Abstract
Pacific Island populations, particularly those of Polynesian descent, have a high prevalence of hyperuricaemia and gout. This is due to an inherently higher urate level among these populations with a demonstrated genetic predisposition. While an excess of urate can cause pathology, urate is also important for human health. It has been implicated as an antioxidant, has a neuroprotective role and is involved in innate immune responses. This paper provides a brief review of urate levels worldwide, with a particular focus on island Southeast Asia and the Pacific. We then present possible evolutionary explanations for the elevated serum urate levels among Pacific populations in the context of the physiological importance of urate and of the settlement history of the region. Finally, we propose that ancestry may play a significant role in hyperuricaemia in these populations and that exposure to malaria prior to population expansion into the wider Pacific may have driven genetic selection for variants contributing to high serum urate.



Similar content being viewed by others
References
Wu X, Wakamiya M, Vaishnav S, Geske R, Montgomery C, Jones P, Bradley A, Caskey CT (1994) Hyperuricemia and urate nephropathy in urate oxidase-deficient mice. Proc Natl Acad Sci USA 91(2):742–746. doi:10.1073/pnas.91.2.742
Oda M, Satta Y, Takenaka O, Takahata N (2002) Loss of urate oxidase activity in hominoids and its evolutionary implications. Mol Biol Evol 19(5):640–653
Johnson RJ, Titte S, Cade JR, Rideout BA, Oliver WJ (2005) Uric acid, evolution and primitive cultures. Semin Nephrol 25(1):3–8. doi:10.1016/j.semnephrol.2004.09.002
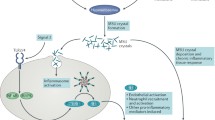
Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440(7081):237–241. doi:10.1038/nature04516
Rock KL, Kataoka H, Lai J-J (2013) Uric acid as a danger signal in gout and its comorbidities. Nat Rev Rheumatol 9(1):13–23. doi:10.1038/nrrheum.2012.143
de Lau LML, Koudstaal PJ, Hofman A, Breteler MMB (2005) Serum uric acid levels and the risk of Parkinson disease. Ann Neurol 58(5):797–800. doi:10.1002/ana.20663
Spitsin S, Hooper DC, Mikheeva T, Koprowski H (2001) Uric acid levels in patients with multiple sclerosis: analysis in mono- and dizygotic twins. Mult Scler 7(3):165–166. doi:10.1177/135245850100700305
Rentzos M, Nikolaou C, Anagnostouli M, Rombos A, Tsakanikas K, Economou M, Dimitrakopoulos A, Karouli M, Vassilopoulos D (2006) Serum uric acid and multiple sclerosis. Clin Neurol Neurosurg 108(6):527–531. doi:10.1016/j.clineuro.2005.08.004
Kim T-S, Pae C-U, Yoon S-J, Jang W-Y, Lee NJ, Kim J-J, Lee S-J, Lee C, Paik I-H, Lee C-U (2006) Decreased plasma antioxidants in patients with Alzheimer’s disease. Int J Geriatr Psychiatry 21(4):344–348. doi:10.1002/gps.1469
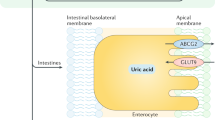
Maesaka JK, Fishbane S (1998) Regulation of renal urate excretion: a critical review. Am J Kidney Dis 32(6):917–933. doi:10.1016/S0272-6386(98)70067-8
Merriman TR, Dalbeth N (2011) The genetic basis of hyperuricaemia and gout. Joint Bone Spine 78(1):35–40. doi:10.1016/j.jbspin.2010.02.027
Hak AE, Choi HK (2008) Menopause, postmenopausal hormone use and serum uric acid levels in US women—the Third National Health and Nutrition Examination Survey. Arthritis Res Ther 10(5):R116. doi:10.1186/ar2519
Healey LA, Caner JEZ, Bassett DR, Decker JL (1966) Serum uric acid and obesity in Hawaiians. JAMA 196(4):364–365. doi:10.1001/jama.1966.03100170106039
Prior I (1981) Epidemiology of rheumatic disorders in the Pacific with particular emphasis on hyperuricaemia and gout. Semin Arthritis Rheum 11(1):213–229. doi:10.1016/0049-0172(81)90101-3
Suzuki T (1993) Paleopathological and paleoepidemiological investigation of human skeletal remains of early Hawaiians from Mokapu Site, Oahu Island, Hawaii. Jpn Rev 4:83–128. doi:10.2307/25790927
Healey LA, Skeith MD, Decker JL, Bayani-Sioson PS (1967) Hyperuricaemia in Filipinos: interaction of heredity and environment. Am J Hum Genet 19(2):81–85
Duff IF, Mikkelsen WM, Dodge HJ, Himes DS (1968) Comparison of uric acid levels in some oriental and Caucasian groups unselected as to gout or hyperuricemia. Arthritis Rheum 11(2):184–190. doi:10.1002/art.1780110209
Reed D, Labarthe D, Stallones R (1972) Epidemiologic studies of serum uric acid levels among Micronesians. Arthritis Rheum 15(4):381–390. doi:10.1002/art.1780150409
Wyatt GB, Griew AR, Martin FIR, Campbell DG (1980) Plasma cholesterol, triglyceride and uric acid in urban and rural communities in Papua New Guinea. Aust N Z J Med 10(5):491–495. doi:10.1111/j.1445-5994.1980.tb04964.x
Jackson L, Taylor R, Faaiuso S, Ainuu SP, Whitehouse S, Zimmet P (1981) Hyperuricaemia and gout in Western Samoans. J Chronic Dis 34(2–3):65–75. doi:10.1016/0021-9681(81)90052-7
Finau SA, Stanhope JM, Prior IAM, Joseph JG, Puloka ST, Leslie PN (1983) The Tonga cardiovascular and metabolic study: design, demographic aspects and medical findings. Community Health Stud 7(1):67–77. doi:10.1111/j.1753-6405.1983.tb00393.x
Tuomilehto J, Zimmet P, Wolf E, Taylor R, Ram P, King H (1988) Plasma uric acid level and its association with diabetes mellitus and some biologic parameters in a biracial population of Fiji. Am J Epidemiol 127(2):321–336
Prior IAM, Welby TJ, Østbye T, Salmond CE, Stokes YM (1987) Migration and gout: the Tokelau Island migrant study. Br Med J (Clin Res Ed) 295(6596):457–461. doi:10.1136/bmj.295.6596.457
Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G (2004) Purine-rich foods, dairy and protein intake, and the risk of gout in men. N Engl J Med 350(11):1093–1103. doi:10.1056/NEJMoa035700
Choi HK, Curhan G (2008) Soft drinks, fructose consumption, and the risk of gout in men: prospective cohort study. BMJ 336(7639):309–312. doi:10.1136/bmj.39449.819271.BE
