Abstract
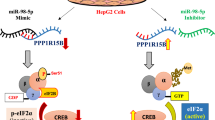
Heparan sulfate proteoglycan (HSPG), such as glypican, plays a role as a co-receptor for growth factor to influence cells proliferation. However the mechanism is still vague. Micro-RNAs (miRNAs) regulate cell proliferation. Their capacity to direct the translation and stability of targeted transcripts can dramatically influence cellular physiological function. To explore how the function of glypican is regulated involved in cell proliferation, glypican-4 was chosen with a bioinformatics search identifying targeting seed sequences for miR-125a within the 3′-untranslated regions (3′UTR). Indeed, luciferase constructs containing the 3′UTR of glypican-4 demonstrated around 54 % less activity in miR-125a expressing cells relative to the controls. The expression of glypican-4 at both the transcript and protein level was down-regulated by transition trasfection of miR-125a in the human embryonic kidney cell line 293T (HEK293T). Although cell proliferation of HEK293T was not influenced by the silence of glypican-4, DNA synthesis in response to FGF2 in the cells was attenuated by knockdown of glypican-4 using siRNA technique. Further study showed that phosphorylation of ERK1/2 and AKT was suppressed by overexpressing miR-125a, whereas the suppressed MAPK and AKT signaling could be recovered by anti-miR-125a treatment. Both DNA synthesis and cell proliferation were impaired by the inhibitor of ERK1/2 signaling. MTT assay demonstrated that the cell proliferation was impaired by miR-125a overexpression, however, rescued by anti-miR-125a in HEK293T cells. These results disclosed new function of miR-125a by targeting gene glypican-4 in cell growth process and illustrated the feasibility of using miRNAs as a therapeutic strategy to suppress cells proliferation.





Similar content being viewed by others
Abbreviations
- HSPG:
-
Heparan sulfate proteoglycan
- miRNAs:
-
Micro-RNAs
- 3′UTR:
-
3′-untranslated regions
References
Bishop, J.R., Schuksz, M., Esko, J.D.: Heparan sulphate proteoglycans fine-tune mammalian physiology. Nature 446, 1030–1037 (2007)
Qiu, H., Yang, B., Pei, Z.C., Zhang, Z., Ding, K.: WSS25 inhibits growth of xenografted hepatocellular cancer cells in nude mice by disrupting angiogenesis via blocking bone morphogenetic protein (BMP)/Smad/Id1 signaling. J. Biol. Chem. 285, 32638–32646 (2010)
Ding, K., Lopez-Burks, M., Sánchez-Duran, J.A., Korc, M., Lander, A.D.: Growth factor-induced shedding of syndecan-1 confers glypican-1 dependence on mitogenic responses of cancer cells. J. Cell. Biol. 171, 729–738 (2005)
Fransson, L.A.: Glypicans. Int. J. Biol. Cell Biol. 35, 125–129 (2003)
De Cat, B., David, G.: Developmental roles of the glypicans. Semin. Cell. Dev. Biol. 12, 117–125 (2001)
Bernfield, M., Gotte, M., Park, P.W., Reizes, O., Fitzgerald, M.L., Lincecum, J., Zako, M.: Annu. Rev. Biochem. 68, 729–777 (1999)
Flimus, J.: Glypicans in growth control and cancer. Glycobiology 11, 19R–23R (2001)
Hagihara, K., Watanabe, K., Chun, J., Yamaguchi, Y.: Glypican-4 is an FGF2-binding heparan sulfate proteoglycan expressed in neural precursor cells. Dev. Dyn. 219, 353–367 (2000)
Karihaloo, A., Kale, S., Rosenblum, N.D., Cantley, L.G.: Hepatocyte growth factor-mediated renal epithelial branching morphogenesis is regulated by glypican-4 expression. Mol. Cell. Biol. 24, 8745–8752 (2004)
Gibbings, D., Voinnet, O.: Control of RNA silencing and localization by endolysosomes. Trends Cell Biol. 20, 491–501 (2010)
Edwards, J.K., Pasqualini, R., Arap, W., Calin, G.A.: MicroRNAs and ultraconserved genes as diagnostic markers and therapeutic targets in cancer and cardiovascular diseases. J. Cardiovasc. Transl. Res. 3, 271–279 (2010)
Carrington, J.C., Ambros, V.: Role of microRNAs in plant and animal development. Science 301, 336–338 (2003)
Lee, R.C., Feinbaum, R., Ambros, L.V.: The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75, 843–854 (1993)
Wang, V., Wu, W.: MicroRNA: A new player in breast cancer development. J. Cancer Mol. 3, 133–138 (2007)
Bartel, D.P.: MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116, 281–297 (2004)
Guo, X., Wu, Y., Hartley, R.S.: MicroRNA-125a represses cell growth by targeting HuR in breast cancer. RNA Biol. 6, 575–583 (2009)
Cowden Dahl, K.D., Dahl, R., Kruichak, J.N., Hudson, L.G.: The epidermal growth factor receptor responsive miR-125a represses mesenchymal morphology in ovarian cancer cells. Neoplasia 11, 1208–1215 (2009)
Lee, Y., Kim, M., Han, J., Yeom, K.H., Lee, S., Baek, S.H., Kim, V.N.: MicroRNA genes are transcribed by RNA polymerase II. EMBO J. 23, 4051–4060 (2004)
Selbach, M., Schwanhäusser, B., Thierfelder, N., Fang, Z., Khanin, R., Rajewsky, N.: Widespread changes in protein synthesis induced by microRNAs. Nature 455, 58–63 (2008)
Ott, V.L., Rapraeger, A.C.: Tyrosine phosphorylation of syndecan-1 and −4 cytoplasmic domains in adherent B82 fibroblasts. J. Biol. Chem. 273, 35291–35298 (1998)
Pellegrini, L., Burke, D.F., Delft, F., Mulloy, V.B., Blundell, T.L.: Crystal structure of fibroblast growth factor receptor ectodomain bound to ligand and heparin. Nature 407, 1029–1034 (2000)
Delehedde, M., Seve, M., Sergeant, N., Wartelle, I., Lyon, M., Rudland, P.S., Fernig, D.G.: Fibroblast growth factor-2 stimulation of p42/44MAPK phosphorylation and IkappaB degradation is regulated by heparan sulfate/heparin in rat mammary fibroblasts. J. Biol. Chem. 275, 33905–33910 (2000)
Jin, K., Mao, X.O., Del Rio, G.G., Jin, L., Greenberg, D.A.: Heparin-binding epidermal growth factor-like growth factor stimulates cell proliferation in cerebral cortical cultures through phosphatidylinositol 3′-kinase and mitogen-activated protein kinase. J. Neurosci. Res. 81, 497–505 (2005)
Garzon, R., Volinia, S., Liu, C.G., Fernandez-Cymering, C., Palumbo, T., Pichiorri, F., Fabbri, M., Coombes, K., Alder, H., Nakamura, T., Flomenberg, N., Marcucci, G., Calin, G.A., Kornblau, S.M., Kantarjian, H., Bloomfield, C.D., Andreeff, M., Croce, C.M.: MicroRNA signatures associated with cytogenetics and prognosis in acute myeloid leukemia. Blood 111, 3183–3189 (2008)
Acknowledgments
This work was supported by National Natural Science Foundation of China (NSFC) (81171914) and National Science Fund for Distinguished Young Scholars (81125025) in China.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Feng, C., Li, J., Ruan, J. et al. MicroRNA-125a inhibits cell growth by targeting glypican-4. Glycoconj J 29, 503–511 (2012). https://doi.org/10.1007/s10719-012-9387-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-012-9387-0