Abstract
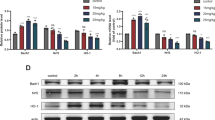
Oxidative stress-induced cell damage is involved in many neurological diseases. Interferon regulatory factor 6 (IRF6), a member of the IRF family of transcription factors, is required for the differentiation of skin, breast epithelium, and oral epithelium. However, the regulation and function of IRF6 in central nervous system remain unknown. This study aimed to investigate the role of IRF6 in hydrogen peroxide (H2O2)-induced oxidative neuronal injury in HT22 mouse hippocampal cells. Treatment with H2O2 significantly increased the expression of IRF6 at both mRNA and protein levels, and knockdown of IRF6 using specific small interfering RNA reduced H2O2-induced cytotoxicity, as evidenced by increased cell viability and decreased apoptosis. Knockdown of IRF6 attenuated intracellular reactive oxygen species (ROS) generation and lipid peroxidation, and also preserved endogenous antioxidant enzyme activities. The inhibitory effect of IRF6 knockdown on mitochondrial dysfunction was demonstrated by reduced mitochondrial oxidative level, preserved mitochondrial membrane potential (MMP) and ATP generation, as well as attenuated mitochondrial swelling. In addition, down-regulation of IRF6 inhibited the activation of mitochondrial apoptotic factors, whereas IRF6 knockdown together with caspase inhibitors had no extra effect on cell viability and LDH release. These results suggest that knockdown of IRF6 has protective effects against H2O2-induced oxidative stress by reducing ROS accumulation and apoptosis, and these protective effects are dependent on preservation of mitochondrial function.





Similar content being viewed by others
References
Alexander M, Forster C, Sugimoto K, Clark HB, Vogel S, Ross ME, Iadecola C (2003) Interferon regulatory factor-1 immunoreactivity in neurons and inflammatory cells following ischemic stroke in rodents and humans. Acta Neuropathol 105(5):420–424. doi:10.1007/s00401-002-0658-x
Armogida M, Nistico R, Mercuri NB (2012) Therapeutic potential of targeting hydrogen peroxide metabolism in the treatment of brain ischaemia. Br J Pharmacol 166(4):1211–1224. doi:10.1111/j.1476-5381.2012.01912.x
Avery SV (2011) Molecular targets of oxidative stress. Biochem J 434(2):201–210. doi:10.1042/BJ20101695
Bailey CM, Abbott DE, Margaryan NV, Khalkhali-Ellis Z, Hendrix MJ (2008) Interferon regulatory factor 6 promotes cell cycle arrest and is regulated by the proteasome in a cell cycle-dependent manner. Mol Cell Biol 28(7):2235–2243. doi:10.1128/MCB.01866-07
Bains M, Hall ED (2012) Antioxidant therapies in traumatic brain and spinal cord injury. Biochim Biophys Acta 1822(5):675–684. doi:10.1016/j.bbadis.2011.10.017
Botti E, Spallone G, Moretti F, Marinari B, Pinetti V, Galanti S, De Meo PD, De Nicola F, Ganci F, Castrignano T, Pesole G, Chimenti S, Guerrini L, Fanciulli M, Blandino G, Karin M, Costanzo A (2011) Developmental factor IRF6 exhibits tumor suppressor activity in squamous cell carcinomas. Proc Natl Acad Sci USA 108(33):13710–13715. doi:10.1073/pnas.1110931108
Chang LK, Putcha GV, Deshmukh M, Johnson EM Jr (2002) Mitochondrial involvement in the point of no return in neuronal apoptosis. Biochimie 84(2–3):223–231
Chen W, Royer WE Jr (2010) Structural insights into interferon regulatory factor activation. Cell Signal 22(6):883–887. doi:10.1016/j.cellsig.2009.12.005
Chen H, Yoshioka H, Kim GS, Jung JE, Okami N, Sakata H, Maier CM, Narasimhan P, Goeders CE, Chan PH (2011a) Oxidative stress in ischemic brain damage: mechanisms of cell death and potential molecular targets for neuroprotection. Antioxid Redox Signal 14(8):1505–1517. doi:10.1089/ars.2010.3576