Batt C, Phipps-Green AJ, Black MA, Cadzow M, Merriman ME, Topless R, Gow P, Harrison A, Highton J, Jones P, Stamp L, Dalbeth N, Merriman TR (2013) Sugar-sweetened beverage consumption: a risk factor for prevalent gout with SLC2A9 genotype-specific effects on serum urate and risk of gout. Ann Rheum Dis. doi:10.1136/annrheumdis-2013-203600
Gray RD, Drummond AJ, Greenhill SJ (2009) Language phylogenies reveal expansion pulses and pauses in Pacific settlement. Science 323(5913):479–483. doi:10.1126/science.1166858
Chang SJ, Ko YC, Wang T-N, Chang FT, Cinokotai FF, Chen CJ (1997) High prevalence of gout and related risk factors in Taiwan’s aborigines. J Rheumatol 27(7):1364–1369
Chou CT, Lai JS (1998) The epidemiology of hyperuricaemia and gout in Taiwan aborigines. Rheumatology 37(3):258–262. doi:10.1093/rheumatology/37.3.258
Cox MP, Nelson MG, Tumonggor MK, Ricaut F-X, Sudoyo H (2012) A small cohort of Island Southeast Asian women founded Madagascar. Proc R Soc Lond [Biol] 279(1739):2761–2768. doi:10.1098/rspb.2012.0012
Capdevielle P, Teyssier P, Valmary J, Perrot J, Delprat J (1980) Goutte et hyperuricemies a Tananarive. Med Trop 40(4):409–418
Fitzpatrick SM, Callaghan R (2008) Seafaring simulations and the origin of prehistoric settlers to Madagascar. In: Clark G, Leach F, O’Connor S (eds) Islands of inquiry: colonisation, seafaring and the archaeology of maritime landscapes, vol 29. ANU Press, Canberra, pp 47–58
Conen D, Wietlisbach V, Bovet P, Shamlaye C, Riesen W, Paccaud F, Burnier M (2004) Prevalence of hyperuricemia and relation of serum uric acid with cardiovascular risk factors in a developing country. BMC Public Health 4(1):9. doi:10.1186/1471-2458-4-9
Kayser M (2010) The human genetic history of Oceania: near and remote views of dispersal. Curr Biol 20(4):R194–R201. doi:10.1016/j.cub.2009.12.004
Friedlaender JS, Friedlaender FR, Hodgson JA, Stoltz M, Koki G, Horvat G, Zhadanov S, Schurr TG, Merriwether DA (2007) Melanesian mtDNA complexity. PLoS One 2(2):e248
Hughes RG, Marks GC (2009) Against the tide of change: diet and health in the Pacific islands. J Am Diet Assoc 109(10):1700–1703. doi:10.1016/j.jada.2009.07.015
Hanna JM, Pelletier DL, Brown VJ (1986) The diet and nutrition of contemporary Samoans. In: Baker PT, Hanna JM, Baker TS (eds) The changing Samoans: behavior and health in transition. Oxford University Press, Oxford, pp 275–296
Coyne T, Hughes R, Langi S (2000) Lifestyle diseases in Pacific communities, vol 219. Secretariat of the Pacific Community, Noumea
Lenane GAQ, Rose BS, Isdale IC (1960) Gout in Maori. Ann Rheum Dis 19:120–125. doi:10.1136/ard.19.2.120
Rose BS (1975) Gout in the Maoris. Semin Arthritis Rheum 5(2):121–145. doi:10.1016/0049-0172(75)90002-5
Johnson RJ, Rideout BA (2004) Uric acid and diet—insights into the epidemic of cardiovascular disease. N Engl J Med 350(11):1071–1073. doi:10.1056/NEJMp048015
Schlesinger N (2005) Dietary factors and hyperuricaemia. Curr Pharm Des 11:4133–4138. doi:10.2174/138161205774913273
Álvarez-Lario B, Macarrón-Vicente J (2010) Uric acid and evolution. Rheumatology 49(11):2010–2015. doi:10.1093/rheumatology/keq204
Buckley H, Tayles N, Halcrow SE, Robb K, Fyfe R (2010) The people of Wairau bar: a re-examination. J Pac Archaeol 1(1):1–20
Campbell M, Hudson B (2011) The Northern Runway Development (NRD) site Community report. Auckland
Buckley HR (2007) Possible gouty arthritis in Lapita-associated skeletons from Teouma, Efate Island, Central Vanuatu. Curr Anthropol 48(5):741–749. doi:10.1086/520967
Rothschild BM, Heathcote GM (1995) Characterization of gout in a skeletal population sample: presumptive diagnosis in a Micronesian population. Am J Phys Anthropol 98(4):519–525. doi:10.1002/ajpa.1330980411
Douglas MT, Pietrusewsky M, Ikehara-Quebral RM (1997) Skeletal biology of Apurguan: a precontact Chamorro site on Guam. Am J Phys Anthropol 104(3):291–313. doi:10.1002/(sici)1096-8644(199711)104:3<291:aid-ajpa3>3.0.co;2-z
Gosling AL, Matisoo-Smith E, Merriman TR (2013) Gout in Maori. Rheumatology. doi:10.1093/rheumatology/ket299
Pollock NJ (1992) These roots remain: food habits in islands of the central and eastern Pacific since Western contact. The Institute for Polynesian Studies, Laie, HI
Köttgen A, Albrecht E, Teumer A, Vitart V, Krumsiek J, Hundertmark C, Pistis G, Ruggiero D, O’Seaghdha CM, Haller T (2012) Genome-wide association analyses identify 18 new loci associated with serum urate concentrations. Nat Genet 45(2):145–154. doi:10.1038/ng.2500
Hollis-Moffatt JE, Xu X, Dalbeth N, Merriman ME, Topless R, Waddell C, Gow PJ, Harrison AA, Highton J, Jones PBB, Stamp LK, Merriman TR (2009) Role of the urate transporter SLC2A9 gene in susceptibility to gout in New Zealand Māori, Pacific Island, and Caucasian case–control sample sets. Arthritis Rheum 60(11):3485–3492. doi:10.1002/art.24938
Phipps-Green AJ, Hollis-Moffatt JE, Dalbeth N, Merriman ME, Topless R, Gow PJ, Harrison AA, Highton J, Jones PBB, Stamp LK, Merriman TR (2010) A strong role for the ABCG2 gene in susceptibility to gout in New Zealand Pacific Island and Caucasian, but not Māori, case and control sample sets. Hum Mol Genet 19(24):4813–4819. doi:10.1093/hmg/ddq412
Hollis-Moffatt JE, Phipps-Green AJ, Chapman B, Jones GT, van Rij A, Gow PJ, Harrison AA, Highton J, Jones PB, Montgomery GW (2012) The renal urate transporter SLC17A1 locus: confirmation of association with gout. Arthritis Res Ther 14(2):R92. doi:10.1186/ar3816
Spriggs M (2011) Archaeology and the Austronesian expansion: where are we now? Antiquity 85:510–528
Wilmshurst JM, Hunt TL, Lipo CP, Anderson AJ (2011) High-precision radiocarbon dating shows recent and rapid initial human colonization of East Polynesia. Proc Natl Acad Sci USA 108(5):1815–1820. doi:10.1073/pnas.1015876108
Pietrusewsky M (1996) The physical anthropology of Polynesia: a review of some cranial and skeletal studies. In: Davidson J, Irwin G, Leach B, Pawley A, Brown D (eds) Oceanic culture history: essays in honour of Roger Green. New Zealand Journal of Archaeology Special Publication. New Zealand Archaeological Association, Dunedin
