications. Wiley, New York, p 231
Bard AJ, Faulkner LR (2001) Electrochemical methods: fundamental and applications. Wiley, New York, p 236
Acknowledgments
This work was financially supported by the Major State Basic Research Development Program of China (973 Program, No. 2010CB227201) and Innovation Project of College Students of Central South University (No. CL12136).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, Z., He, Y., Chen, C. et al. Study of the electrochemical performance of VO2+/VO2 + redox couple in sulfamic acid for vanadium redox flow battery. Ionics 20, 949–955 (2014). https://doi.org/10.1007/s11581-013-1051-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-013-1051-6