Abstract
Background
Lubiprostone has been used to treat constipation through its effects to stimulate Cl− secretion, resulting in water and electrolyte secretion.
Aim
Potential associated changes in intestinal mucus and the colonizing bacteria (microbiome) have not been studied. As mucus obstructions may play a role in cystic fibrosis, the hypothesis that lubiprostone alters intestinal mucus and the microbiome was investigated.
Methods
Ion transport studies were performed ex vivo. For mucus and microbiome studies, mice were gavaged daily with lubiprostone or vehicle. Mucin from intestinal sections was analyzed in Carnoy’s fixed tissues stained with Alcian blue. Microbiome composition was analyzed by 16S rRNA gene-based sequencing.
Results
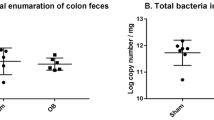
Lubiprostone stimulated short circuit current in all mouse intestinal segments after both serosal and mucosal additions, albeit at lower concentrations in the latter. Current was Cl-dependent and blocked by mucosal diphenylcarboxylic acid, serosal bumetanide, and serosal Ba++. The CFTR inhibitor CFTRinh172 had a marginal effect. Mucus near epithelial cells (inner layer mucus) was not present in the small intestine of any mice. Proximal colon inner mucus layer was thicker in ∆F/∆F compared with +/∆F and +/+ mice. Lubiprostone decreased inner mucus layer thickness in both proximal and distal colon of all mice. Furthermore, lubiprostone altered the intestinal microbiome by increasing abundance of Lactobacillus and Alistipes.
Conclusions
Lubiprostone activates non-CFTR Cl− secretion and alters the colonic inner mucus layer, which is associated with changes in the composition of the enteric microbiome.




Similar content being viewed by others
References
Rowe SM. Cystic fibrosis. New Eng J Med. 2005;352:1992–2001.
Gouyer V, Gottrand F, Desseyn JL. The extraordinarily complex but highly structured organization of intestinal mucus-gel unveiled in multicolor images. PLOS One. 2011;6:e18761.
Gilger MA. Cystic fibrosis: nutritional and intestinal disorders. Pediatr Pulmonol. 1995;20:56–57.
Werlin SL, Benuri-Silbiger I, Kerem E, et al. Evidence of intestinal inflammation in patients with cystic fibrosis. J Pediatr Gastroenterol Nut. 2010;51:304–308.
Saiman L, Siegel J. Infection control in cystic fibrosis. Clin Microbiol Rev. 2004;17:57–71.
Fridge JL, Conrad C, Gerson L, et al. Risk factors for small bowel bacterial overgrowth in cystic fibrosis. J Pediatr Gastroenterol Nut. 2007;44:212–218.
Eckburg PB, Bil EM, Bernstein CN, et al. Divesity of the human intestinal microbial flora. Science. 2005;308:1635–1638.
Lacy BE, Levy LC. Lubiprostone: a chloride channel activator. J Clin Gastroenterol. 2007;41:345–351.
Ueno R, Osama H, Habe T. Oral SPI-0211 increases intestinal fluid secretion and chloride concentration without altering serum electrolyte levels. Gastroenterology. 2004;126:A298.
Cuppoletti J, Malinowska DH, Tewari KP, et al. SPI-0211 activates T84 cell chloride transport and recombinant human ClC-2 chloride currents. Am J Physiol. 2004;287:C1173–C1183.
Fei G, Wang YZ, Liu S, et al. Stimulation of mucosal secretion by lubiprostone (SPI-0211) in Guinea pig small intestine and colon. Am J Physiol Gastrointest Liver Physiol. 2009;296:823–832.
Mizumori M, Akiba Y, Kaunitz JD. Lubiprostone stimulates duodenal bicarbonate secretion in rats. Dig Dis Sci. 2009;54:2063–2069.
Bijvelds MJ, Bot AG, Escher JC, De Jonge HR. Activation of intestinal Cl− secretion by lubiprostone requires the cystic fibrosis transmembrane conductance regulator. Gastroenterology. 2009;137:976–985.
Ao M, Venkatasubramanian J, Boonkaewwan C, Benya R, Rao M. Lubiprostone activates Cl− secretion via cAMP signaling and increases membrane CFTR in the human colon carcinoma cell line, T84. Dig Dis Sci. 2011;56:339–351.
MacVinish LJ, Cope G, Ropenga A, Cuthbert AW. Chloride transporting capability of Calu-3 epithelia following persistent knockdown of the cystic fibrosis transmembrane conductance regulator, CFTR. Br J Pharmacol. 2007;150:1055–1065.