Kayser M, Brauer S, Cordaux R, Casto A, Lao O, Zhivotovsky LA, Moyse-Faurie C, Rutledge RB, Schiefenhoevel W, Gil D, Lin AA, Underhill PA, Oefner PJ, Trent RJ, Stoneking M (2006) Melanesian and Asian origins of Polynesians: mtDNA and Y chromosome gradients across the Pacific. Mol Biol Evol 23(11):2234–2244. doi:10.1093/molbev/msl093
Ames BN, Cathcart R, Schwiers E, Hochstein P (1981) Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci USA 78(11):6858–6862
Scott GS, Hooper DC (2001) The role of uric acid in protection against peroxynitrite-mediated pathology. Med Hypotheses 56(1):95–100. doi:10.1054/mehy.2000.1118
Kutzing MK, Firestein BL (2008) Altered uric acid levels and disease states. J Pharmacol Exp Ther 324(1):1–7. doi:10.1124/jpet.107.129031
Cannon PJ, Stason WB, Demartini FE, Sommers SC, Laragh JH (1966) Hyperuricemia in primary and renal hypertension. N Engl J Med 275(9):457–464. doi:10.1056/NEJM196609012750902
Jossa F, Farinaro E, Panico S, Krogh V, Celentano E, Galasso R, Mancini M, Trevisan M (1994) Serum uric acid and hypertension: the Olivetti heart study. J Hum Hypertens 8(9):677–681
Johnson RJ, Kang D-H, Feig D, Kivlighn S, Kanellis J, Watanabe S, Tuttle KR, Rodriguez-Iturbe B, Herrera-Acosta J, Mazzali M (2003) Is there a pathogenetic role for uric acid in hypertension and cardiovascular and renal disease? Hypertension 41(6):1183–1190. doi:10.1161/01.hyp.0000069700.62727.c5
Watanabe S, Kang D-H, Feng L, Nakagawa T, Kanellis J, Lan H, Mazzali M, Johnson RJ (2002) Uric acid, hominoid evolution, and the pathogenesis of salt-sensitivity. Hypertension 40(3):355–360. doi:10.1161/01.hyp.0000028589.66335.aa
Johnson R, Sautin Y, Oliver W, Roncal C, Mu W, Gabriela Sanchez-Lozada L, Rodriguez-Iturbe B, Nakagawa T, Benner S (2009) Lessons from comparative physiology: could uric acid represent a physiologic alarm signal gone awry in western society? J Comp Physiol [B] 179(1):67–76. doi:10.1007/s00360-008-0291-7
Gumaa KA, Mustafa KY, Mahmoud NA, Gader AMA (1978) The effects of fasting in Ramadan. Br J Nutr 49:573–581. doi:10.1079/BJN19780161
Lennox WG (1924) Increase of uric acid in the blood during prolonged starvation. JAMA 82(8):602–604. doi:10.1001/jama.1924.02650340012004
Shi Y, Evans JE, Rock KL (2003) Molecular identification of a danger signal that alerts the immune system to dying cells. Nature 425(6957):516–521. doi:10.1038/nature01991
Shi Y, Mucsi AD, Ng G (2010) Monosodium urate crystals in inflammation and immunity. Immunol Rev 233(1):203–217. doi:10.1111/j.0105-2896.2009.00851.x
Martinon F (2010) Mechanisms of uric acid crystal-mediated autoinflammation. Immunol Rev 233(1):218–232. doi:10.1111/j.0105-2896.2009.00860.x
Sabeti PC, Schaffner SF, Fry B, Lohmueller J, Varilly P, Shamovsky O, Palma A, Mikkelsen TS, Altshuler D, Lander ES (2006) Positive natural selection in the human lineage. Science 312(5780):1614–1620. doi:10.1126/science.1124309
Schofield L, Grau GE (2005) Immunological processes in malaria pathogenesis. Nat Rev Immunol 5:722–735. doi:10.1038/nri1686
Conway DJ, Fanello C, Lloyd JM, Al-Joubori BMAS, Baloch AH, Somanath SD, Roper C, Oduola AMJ, Mulder B, Povoa MM, Singh B, Thomas AW (2000) Origin of plasmodium falciparum malaria is traced by mitochondrial DNA. Mol Biochem Parasitol 111(1):163–171. doi:10.1016/S0166-6851(00)00313-3
Kwiatkowski DP (2005) How malaria has affected the human genome and what human genetics can teach us about malaria. Am J Med Genet 77(2):171–192. doi:10.1086/432519
Griffith JW, Sun T, McIntosh MT, Bucala R (2009) Pure hemozoin is inflammatory in vivo and activates the NALP3 inflammasome via release of uric acid. J Immunol 183(8):5208–5220. doi:10.4049/jimmunol.0713552
Shio MT, Kassa FA, Bellemare M-J, Olivier M (2010) Innate inflammatory response to the malarial pigment hemozoin. Microbes Infect 12(12–13):889–899. doi:10.1016/j.micinf.2010.07.001
Orengo JM, Leliwa-Sytek A, Evans JE, Evans B, van de Hoef D, Nyako M, Day K, Rodriguez A (2009) Uric acid is a mediator of the Plasmodium falciparum-induced inflammatory response. PLoS ONE 4(4):e5194. doi:10.1371/journal.pone.0005194
Buckley H (2006) ‘The predators within’: investigating the relationship between malaria and health in the prehistoric Pacific Islands. In: Oxenham M, Tayles N (eds) Bioarchaeology of Southeast Asia. Cambridge University Press, Cambridge, pp 309–332
Hay SI, Guerra CA, Tatem AJ, Noor AM, Snow RW (2004) The global distribution and population at risk of malaria: past, present, and future. Lancet Infect Dis 4(6):327–336. doi:10.1016/S1473-3099(04)01043-6
Hill AVS, Bowden DK, Trent RJ, Higgs DR, Oppenheimer SJ, Thein SL, Mickleson KNP, Weatherall DJ, Clegg JB (1985) Melanesians and Polynesians share a unique α-thalassaemia mutation. Am J Hum Genet 37:571–580
Flint J, Hill A, Bowden D, Oppenheimer S, Sill PR, Serjeantson S, Bana-Koiri J, Bhatia K, Alpers MP, Boyce AJ, Weatherall DJ, Clegg JB (1986) High frequencies of α-thalassaemia are the result of natural selection by malaria. Nature 321:744–750. doi:10.1038/321744a0
Hill AVS, Gentile B, Bonnardot JM, Roux J, Weatherall DJ, Clegg JB (1987) Polynesian origins and affinities: globin gene variants in east Polynesia. Am J Hum Genet 40:453–463
Hill AVS, O’Shaughnessy D, Clegg JB (1989) Haemoglobin and globin gene variants in the Pacific. In: Hill AVS, Sergeantson SW (eds) The colonization of the Pacific: a genetic trail. Oxford University Press, Oxford
Clark JT, Kelly KM (1993) Human genetics, paleoenvironments, and malaria: relationships and implications for the settlement of Oceania. Am Anthropol 95(3):612–630. doi:10.1525/aa.1993.95.3.02a00040
Hornabrook RW, Sergeantson S, Stanhope J (1975) Normal serum biochemistry in Papua New Guinean adults. P N G Med J 18(4):232–238