Chen T, Liu W, Chao X, Zhang L, Qu Y, Huo J, Fei Z (2011b) Salvianolic acid B attenuates brain damage and inflammation after traumatic brain injury in mice. Brain Res Bull 84(2):163–168. doi:10.1016/j.brainresbull.2010.11.015
Chen T, Fei F, Jiang XF, Zhang L, Qu Y, Huo K, Fei Z (2012) Down-regulation of Homer1b/c attenuates glutamate-mediated excitotoxicity through endoplasmic reticulum and mitochondria pathways in rat cortical neurons. Free Radic Biol Med 52(1):208–217. doi:10.1016/j.freeradbiomed.2011.10.451
Chen T, Yang YF, Luo P, Liu W, Dai SH, Zheng XR, Fei Z, Jiang XF (2013a) Homer1 knockdown protects dopamine neurons through regulating calcium homeostasis in an in vitro model of Parkinson’s disease. Cell Signal 25(12):2863–2870. doi:10.1016/j.cellsig.2013.09.004
Chen T, Zhu J, Zhang C, Huo K, Fei Z, Jiang XF (2013b) Protective effects of SKF-96365, a non-specific inhibitor of SOCE, against MPP + -induced cytotoxicity in PC12 cells: potential role of Homer1. PLoS One 8(1):e55601. doi:10.1371/journal.pone.0055601
Circu ML, Aw TY (2010) Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med 48(6):749–762. doi:10.1016/j.freeradbiomed.2009.12.022
Cornelius C, Crupi R, Calabrese V, Graziano A, Milone P, Pennisi G, Radak Z, Calabrese EJ, Cuzzocrea S (2013) Traumatic brain injury: oxidative stress and neuroprotection. Antioxid Redox Signal 19(8):836–853. doi:10.1089/ars.2012.4981
Dai SH, Chen T, Wang YH, Zhu J, Luo P, Rao W, Yang YF, Fei Z, Jiang XF (2014a) Sirt3 attenuates hydrogen peroxide-induced oxidative stress through the preservation of mitochondrial function in HT22 cells. Int J Mol Med 34(4):1159–1168. doi:10.3892/ijmm.2014.1876
Dai SH, Chen T, Wang YH, Zhu J, Luo P, Rao W, Yang YF, Fei Z, Jiang XF (2014b) Sirt3 protects cortical neurons against oxidative stress via regulating mitochondrial Ca2+ and mitochondrial biogenesis. Int J Mol Sci 15(8):14591–14609. doi:10.3390/ijms150814591
Gough DR, Cotter TG (2011) Hydrogen peroxide: a Jekyll and Hyde signalling molecule. Cell Death Dis 2:e213. doi:10.1038/cddis.2011.96
Hacker G, Paschen SA (2007) Therapeutic targets in the mitochondrial apoptotic pathway. Expert Opin Ther Targets 11(4):515–526. doi:10.1517/14728222.11.4.515
Hall ED, Vaishnav RA, Mustafa AG (2010) Antioxidant therapies for traumatic brain injury. Neurotherapeutics 7(1):51–61. doi:10.1016/j.nurt.2009.10.021
Hiebert JB, Shen Q, Thimmesch AR, Pierce JD (2015) Traumatic brain injury and mitochondrial dysfunction. Am J Med Sci 350(2):132–138. doi:10.1097/MAJ.0000000000000506
Jones JL, Canady JW, Brookes JT, Wehby GL, L’Heureux J, Schutte BC, Murray JC, Dunnwald M (2010) Wound complications after cleft repair in children with Van der Woude syndrome. J Craniofac Surg 21(5):1350–1353. doi:10.1097/SCS.0b013e3181ec6aad
Kondo S, Schutte BC, Richardson RJ, Bjork BC, Knight AS, Watanabe Y, Howard E, de Lima RL, Daack-Hirsch S, Sander A, McDonald-McGinn DM, Zackai EH, Lammer EJ, Aylsworth AS, Ardinger HH, Lidral AC, Pober BR, Moreno L, Arcos-Burgos M, Valencia C, Houdayer C, Bahuau M, Moretti-Ferreira D, Richieri-Costa A, Dixon MJ, Murray JC (2002) Mutations in IRF6 cause Van der Woude and popliteal pterygium syndromes. Nat Genet 32(2):285–289. doi:10.1038/ng985
Laker RC, Xu P, Ryall KA, Sujkowski A, Kenwood BM, Chain KH, Zhang M, Royal MA, Hoehn KL, Driscoll M, Adler PN, Wessells RJ, Saucerman JJ, Yan Z (2014) A novel MitoTimer reporter gene for mitochondrial content, structure, stress, and damage in vivo. J Biol Chem 289(17):12005–12015. doi:10.1074/jbc.M113.530527
Lei B, Adachi N, Arai T (1998) Measurement of the extracellular H2O2 in the brain by microdialysis. Brain Res Brain Res Protoc 3(1):33–36