Bao HF, Liu L, Self J, Duke BJ, Ueno R, Eaton DC. A synthetic prostone activates apical chloride channels in A6 epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2008;295:G234–G251.
Zeiher BG, Eichwald E, Zabner J, et al. A mouse model for the delta F508 allele of cystic fibrosis. J Clin Invest. 1995;96:2051–2064.
Wang Y, Hoenig JD, Malin KJ, et al. 16S rRNA gene-based analysis of fecal microbiota from preterm infants with and without necrotizing enterocolitis. ISME J. 2009;3:944–954.
Cole JR, Chai B, Marsh TL, et al. The Ribosomal Database Project (RDP-II): previewing a new autoaligner that allows regular updates and the new prokaryotic taxonomy. Nucleic Acid Res. 2003;31:442–443.
Ludwig W, Strunk O, Westram R, et al. ARB: a software environment for sequence data. Nucleic Acids Res. 2004;32:1363–1371.
Lozupone C, Hamady M, Knight R. UniFrac—an online tool for comparing microbial community diversity in a phylogenetic context. BMC Bioinformatics. 2006;7:371.
Drossman DA, Chey WD, Johanson JR, et al. Clinical trial: lubiprostone in patients with constipation-associated irritable bowel syndrome—results of two randomized, placebo-controlled studies. Aliment Pharmacol Ther. 2009;29:329–341.
MacDonald KD, McKenzie KR, Henderson MJ, et al. Lubiprostone activated non-CFTR dependent respiratory epithlelial chloride secretion in cystic fibrosis mice. Am J Physiol Lung Cell Mol Physiol. 2008;295:L933–L940.
Wang XF, Reddy MM, Quinton PM. Effects of a new cystic fibrosis transmembrane conductance regulator inhibitor on Cl− conductance in human sweat ducts. Exp Physiol. 2004;89:417–425.
Atuma C, Strugala V, Allen A, Holm L. The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am J Physiol. 2001;280:G922–G929.
Gustafsson JK, Ermund A, Johansson MEV, Schutte A, Hansson GC, Sjovall H. An ex vivo method for studying mucus formation, properties, and thickness in human colonic biopsies and mouse small and large intestinal explants. Am J Physiol. 2012;302:G430–G438.
Johansson MEV, Phillipson M, Petersson J, Velcich A, Holm L, Hansson GC. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc Natl Acad Sci USA. 2008;205:15064–15069.
Johansson MEV, Holmen Larsson JM, Hansson GC. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc Natl Acad Sci USA. 2011;108:4659–4665.
Swidsinski A, Loening-Baucke V, Theissig T, et al. Comparative study of the intestinal mucus barrier in normal and inflamed colon. Gut. 2007;56:343–350.
Paolillo R, RomanoCarratelli C, Sorrentino S, Mazzola N, Rizzo A. Immunomodulatory effects of Lactobacillus plantarum on human colon cancer cells. Int Immunopharmacol. 2009;9:1265–1271.
Tao Y, Drabik KA, Waypa TS, et al. Soluble factors from Lactobacillus GG activate MAPKs and induce cytoprotective heat shock proteins in intestinal epithelial cells. Am J Physiol Cell Physiol. 2006;290:C1018–C1030.
Kazaie K, Zadeh M, Khan MW, et al. Ablating colon cancer polyposis by Lactobacillus acidophilus deficient in lipoteichoic acid. Proc Natl Acad Sci USA. 2012;109:10462–10467.
Asha GD, Devaraja TN. Lactobacillus sp. As probiotics for human health with special emphasis on colorectal cancer. Ind J Sci Technol. 2011;4:1008–1014.
Mazmanian SK, Liu CH, Tzianabos AO, Kasper DL. An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell. 2005;122:107–108.
Guraner F, Malagelada JR. Gut flora in health and disease. Lancet. 2003;36:512–519.
O’Hara AM, Shanahan F. The gut flora as a forgotten organ. EMBO Rep. 2006;7:688–693.
Acknowledgments
Supported by a grant from Takeda Pharmaceuticals North America, Inc., NIH Digestive Disease Research Center grant DK-42086 (E.B.C.), NIH grant R37 DK47722 (E.B.C.), the University of Chicago Cancer Research Center (CA-01459), a fellowship grant from the Crohn’s and Colitis Foundation of America (Y.W.), NIH grant HD059123 (E.C.C.).
Conflict of interest
The authors disclose no other potential conflicts of interests or financial relationship with the organization that sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Musch, M.W., Wang, Y., Claud, E.C. et al. Lubiprostone Decreases Mouse Colonic Inner Mucus Layer Thickness and Alters Intestinal Microbiota. Dig Dis Sci 58, 668–677 (2013). https://doi.org/10.1007/s10620-012-2509-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2509-5