Summerhayes GR, Leavesley M, Fairbairn A, Mandui H, Field J, Ford A, Fullagar R (2010) Human adaptation and plant use in highland New Guinea 49,000 to 44,000 years ago. Science 330(6000):78–81. doi:10.1126/science.1193130
Müller I, Bockarie M, Alpers M, Smith T (2003) The epidemiology of malaria in Papua New Guinea. Trends Parasitol 19(6):253–259. doi:10.1016/S1471-4922(03)00091-6
Kirch PV, Rallu J-L (eds) (2007) The growth and collapse of Pacific island societies: archaeological and demographic perspectives. University of Hawai’i Press, Honolulu
Denoon D (1995) Pacific Island depopulation: natural or un-natural history? In: Bryder L, Dow DA (eds) New countries and old medicine: proceedings of an international conference on the history of medicine and health. Pyramid Press, Auckland
McArthur N (1968) Island populations of the Pacific. Australian National University Press, Canberra
Harrison GA, Tanner JM, Pilbeam DR, Baker DT (1988) Human biology: an introduction to human evolution, variation, growth and adaptability. Oxford University Press, Oxford
Chang HY, Pan WH, Yeh WT, Tsai KS (2001) Hyperuricemia and gout in Taiwan: results from the Nutritional and Health Survey in Taiwan (1993–96). J Rheumatol 28(7):1640–1646
Brauer GW, Prior IA (1978) A prospective study of gout in New Zealand Maoris. Ann Rheum Dis 37(5):466–472. doi:10.1136/ard.37.5.466
Stamp LK, Wells JE, Pitama S, Faatoese A, Doughty RN, Whalley G, Richards AM, Cameron VA (2012) Hyperuricaemia and gout in New Zealand rural and urban Māori and non-Māori communities. Intern Med J 43(6):678–684. doi:10.1111/imj.12062
Prior IA, Rose BS (1966) Uric acid, gout and public health in the South Pacific. N Z Med J 65:295–300
Hasselot N, Ott D, Blanchot M (1989) Détermination de quelques valeurs usuelles fréquentes en biochimie clinique dan un échantillon de populations polynésiennes: comparaison avec les valeurs de référence françaises. Med Trop 49(2):179–182
Healey LA, Caner EZJ, Decker JL (1966) Ethnic variations in serum uric acid I. Filipino hyperuricemia in a controlled environment. Arthritis Rheum 9(2):288–294. doi:10.1002/art.1780090204
Healey LA, Jones KW (1971) Hyperuricemia in American Samoans. Arthritis Rheum 14(2):283–285. doi:10.1002/art.1780140215
Østbye T, Welby T, Prior I, Salmond C, Stokes Y (1989) Type 2 (non-insulin-dependent) diabetes mellitus, migration and westernisation: the Tokelau Island Migrant study. Diabetologia 32(8):585–590. doi:10.1007/bf00285332
Jackson L, Taylor R, Whitehouse S, Zimmet P (1980) The high prevalence of hyperuricaemia in a Polynesian populations. Community Health Stud 4(3):175–179
Zimmet PZ, Whitehouse S, Jackson L, Thoma K (1978) High prevalence of hyperuricaemia and gout in an urbanised Micronesian population. BMJ 1(6122):1237–1239. doi:10.1136/bmj.16122.1237
Adams WH, Harper JA, Heotis PM, Jamner AH (1984) Hyperuricemia in the inhabitants of the Marshall Islands. Arthritis Rheum 27(6):713–716. doi:10.1002/art.1780270620
Burch TA, O’Brien WM, Need R, Kurland LT (1966) Hyperuricaemia and gout in the Mariana Islands. Ann Rheum Dis 25(2):114–116. doi:10.1136/annrheumd00507-0008
Page LB, Damon A, Moellering RC (1974) Antecedents of cardiovascular disease in six Solomon Islands societies. Circulation 49(6):1132–1146. doi:10.1161/01.cir.49.6.1132
Lindeberg S, Cordain L, Råstam L, Ahrén B (2004) Serum uric acid in traditional Pacific Islanders and in Swedes. J Intern Med 255(3):373–378. doi:10.1046/j.1365-2796.2003.01272.x
Sinnett PF, Whyte HM (1973) Epidemiological studies in a total highland population, Tukisenta, New Guinea: cardiovascular disease and relevant clinical, electrocardiographic, radiological and biochemical findings. J Chronic Dis 26(5):265–290. doi:10.1016/0021-9681(73)90031-3
Brennan PJ, Simpson JM, McGilchrist CA, Blacket RB (1980) Serum lipids and other coronary risk factors in Sydney blood donors: a comparison with other Australian subjects. Community Health Stud 4(1):1–6
Nabipour I, Sambrook PN, Blyth FM, Janu MR, Waite LM, Naganathan V, Handelsman DJ, Le Couteur DG, Cumming RG, Seibel MJ (2011) Serum uric acid is associated with bone health in older men: a cross-sectional population-based study. J Bone Miner Res 26(5):955–964. doi:10.1002/jbmr.286
Emmerson B, Douglas W, Doherty R, Feigl P (1969) Serum urate concentrations in the Australian aboriginal. Ann Rheum Dis 28(2):150–156. doi:10.1136/ard.28.2.150
Darmawan J, Valkenburg H, Muirden KD, Wigley RD (1992) The epidemiology of gout and hyperuricaemia in a rural population of Java. J Rheumatol 19:1595–1599
Reddy K, Bulliyya G, Rama Chandraiah T, Kumari K, Reddanna P, Thyagaraju K (1991) Serum lipids and lipid peroxidation pattern in industrial and rural workers in India. Age 14(2):33–38. doi:10.1007/bf02434835
Chen W, Liu Q, Wang H, Chen W, Johnson RJ, Dong X, Li H, Ba S, Tan J, Luo N, Liu T, He H, Yu X (2011) Prevalence and risk factors of chronic kidney disease: a population study in the Tibetan population. Nephrol Dial Transplant 26(5):1592–1599. doi:10.1093/ndt/gfq608
Li Y, Stamler J, Xiao Z, Folsom A, Tao S, Zhang H (1997) Serum uric acid and its correlates in Chinese adult populations, urban and rural, of Beijing. The PRC-USA Collaborative Study in Cardiovascular and Cardiopulmonary Epidemiology. Int J Epidemiol 26(2):288–296. doi:10.1093/ije/26.2.288
Nan H, Qiao Q, Dong Y, Gao W, Tang B, Qian R, Tuomilehto J (2006) The prevalence of hyperuricemia in a population of the coastal city of Qingdao,China. J Rheumatol 33(7):1346–1350
Li D, Yu X, Zhou X, Siriamornpun S, Wahlqvist ML (2006) Uric acid status and its correlates in Hangzhou urban population. Asia Pac J Clin Nutr 15(1):102–106
Ford DK, Agatha M (1964) Serum uric acid levels of healthy Caucasian, Chinese and Haida Indian Males in British Columbia. Can Med Assoc J 90(23):1295
Jee SH, Lee SY, Kim MT (2004) Serum uric acid and risk of death from cancer, cardiovascular disease or all causes in men. Eur J Cardiovasc Prev Rehabil 11(3):185–191. doi:10.1097/01.hjr.0000130222.50258.22
