Abstract
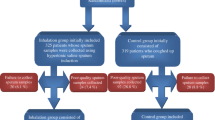
The diagnostic yield of pulmonary tuberculosis (TB) by sputum induction (SI) at the first point of contact with health services, conducted in all patients with suspected TB regardless of the ability to expectorate spontaneously, has not been evaluated. We compared the diagnostic yield of SI to routine sputum collection in a South African community setting. Ambulatory patients with suspected TB provided a ‘spot’ expectorated sputum sample, an SI sample by hypertonic (5 %) saline nebulization, and early morning expectorated sputum sample. The diagnostic yield of sputum smear microscopy and liquid culture (denominator all subjects with any positive Mycobacterium tuberculosis culture), and time-to-positivity of culture were compared between SI and expectorated samples. A total of 555 subjects completed the SI procedure, of whom 132 (24 %) were human immunodeficiency virus (HIV)-infected. One hundred and twenty-nine samples (129, 23 %) were M. tuberculosis culture-positive. The time-to-positivity of Mycobacteria Growth Indicator Tube (MGIT) culture was shorter for SI (median difference 2 days, p = 0.63) and for early morning expectorated sputum (median difference 2 days, p = 0.02) compared to spot expectorated sputum. However, there was no difference in the culture-positive diagnostic yield between SI and spot expectorated sputum [difference +0.7 %; confidence interval (CI) −7.0 to +8.5 %, p = 0.82] or SI and early morning expectorated sputum (difference +4.7 %; CI −3.2 to +12.5 %, p = 0.20) for all subjects or for HIV-infected subjects. SI reduces the time to positive M. tuberculosis culture, but does not increase the rate of positive culture compared to routine expectorated sputum collection. SI cannot be recommended as the routine collection method at first contact among ambulatory patients with suspected TB in high-burden communities.

Similar content being viewed by others
References
Mase SR, Ramsay A, Ng V et al (2007) Yield of serial sputum specimen examinations in the diagnosis of pulmonary tuberculosis: a systematic review. Int J Tuberc Lung Dis 11(5):485–495
Harries AD, Mphasa NB, Mundy C, Banerjee A, Kwanjana JH, Salaniponi FM (2000) Screening tuberculosis suspects using two sputum smears. Int J Tuberc Lung Dis 4(1):36–40
Boehme CC, Nabeta P, Hillemann D et al (2010) Rapid molecular detection of tuberculosis and rifampin resistance. N Engl J Med 363(11):1005–1015
Peter JG, Cashmore TJ, Meldau R, Theron G, van Zyl-Smit R, Dheda K (2012) Diagnostic accuracy of induced sputum LAM ELISA for tuberculosis diagnosis in sputum-scarce patients. Int J Tuberc Lung Dis 16(8):1108–1112
Parry CM, Kamoto O, Harries AD et al (1995) The use of sputum induction for establishing a diagnosis in patients with suspected pulmonary tuberculosis in Malawi. Tuber Lung Dis 76(1):72–76
Menzies D (2003) Sputum induction: simpler, cheaper, and safer—but is it better? Am J Respir Crit Care Med 167(5):676–677
Gonzalez-Angulo Y, Wiysonge CS, Geldenhuys H et al (2012) Sputum induction for the diagnosis of pulmonary tuberculosis: a systematic review and meta-analysis. Eur J Clin Microbiol Infect Dis 31(7):1619–1630
Paggiaro PL, Chanez P, Holz O et al (2002) Sputum induction. Eur Respir J Suppl 37:3s–8s
Schoch OD, Rieder P, Tueller C et al (2007) Diagnostic yield of sputum, induced sputum, and bronchoscopy after radiologic tuberculosis screening. Am J Respir Crit Care Med 175(1):80–86
Saglam L, Akgun M, Aktas E (2005) Usefulness of induced sputum and fibreoptic bronchoscopy specimens in the diagnosis of pulmonary tuberculosis. J Int Med Res 33(2):260–265
McWilliams T, Wells AU, Harrison AC, Lindstrom S, Cameron RJ, Foskin E (2002) Induced sputum and bronchoscopy in the diagnosis of pulmonary tuberculosis. Thorax 57(12):1010–1014
Peter JG, Theron G, Pooran A, Thomas J, Pascoe M, Dheda K (2013) Comparison of two methods for acquisition of sputum samples for diagnosis of suspected tuberculosis in smear-negative or sputum-scarce people: a randomised controlled trial. Lancet Respir Med 1(6):471–478
Zar HJ, Hanslo D, Apolles P, Swingler G, Hussey G (2005) Induced sputum versus gastric lavage for microbiological confirmation of pulmonary tuberculosis in infants and young children: a prospective study. Lancet 365(9454):130–134
Hatherill M, Hawkridge T, Zar HJ et al (2009) Induced sputum or gastric lavage for community-based diagnosis of childhood pulmonary tuberculosis? Arch Dis Child 94(3):195–201
Geldenhuys HD, Kleynhans W, Buckerfield N et al (2012) Safety and tolerability of sputum induction in adolescents and adults with suspected pulmonary tuberculosis. Eur J Clin Microbiol Infect Dis 31(4):529–537
English R (2008) Boland/Overberg region annual health status report 2007/08. Department of Health, Worcester
Hesseling AC, Walzl G, Enarson DA et al (2010) Baseline sputum time to detection predicts month two culture conversion and relapse in non-HIV-infected patients. Int J Tuberc Lung Dis 14(5):560–570
Bark CM, Thiel BA, Johnson JL (2012) Pretreatment time to detection of Mycobacterium tuberculosis in liquid culture is associated with relapse after therapy. J Clin Microbiol 50(2):538
Acknowledgments
The authors wish to thank the following for the collection of data: 1,2Manshil Misra, 1,2William Kleynhans, 1,2Norma Buckerfield, 1,2Susan Rossouw, 1,2Elizabeth Filander, and the 1,2SATVI Induced Sputum team.
1South African Tuberculosis Vaccine Initiative (SATVI), Institute of Infectious Disease and Molecular Medicine (IDM), and 2School of Child and Adolescent Health, University of Cape Town, Cape Town, South Africa.
Contributions of authors
HDG and MH contributed towards the study planning, study conduct, and drafted the early versions of the manuscript. All other authors were involved in planning the study, implementation, and commenting on the draft manuscript. All authors approved the final version.
The study was approved by the University of Cape Town Human Research Ethics Committee (Human Subjects Assurance number 00001637) and written informed consent was obtained for participation. The study was conducted in accordance with the Declaration of Helsinki.
Conflict of interest
All authors declare that they have no conflict of interest.
Funding
This work was supported by a grant from the National Institutes of Health (NIH), USA (grant number R01 AI75603-02).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Geldenhuys, H.D., Whitelaw, A., Tameris, M.D. et al. A controlled trial of sputum induction and routine collection methods for TB diagnosis in a South African community. Eur J Clin Microbiol Infect Dis 33, 2259–2266 (2014). https://doi.org/10.1007/s10096-014-2198-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-014-2198-4