Lin Y, Xu D, Li X, Liu C, Liu X, Huang S, Huang Y (2015) Upregulation of interferon regulatory factor 6 promotes neuronal apoptosis after traumatic brain injury in adult rats. Cell Mol Neurobiol. doi:10.1007/s10571-015-0217-3
Niizuma K, Endo H, Chan PH (2009) Oxidative stress and mitochondrial dysfunction as determinants of ischemic neuronal death and survival. J Neurochem 109(Suppl 1):133–138. doi:10.1111/j.1471-4159.2009.05897.x
Parone PA, Da Cruz S, Han JS, McAlonis-Downes M, Vetto AP, Lee SK, Tseng E, Cleveland DW (2013) Enhancing mitochondrial calcium buffering capacity reduces aggregation of misfolded SOD1 and motor neuron cell death without extending survival in mouse models of inherited amyotrophic lateral sclerosis. J Neurosci 33(11):4657–4671. doi:10.1523/JNEUROSCI.1119-12.2013
Richardson RJ, Dixon J, Malhotra S, Hardman MJ, Knowles L, Boot-Handford RP, Shore P, Whitmarsh A, Dixon MJ (2006) Irf6 is a key determinant of the keratinocyte proliferation-differentiation switch. Nat Genet 38(11):1329–1334. doi:10.1038/ng1894
Robertson CL, Saraswati M, Fiskum G (2007) Mitochondrial dysfunction early after traumatic brain injury in immature rats. J Neurochem 101(5):1248–1257. doi:10.1111/j.1471-4159.2007.04489.x
Salahshourifar I, Wan Sulaiman WA, Halim AS, Zilfalil BA (2012) Mutation screening of IRF6 among families with non-syndromic oral clefts and identification of two novel variants: review of the literature. Eur J Med Genet 55(6–7):389–393. doi:10.1016/j.ejmg.2012.02.006
Schon EA, Manfredi G (2003) Neuronal degeneration and mitochondrial dysfunction. J Clin Investig 111(3):303–312. doi:10.1172/JCI17741
Schutte BC, Saal HM, Goudy S, Leslie E (1993) IRF6-related disorders. In: Pagon RA, Adam MP, Ardinger HH et al (eds) GeneReviews(R). University of Washington, Seattle
Senderowicz AM, Sausville EA (2000) Preclinical and clinical development of cyclin-dependent kinase modulators. J Natl Cancer Inst 92(5):376–387
Sundaresan M, Yu ZX, Ferrans VJ, Irani K, Finkel T (1995) Requirement for generation of H2O2 for platelet-derived growth factor signal transduction. Science 270(5234):296–299
Xiang M, Wang L, Guo S, Lu YY, Lei H, Jiang DS, Zhang Y, Liu Y, Zhou Y, Zhang XD, Li H (2014) Interferon regulatory factor 8 protects against cerebral ischaemic-reperfusion injury. J Neurochem 129(6):988–1001. doi:10.1111/jnc.12682
Yanai H, Negishi H, Taniguchi T (2012) The IRF family of transcription factors: inception, impact and implications in oncogenesis. Oncoimmunology 1(8):1376–1386. doi:10.4161/onci.22475
Yu Z, Liu N, Li Y, Xu J, Wang X (2013) Neuroglobin overexpression inhibits oxygen-glucose deprivation-induced mitochondrial permeability transition pore opening in primary cultured mouse cortical neurons. Neurobiol Dis 56:95–103. doi:10.1016/j.nbd.2013.04.015
Zhao GN, Jiang DS, Li H (2015) Interferon regulatory factors: at the crossroads of immunity, metabolism, and disease. Biochim Biophys Acta 1852(2):365–378. doi:10.1016/j.bbadis.2014.04.030
Acknowledgments
This study has been funded by the Shaanxi Province Scientific and Technological Research and Development Program (No. 2004K14-G; No.2010, K01-154) and Shaanxi Province Natural Science Foundation Research Program (No. 2014JM4131). The authors would like to thank Dr. Terry Chen for his technical support for the experiments and the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Rights and permissions
About this article
Cite this article
Guo, XM., Chen, B., Lv, JM. et al. Knockdown of IRF6 Attenuates Hydrogen Dioxide-Induced Oxidative Stress via Inhibiting Mitochondrial Dysfunction in HT22 Cells. Cell Mol Neurobiol 36, 1077–1086 (2016). https://doi.org/10.1007/s10571-015-0301-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-015-0301-8