Akizuki S (1982) Serum uric acid levels among thirty-four thousand people in Japan. Ann Rheum Dis 41(3):272–274. doi:10.1136/ard.41.3.272
Nagahama K, Iseki K, Inoue T, Touma T, Ikemiya Y, Takishita S (2004) Hyperuricemia and cardiovascular risk factor clustering in a screened cohort in Okinawa, Japan. Hypertens Res 27(4):227. doi:10.1291/hypres.27.227
Yano K, Rhoads GG, Kagan A (1977) Epidemiology of serum uric acid among 8000 Japanese-American men in Hawaii. J Chronic Dis 30(3):171–184. doi:10.1016/0021-9681(77)90083-2
Lohsoonthorn V, Dhanamun B, Williams MA (2006) Prevalence of hyperuricemia and its relationship with metabolic syndrome in Thai adults receiving annual health exams. Arch Med Res 37(7):883–889. doi:10.1016/j.arcmed.2006.03.008
Mikkelsen WM, Dodge HJ, Valkenburg H, Himes S (1965) The distribution of serum uric acid values in a population unselected as to gout or hyperuricemia: Tecumseh, Michigan 1959–1960. Am J Med 39(2):242–251
Acheson RM (1969) Social class gradients and serum uric acid in males and females. BMJ 4(5675):65. doi:10.1136/bmj.4.5675.65
Zhu Y, Pandya BJ, Choi HK (2011) Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum 63(10):3136–3141. doi:10.1002/art.30520
O’Brien WM, Burch TA, Bunim JJ (1966) Genetics of hyperuricaemia in Blackfeet and Pima Indians. Ann Rheum Dis 25:117–119. doi:10.1136/annrheumd00507-0011
Voruganti VS, Göring HH, Mottl A, Franceschini N, Haack K, Laston S, Almasy L, Fabsitz R, Lee E, Best L, Devereux R, Howard B, MacCluer J, Comuzzie A, Umans J, Cole S (2009) Genetic influence on variation in serum uric acid in American Indians: the strong heart family study. Hum Genet 126(5):667–676. doi:10.1007/s00439-009-0716-8
Acheson R, Florey C (1969) Body-weight, ABO blood-groups, and altitude of domicile as determinants of serum uric acid in military recruits in four countries. Lancet 294(7617):391–395. doi:10.1016/s0140-6736(69)90107-x
Tavares E, Vieira-Filho J, Andriolo A, Sanudo A (2003) Metabolic profile and cardiovascular risk patterns in an indigenous population of Amazonia. Hum Biol 75(1):31–46
Gimeno SGA, Rodrigues D, Canó EN, Lima EES, Schaper M, Pagliaro H, Lafer MM, Baruzzi RG (2009) Cardiovascular risk factors among Brazilian Karib indigenous peoples: upper Xingu, Central Brazil, 2000–3. J Epidemiol Community Health 63(4):299–304. doi:10.1136/jech.2008.077966
Sturge RA, Scott JT, Kennedy AC, Hart DP, Buchanan WW (1977) Serum uric acid in England and Scotland. Ann Rheum Dis 36(5):420–427. doi:10.1136/ard.36.5.420
Badley EM, Meyrick JS, Wood PHN (1978) Gout and serum uric acid levels in the Cotwolds. Rheumatology 17(3):133–142. doi:10.1093/rheumatology/17.3.133
Reunanen A, Takkunen H, Knekt P, Aromaa A (1982) Hyperuricemia as a risk factor for cardiovascular mortality. Acta Med Scand 212(S668):49–59. doi:10.1111/j.0954-6820.1982.tb08521.x
Zalokar J, Lellouch J, Claude JR, Kuntz D (1972) Serum uric acid in 23,923 men and gout in a subsample of 4257 men in France. J Chronic Dis 25(5):305–312. doi:10.1016/0021-9681(72)90166-x
Strasak A, Ruttmann E, Brant L, Kelleher C, Klenk J, Concin H, Diem G, Pfeiffer K, Ulmer H; the VHM&PP Study Group (2008) Serum uric acid and risk of cardiovascular mortality: a prospective long-term study of 83 683 Austrian men. Clin Chem 54(2):273–284. doi:10.1373/clinchem.2007.094425
Sari I, Akar S, Pakoz B, Sisman A, Gurler O, Birlik M, Onen F, Akkoc N (2009) Hyperuricemia and its related factors in an urban population, Izmir, Turkey. Rheumatol Int 29(8):869–874. doi:10.1007/s00296-008-0806-2
Goldbourt U, Medalie JH, Herman JB, Neufeld HN (1980) Serum uric acid: correlation with biochemical, anthropometric, clinical and behavioral parameters in 10,000 Israeli men. J Chronic Dis 33(7):435–443. doi:10.1016/0021-9681(80)90040-5
Snook C, Chang W (1978) Serum uric acid in Jordanian Arabs. J Trop Med Hyg 81:166–171
Hadidy S, Ibrahim AS, Hallag Z, Tabbakh H (1983) Normal serum concentrations of uric acid among a Syrian population. J Trop Med Hyg 86:47–50
Onsi A, Younis K (1961) Blood uric acid in Kuwait. J Kuwait Med Assoc 2:208–210
Al-Aref SA (1971) Hyperuricaemia: an epidemiological study. J Kuwait Med Assoc 5:207–214
Bacchus R, Kilshaw BH, Madkour M, Bassam SA, Farhan BA (1980) Preliminary studies on a reference range for Saudi Arabian males: (1) serum uric acid. Saudi Med J 1(3):160–163
Al-Arfaj AS (2001) Hyperuricemia in Saudi Arabia. Rheumatol Int 20(2):61–64. doi:10.1007/s002960000076
Beighton P, Solomon L, Soskolne C, Sweet B (1973) Serum uric acid concentrations in a rural Tswana community in Southern Africa. Ann Rheum Dis 32(4):346–350. doi:10.1136/ard.32.4.346
Beighton P, Solomon L, Soskolne C, Sweet B, Robin G (1974) Serum uric acid concentrations in an urbanized South African Negro population. Ann Rheum Dis 33(5):442–445. doi:10.1136/ard.33.5.442
Beighton P, Daynes G, Soskolne CL (1976) Serum uric acid concentrations in a Xhosa community in the Transkei of Southern Africa. Ann Rheum Dis 35(1):77–80. doi:10.1136/ard.35.1.77
Palmer IM, Schutte AE, Huisman HW (2010) Uric acid and the cardiovascular profile of African and Caucasian men. J Hum Hypertens 24(10):639–645. doi:10.1038/jhh.2010.1
Kayembe D, Sigg-Farner C, Deplaen J (1973) La goutte dans un hopital universitaire Africain. Ann Soc Belg Med Trop 53(2):89–95
Snook C, Cartwright I (1975) Serum uric acid in Zambians. East Afr Med J 52(4):196–201
Saathoff E, Schneider P, Kleinfeldt V, Geis S, Haule D, Maboko L, Samky E, de Souza M, Robb M, Hoelscher M (2008) Laboratory reference values for healthy adults from southern Tanzania. Trop Med Int Health 13(5):612–625. doi:10.1111/j.1365-3156.2008.02047.x
Author information
Authors and Affiliations
Corresponding author
Additional information
Elizabeth Matisoo-Smith and Tony R. Merriman have contributed equally to this study.
