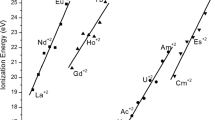
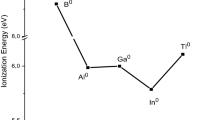
Abstract
The usefulness of isoelectronic series (same number of total electrons and atoms and of valence electrons) across Periods is often overlooked. Here we show the ubiquitousness of isoelectronic sets by means of matrices, arrays, and sequential series. Some of these series have not previously been identified. In addition, we recommend the use of the term valence-isoelectronic for species which differ in the number of core electrons and pseudo-isoelectronic for matching (n) and (n + 10) species.
Similar content being viewed by others
References
Bickelhaupt, F.M., DeKock, R.L., Baerends, E.J.: The short N–F bond in N2F+ and how Pauli repulsion influences bond lengths. J. Am. Chem. Soc. 124, 1500–1505 (2002)
Carroll, L.: More Annotated Alice: Alice’s Adventures in Wonderland and Through the Looking Glass and What Alice Found There, with notes by M. Gardner, p. 253. Random House, New York (1990)
Elliott, B.M., Boldyrev, A.I.: Ozonic acid and its ionic salts: Ab initio probing of the O4 2− dianion. Inorg. Chem. 43, 4109–4111 (2004)
Greenwood, N.N., Earnshaw, A.: Chemistry of the Elements, 2nd edn. Butterworth-Heinemann, Oxford (1997)
Huheey, J.E., Keitler, E.A., Keitler, R.L.: Inorganic Chemistry: Principles of Structure and Reactivity, p. 640. HarperCollins, New York (1993)
LaCagnin, L.B., et al.: The carbon dioxide anion radical adduct in the perfused rat liver. Mol. Pharmacol. 33, 351–357 (1988)
Langmuir, I.: The arrangement of electrons in atoms and molecules. J. Am. Chem. Soc. 41, 868–934 (1919)
Larsson, M., et al.: X-ray photoelectron, auger electron and ion fragment spectra of O2 and potential curves of O2 2+. J. Phys. B: At. Mol. Opt. Phys. 23, 1175–1195 (1990)
Moody, B.: Comparative Inorganic Chemistry, 2nd edn, p. 51. Edward Arnold, London (1969)
Penney, W.G., Sutherland, G.B.B.M.: The relation between the form, force constants and vibrational frequencies of triatomic species. Proc. R. Soc. (London) A156, 654 (1936)
Rayner-Canham, G.W., Overton,T.L.: Descriptive Inorganic Chemistry, 4th edn, p. 30. W.H. Freeman Publishing Co., New York (2006). The metals next to the semi-metals show differences in behaviour from ‘typical’ metals. In particular, they are amphoteric, readily forming oxyanions in basic solution, such as stannates. For this reason, one can use the term weak metals in contrast to the supermetals, a designation sometimes used for the low-density, highly reactive, alkali metals
Rayner-Canham, G., Oldford, M.: The chemical ‘knight’s move’ relationship: what is its significance? Foundations Chem. 9, 119–125 (2007)
Rouvray, D.H., King, R.B. (eds.): The Periodic Table: Into the 21st Century. Research Studies Press Ltd, Hertfordshire, England (2004)
Sima, J.: Isoelectronic series: the stability of their members. J. Chem. Educ. 72, 310 (1995)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rayner-Canham, G. Isoelectronic series: a fundamental periodic property. Found Chem 11, 123–129 (2009). https://doi.org/10.1007/s10698-008-9055-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10698-008-9055-4