Appendix: Mean SU levels and prevalence of gout and hyperuricaemia in different adult male populations worldwide summarised from the published literature
Appendix: Mean SU levels and prevalence of gout and hyperuricaemia in different adult male populations worldwide summarised from the published literature
Location | Population | n | Age range | Mean SU | Prevalence | References | ||
|---|---|---|---|---|---|---|---|---|
mg/dL | mmol/L | HU (%) | Gout (%) | |||||
Oceania | ||||||||
New Zealand | Māori | 388 | >15 years | 7.1 ± 1.5 | 0.42 ± 0.09 | 49 | – | [94] |
New Zealand | Māori | 215 | 20–64 years | – | – | 27.8 | 18.4 | [95] |
New Zealand | NZ “non-Māori” | 125 | 20–64 years | – | – | 15.3 | 4.0 | [95] |
Cook Islands | Rarotongans | 243 | >20 years | 7.0 ± 1.4 | 0.41 ± 0.08 | 44 | 2.5 | [96] |
Cook Islands | Pukapukans | 188 | >20 years | 7.0 ± 1.1 | 0.42 ± 0.07 | 48.5 | 5.4 | [96] |
Papeete, Tahiti | Tahitians | 179 | 20–50 years | 7.5 | 0.45 | – | – | [97] |
Hawaii | Hawaiians | 49 | 22–72 years | 5.4 ± 1.1 | 0.32 ± 0.07 | – | – | [13] |
Kaneohe, Hawaii | Hawaiians | 11 | “Adult” | 6.4 ± 1.4 | 0.38 ± 0.08 | – | – | [98] |
Apia, Samoa | Samoans (urban) | 325 | >20 years | 6.7 ± 1.4 | 0.40 ± 0.08 | 36.4 | 7.2 | [20] |
Samoa | Samoans (rural) | 358 | >20 years | 6.9 ± 1.2 | 0.41 ± 0.07 | 43.3 | 6.2 | [20] |
American Samoa | American Samoans | 71 | – | 6.8 ± 1.2 | 0.40 ± 0.07 | – | – | [99] |
Nuku’alofa, Tonga | Tongans (urban) | 177 | 20–69 years | 6.5 ± 0.1 | 0.39 ± 0.00 | – | 2.7 | [21] |
Foa Island, Tonga | Tongans (rural) | 199 | 20–69 years | 5.3 ± 0.0 | 0.32 ± 0.00 | – | 0 | [21] |
Suva, Fiji | Fijians (urban) | 401 | >20 years | 6.6 ± 1.2 | 0.39 ± 0.07 | 32.4 | 0.002 | [22] |
Fiji | Fijians (rural) | 242 | >20 years | 6.0 ± 1.0 | 0.36 ± 0.06 | 16.9 | 0 | [22] |
Tokelau | Tokelauans | 433 | 35–74 years | 6.6 ± 1.3 | 0.39 ± 0.08 | – | – | [100] |
Tuvalu | Tuvaluans | 189 | >20 years | 6.6 | 0.39 | 32.3 | 0 | [101] |
Nauru | Nauruans | 217 | >20 years | 7.6 ± 1.6 | 0.45 ± 0.10 | 64 | 6.9 | [102] |
Marshall Islands | Marshall Islanders | 188 | >15 years | 6.6 ± 1.3 | 0.39 ± 0.08 | – | 1.7 | [103] |
Rongelap, Marshall Islands | Marshall Islanders | 37 | >15 years | 6.1 ± 1.0 | 0.36 ± 0.06 | – | – | [103] |
Utirik, Marshall Islands | Marshall Islanders | 68 | >15 years | 6.5 ± 1.4 | 0.39 ± 0.08 | – | – | [103] |
Ebeye, Marshall Islands | Marshall Islanders | 45 | >15 years | 6.9 ± 1.4 | 0.41 ± 0.08 | – | – | [103] |
Majuro, Marshall Islands | Marshall Islanders | 29 | >15 years | 6.9 ± 1.2 | 0.41 ± 0.07 | – | – | [103] |
Caroline Islands | Carolinians | 26 | >40 years | 7.3 ± 1.7 | 0.43 ± 0.10 | – | – | [104] |
Mariana Islands | Chamorro | 160 | >40 years | 6.2 ± 1.5 | 0.37 ± 0.09 | – | – | [104] |
Guam, Mariana Islands | Chamorro (urban) | 273 | >20 years | 7.1 ± 1.5 | 0.42 ± 0.09 | 48 | 1.5 | [18] |
Rota, Mariana Islands | Chamorro (rural) | 122 | >20 years | 7.2 ± 1.4 | 0.43 ± 0.08 | 52 | 4.1 | [18] |
Palau | Palauans | 510 | >20 years | 6.7 ± 1.6 | 0.40 ± 0.10 | 37 | 1.3 | [18] |
Kwaio, Solomon Islands | Solomon Islanders | 127 | 18–54 years | 6.5 ± 1.6 | 0.39 ± 0.10 | – | – | [105] |
Aita, Solomon Islands | Solomon Islanders | 81 | 18–54 years | 6.1 ± 1.1 | 0.36 ± 0.07 | – | – | [105] |
Baegu, Solomon Islands | Solomon Islanders | 126 | 18–54 years | 6.2 ± 1.6 | 0.37 ± 0.09 | – | – | [105] |
Lau, Solomon Islands | Solomon Islanders | 77 | 18–54 years | 5.5 ± 0.9 | 0.33 ± 0.06 | – | – | [105] |
Nagovisi, Solomon Islands | Solomon Islanders | 109 | 18–54 years | 5.3 ± 0.9 | 0.31 ± 0.05 | – | – | [105] |
Nasioi, Solomon Islands | Solomon Islanders | 59 | 18–54 years | 6.0 ± 0.9 | 0.36 ± 0.05 | – | – | [105] |
Kitava, Trobiand Island, PNG | Trobriand Islanders | 41 | 40–59 years | 5.1 | 0.30 | – | – | [106] |
East Sepik, PNG | Salatas | 71 | 4.9 ± 1.4 | 0.29 ± 0.09 | – | – | [17] | |
Eastern Highlands Province, PNG | Lufa (Highlanders) | 329 | >15 years | 5.6 | 0.32 | – | – | [86] |
Simbu Province, PNG | Nomane (Highlanders) | 39 | >15 years | 6.1 | 0.36 | – | – | [86] |
Oro Province, PNG | Managalas (Highlanders) | 20 | >15 years | 5.5 | 0.33 | – | – | [86] |
Madang Coast. PNG | Madang Coast (Lowlanders) | 43 | >15 years | 4.8 | 0.29 | – | – | [86] |
Karkar Island, PNG | Karkar Islanders | 260 | >15 years | 5.7 | 0.34 | – | – | [86] |
Western Province, PNG | Kiunga (Lowlanders) | 32 | >15 years | 5.3 | 0.32 | – | – | [86] |
Tukisenta, Western Highlands, PNG | Murapin Highlanders | 391 | >15 years | 5.4 | 0.32 | – | – | [107] |
Koki, Port Moresby, PNG | “Melanesians” (urban) | 86 | >18 years | 6.2 ± 1.3 | 0.37 ± 0.08 | 20 | – | [19] |
Kalo Village, Central Province, PNG | “Melanesians” (rural) | 22 | >18 years | 4.2 ± 1.3 | 0.25 ± 0.08 | – | – | [19] |
Australia | Australian Caucasians | 200 | 30–39 years | 6.6 ± 0.2 | 0.39 ± 0.01 | 12 | – | [108] |
Australia | Representative Australian population | 1,673 | 70–97 years | 6.0 ± 1.3 | 0.36 ± 0.08 | 23 | – | [109] |
Mitchell River, Australia | Aborigines | 27 | 20–98 years | 5.5 ± 1.0 | 0.33 ± 0.06 | – | – | [110] |
Aurukun, Australia | Aborigines | 82 | 20–70 years | 6.0 ± 1.2 | 0.36 ± 0.07 | – | – | [110] |
Weipa, Australia | Aborigines | 3 | >20 years | 5.3 ± 1.7 | 0.31 ± 0.10 | – | – | [110] |
Island Southeast Asia | ||||||||
Java, Indonesia | Malayo-Polynesians | 2,184 | >15 years | 6.2 ± 1.3 | 0.37 ± 0.08 | 24.3 | 1.7 | [111] |
Manila, Philippines | Filipinos (urban) | 140 | 21–87 years | 5.3 ± 1.4 | 0.32 ± 0.08 | – | – | [16] |
Cebu, Philippines | Filipinos (rural) | 146 | 21–87 years | 5.1 ± 1.2 | 0.30 ± 0.07 | – | – | [16] |
La Union, Philippines | Filipinos (rural) | 197 | 21–87 years | 5.3 ± 1.3 | 0.32 ± 0.08 | – | – | [16] |
Seattle, USA | Filipinos | 113 | 21–87 years | 6.3 ± 1.4 | 0.37 ± 0.08 | – | – | [16] |
Hawaii | Filipinos (urban) | 60 | 21–87 years | 6.1 ± 1.3 | 0.36 ± 0.08 | – | – | [16] |
Singapore, Malaysia | Malays (urban) | 169 | 15–74 years | 6.3 ± 1.3 | 0.38 ± 0.08 | – | – | [17] |
Lenggong, Malaysia | Malays (rural) | 35 | 15–74 years | 7.1 ± 1.5 | 0.42 ± 0.09 | – | – | [17] |
Taiwan | Aborigines | 145 | – | 7.9 ± 1.7 | 0.47 ± 0.10 | 53.8 | 11.7 | [29] |
Taiwan | Taiwanese Aborigines and Han Chinese | 1,348 | >18 years | 6.7 | 0.40 | 26.1 | 3.3 | [93] |
Mainland Asia | ||||||||
India | Indians (rural) | 85 | 20–70 years | 5.3 ± 1.7 | 0.31 ± 0.10 | – | – | [112] |
Tirupati, India | Indians (urban) | 97 | 20–70 years | 6.1 ± 2.0 | 0.36 ± 0.12 | – | – | [112] |
Lhasa City, Tibet, China | Tibetans | 1,289 | >18 years | 6.9 ± 1.6 | 0.41 ± 1.0 | – | – | [113] |
Beijing, China | Han Chinese (rural) | 558 | 40–58 years | 5.6 ± 1.3 | 0.33 ± 0.08 | 11.3 | – | [114] |
Beijing, China | Han Chinese (urban) | 1,062 | 40–58 years | 5.8 ± 1.3 | 0.34 ± 0.08 | 15.4 | – | [114] |
Qingdao, China | Han Chinese | 903 | 20–74 years | 6.5 | 0.39 | 32.1 | – | [115] |
Hangzhou, China | Han Chinese | 186 | “adults” | 5.5 ± 1.2 | 0.33 ± 0.07 | 11 | – | [116] |
Vancouver, Canada | Canadian Chinese | 100 | – | 5.4 ± 1.1 | 0.32 ± 0.06 | – | – | [117] |
Korea | Koreans | 22,698 | 30–77 years | 5.8 ± 1.6 | 0.35 ± 0.10 | – | – | [118] |
Nagano Prefecture, Japan | Japanese | 15,712 | >15 years | 5.8 ± 1.1 | 0.35 ± 0.06 | – | – | [119] |
Okinawa, Japan | Japanese | 6,163 | 18–89 years | 6.5 ± 1.3 | 0.39 ± 0.08 | 34.5 | – | [120] |
Hawaii, USA | Japanese Americans | 7,971 | 45–69 years | 6.0 ± 1.5 | 0.36 ± 0.09 | – | – | [121] |
Thailand | Thai | 376 | – | 6.0 ± 1.4 | 0.36 ± 0.08 | 18.4 | – | [122] |
Americas | ||||||||
Vancouver, Canada | Caucasian Canadians | 200 | – | 4.6 ± 1.0 | 0.27 ± 0.06 | – | – | [117] |
Canada | Haida (Amerindians) | 236 | – | 4.4 ± 1.0 | 0.26 ± 0.06 | – | – | [117] |
Tecumeseh, Michigan, USA | Caucasian Americans | 2,987 | >3 years | 4.9 ± 2.0 | 0.29 ± 0.12 | 7.5 | – | [123] |
New England, USA | Caucasian Americans | 534 | >20 years | 6.4 ± 1.4 | 0.38 ± 0.08 | – | – | [124] |
NHANES Cohort, USA | Americans of multiple ancestral backgrounds | 8,816 | >20 years | 6.1 | 0.36 | 21.2 | 5.9 | [125] |
Montana, USAa | Blackfeet (Amerindians) | 1,018 | – | 4.9 ± 1.2 | 0.29 ± 0.07 | 7.17 | – | [126] |
Arizona, USAa | Pima (Amerindians) | 949 | – | 4.2 ± 1.2 | 0.25 ± 0.07 | 3.27 | – | [126] |
Arizona, USA | “American Indians” | – | >18 years | 5.7 ± 1.4 | 0.33 ± 0.08 | – | – | [127] |
Dakotas, USA | “American Indians” | – | >18 years | 6.1 ± 1.5 | 0.36 ± 0.09 | – | – | [127] |
Oklahoma, USA | “American Indians” | – | >18 years | 6.1 ± 1.4 | 0.36 ± 0.08 | – | – | [127] |
USA | American Army Recruits | 2,084 | – | 4.9 | 0.29 | – | – | [128] |
Colombia | Colombian Army Recruits | 481 | – | 5.0 | 0.30 | – | – | [128] |
Argentina | Argentinian Army Recruits | 491 | – | 4.8 | 0.29 | – | – | [128] |
Brazil | Brazilian Army Recruits | 1,990 | – | 4.1 | 0.24 | – | – | [128] |
Brazil | Parkatêjê (Amerindians) | 53 | 20–80 years | 5.8 ± 0.9 | 0.34 ± 0.05 | 5.6 | – | [129] |
Brazil | Kalapalo (Amerindians) | 53 | 20–80 years | 5.1 ± 1.3 | 0.30 ± 0.07 | 3.2 | – | [130] |
Brazil | Kuikuro (Amerindians) | 54 | 20–80 years | 4.6 ± 1.0 | 0.27 ± 0.06 | 0 | – | [130] |
Brazil | Matipo (Amerindians) | 5 | 20–80 years | 5.1 ± 0.9 | 0.30 ± 0.05 | 0 | – | [130] |
Brazil | Nahukwá (Amerindians) | 13 | 20–80 years | 4.5 ± 1.0 | 0.27 ± 0.06 | 0 | – | [130] |
Europe | ||||||||
Birmingham and Dorset, England | English | 512 | >15 years | 5.5 ± 1.0 | 0.33 ± 0.06 | 6.6 | – | [131] |
Cotwolds, England | English | 1,727 | >35 years | 5.1 ± 1.4 | 0.30 ± 0.08 | 6.75 | 1.68 | [132] |
Watford, England | English | 158 | >15 years | 5.0 ± 1.2 | 0.30 ± 0.07 | – | – | [124] |
Wensleydale, England | English (rural) | 435 | >15 years | 4.7 ± 0.9 | 0.28 ± 0.05 | – | – | [124] |
Glasgow, Scotland | Scottish (urban) | 337 | >15 years | 5.5 ± 1.0 | 0.33 ± 0.06 | 8.0 | – | [131] |
Finland | Finnish | 3,295 | 40–69 years | 5.1 ± 1.2 | 0.30 ± 0.07 | 6.6 | – | [133] |
France | French | 23,923 | 20–55 years | 5.9 ± 1.2 | 0.35 ± 0.07 | 17.6 | – | [134] |
Austria | Austrians | 83,683 | >18 years | 5.7 ± 1.4 | 0.34 ± 0.08 | – | – | [135] |
Izmir, Turkey | Turks (urban) | 63 | “Adult” | 5.5 ± 1.3 | 0.33 ± 0.07 | 19.0 | – | [136] |
Middle East | ||||||||
Israel | Israelis | 9,909 | >40 years | 4.8 ± 1.0 | 0.28 ± 0.06 | – | – | [137] |
Jordan | Jordanians | 96 | 18–60 years | 5.7 ± 1.1 | 0.35 ± 0.07 | – | – | [138] |
Syria | Syrians | 1,042 | 20–60 years | 5.2 ± 1.2 | 0.31 ± 0.07 | – | – | [139] |
Kuwait | Kuwait | 110 | 20–40 years | 4.7 ± 0.9 | 0.28 ± 0.05 | – | – | [140] |
Kuwait | Kuwait | 79 | >20 years | 5.5 ± 1.4 | 0.33 ± 0.08 | – | – | [141] |
Saudi Arabia | Saudis | 278 | 14–65 years | 5.2 ± 1.0 | 0.31 ± 0.06 | – | – | [142] |
Saudi Arabia | Saudis | 250 | 14–83 years | 5.2 ± 1.5 | 0.31 ± 0.09 | 8 | – | [143] |
Africa | ||||||||
Phokeng, South Africa | Tswana (rural) | 128 | 14–84 years | 4.9 ± 1.9 | 0.29 ± 0.11 | – | – | [144] |
Soweto, South Africa | Tswana, Zuli and Xhosa (urban) | 144 | 15–90 years | 6.1 ± 1.4 | 0.36 ± 0.08 | – | – | [145] |
Transkei, South Africa | Xhosa (tribal) | 80 | >15 years | 4.7 ± 1.1 | 0.28 ± 0.06 | – | – | [146] |
South Africa | “Africans” | 87 | “Adult” | 5.9 ± 1.5 | 0.35 ± 0.09 | – | – | [147] |
South Africa | Caucasians | 121 | “Adult” | 6.7 ± 1.6 | 0.40 ± 0.10 | – | – | [147] |
Zairea | “Zairois” | 75 | – | 5.6 ± 1.1 | 0.33 ± 0.06 | – | – | [148] |
Kinshasa, Zairea | “Zairois” (urban) | 100 | – | 5.7 ± 1.2 | 0.34 ± 0.07 | – | – | [148] |
Kasongo-Batetela, Zaire | “Zairois” (rural) | 33 | – | 5.5 ± 1.1 | 0.33 ± 0.06 | – | – | [148] |
Zairea | Caucasians | 19 | – | 5.4 ± 1.1 | 0.32 ± 0.06 | – | – | [148] |
Zambia | Zambians | 50 | – | 5.2 ± 1.5 | 0.31 ± 0.09 | – | – | [149] |
Mbeya, Tanzania | Tanzanians (mixed tribes) | 137 | 19–48 years | 5.3 | 0.32 | – | – | [150] |
Seychelles | Seychellois | 482 | 25–64 years | 6.9 ± 2.8 | 0.41 ± 0.17 | 35.2 | – | [33] |
Sudan | Sudanese | 16 | 20–22 years | 4.3 ± 0.2 | 0.26 ± 0.01 | – | – | [67] |
Rights and permissions
About this article
Cite this article
Gosling, A.L., Matisoo-Smith, E. & Merriman, T.R. Hyperuricaemia in the Pacific: why the elevated serum urate levels?. Rheumatol Int 34, 743–757 (2014). https://doi.org/10.1007/s00296-013-2922-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-013-2922-x