Abstract
Purpose
Spatial covariance analysis has been used with FDG PET to identify a specific metabolic network associated with Parkinson’s disease (PD). In the current study, we utilized a new, fully automated voxel-based method to quantify network expression in ECD SPECT images from patients with classical PD, patients with multiple system atrophy (MSA), and healthy control subjects.
Methods
We applied a previously validated voxel-based PD-related covariance pattern (PDRP) to quantify network expression in the ECD SPECT scans of 35 PD patients, 15 age- and disease severity-matched MSA patients, and 35 age-matched healthy control subjects. PDRP scores were compared across groups using analysis of variance. The sensitivity and specificity of the prospectively computed PDRP scores in the differential diagnosis of individual subjects were assessed by receiver operating characteristic (ROC) analysis.
Results
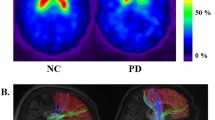
PDRP scores were significantly increased (p < 0.001) in the PD group relative to the MSA and control groups. ROC analysis indicated that the overall diagnostic accuracy of the PDRP measures was 0.91 (AUC). The optimal cutoff value was consistent with a sensitivity of 0.97 and a specificity of 0.80 and 0.71 for discriminating PD patients from MSA and normal controls, respectively.
Conclusion
Our findings suggest that fully automated voxel-based network assessment techniques can be used to quantify network expression in the ECD SPECT scans of parkinsonian patients.



Similar content being viewed by others
References
Lang AE, Lozano AM. Parkinson’s disease. First of two parts. N Engl J Med 1998;339:1044–53.
Gelb DJ, Oliver E, Gilman S. Diagnostic criteria for Parkinson disease. Arch Neurol 1999;56:33–9.
Hughes AJ, Daniel SE, Kilford L, Lees AJ. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 1992;55:181–4.
Vanacore N. Epidemiological evidence on multiple system atrophy. J Neural Transm 2005;112:1605–12.
Eckert T, Eidelberg D. The role of functional neuroimaging in the differential diagnosis of idiopathic Parkinson’s disease and multiple system atrophy. Clin Auton Res 2004;14:84–91.
Eckert T, Sailer M, Kaufmann J, Schrader C, Peschel T, Bodammer N, et al. Differentiation of idiopathic Parkinson’s disease, multiple system atrophy, progressive supranuclear palsy, and healthy controls using magnetization transfer imaging. Neuroimage 2004;21:229–35.
Schrag A, Good CD, Miszkiel K, Morris HR, Mathias CJ, Lees AJ, et al. Differentiation of atypical parkinsonian syndromes with routine MRI. Neurology 2000;54:697–702.
Seppi K, Schocke MF, Esterhammer R, Kremser C, Brenneis C, Mueller J, et al. Diffusion-weighted imaging discriminates progressive supranuclear palsy from PD, but not from the parkinson variant of multiple system atrophy. Neurology 2003;60:922–7.
Van Laere K, Santens P, Bosman T, De Reuck J, Mortelmans L, Dierckx R. Statistical parametric mapping of 99mTc-ECD SPECT in idiopathic Parkinson’s disease and multiple system atrophy with predominant parkinsonian features: correlation with clinical parameters. J Nucl Med 2004;45:933–42.
Eckert T, Barnes A, Dhawan V, Frucht S, Gordon MF, Feigin AS, et al. FDG PET in the differential diagnosis of parkinsonian disorders. Neuroimage 2005;26:912–21.
Eckert T, Eidelberg D. Neuroimaging and therapeutics in movement disorders. Neurorx 2005;2:361–71.
Ma Y, Tang C, Spetsieres P, Dhawan V, Eidelberg D. Abnormal metabolic network activity in Parkinson’s disease: test-retest reproducibility. J Cereb Blood Flow Metab 2006; in press.
Moeller JR, Nakamura T, Mentis MJ, Dhawan V, Spetsieres P, Antonini A, et al. Reproducibility of regional metabolic covariance patterns: comparison of four populations. J Nucl Med 1999;40:1264–9.
Lozza C, Baron JC, Eidelberg D, Mentis MJ, Carbon M, Marie RM. Executive processes in Parkinson’s disease: FDG-PET and network analysis. Hum Brain Mapp 2004;22:236–45.
Asanuma K, Ma Y, Huang C, Carbon-Correll M, Edwards C, Raymond D, et al. The metabolic pathology of dopa-responsive dystonia. Ann Neurol 2005;57:596–600.
Feigin A, Fukuda M, Dhawan V, Przedborski S, Jackson-Lewis V, Mentis MJ, et al. Metabolic correlates of levodopa response in Parkinson’s disease. Neurology 2001;57:2083–8.
Asanuma K, Tang C, Ma Y, Dhawan V, Mattis P, Edwards C, et al. Network modulation in the treatment of Parkinson’s disease. Brain 2006;129:2667–78.
Fukuda M, Mentis MJ, Ma Y, Dhawan V, Antonini A, Lang AE, et al. Networks mediating the clinical effects of pallidal brain stimulation for Parkinson’s disease: a PET study of resting-state glucose metabolism. Brain 2001;124:1601–9.
Trošt M, Su S, Su P, Yen RF, Tseng HM, Barnes A, et al. Network modulation by the subthalamic nucleus in the treatment of Parkinson’s disease. Neuroimage 2006;31:301–7.
Feigin A, Antonini A, Fukuda M, De Notaris R, Benti R, Pezzoli G, et al. Tc-99m ethylene cysteinate dimer SPECT in the differential diagnosis of parkinsonism. Mov Disord 2002;17:1265–70.
Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology 1967;17:427–42.
Wenning GK, Ben Shlomo Y, Magalhaes M, Daniel SE, Quinn NP. Clinical features and natural history of multiple system atrophy. An analysis of 100 cases. Brain 1994;117:835–45.
Van Laere K, Koole M, Versijpt J, Vandenberghe S, Brans B, D’sAsseler Y, et al. Transfer of normal 99mTc-ECD brain SPET databases between different gamma cameras. Eur J Nucl Med 2001;28:435–49.
Spetsieris P, Ma Y, Dhawan V, Moeller J, Eidelberg D. Highly automated computer-aided diagnosis of neurological disorders using functional brain imaging. Proc SPIE: Medical Imaging 2006;6144:61445M1–12.
Ludbrook J. Multiple comparison procedures updated. Clin Exp Pharmacol Physiol 1998;25:1032–7.
Obuchowski NA. Receiver operating characteristic curves and their use in radiology. Radiology 2003;229:3–8.
Huang C, Feigin A, Ma Y, Eidelberg D. Imaging measures of longitudinal change in Parkinson’s disease. Neurology 2005;64(6, Suppl 1):A325.
Moeller JR, Eidelberg D. Divergent expression of regional metabolic topographies in Parkinson’s disease and normal ageing. Brain 1997;120:2197–206.
Eidelberg D, Moeller JR, Ishikawa T, Dhawan V, Spetsieris P, Chaly T, et al. Assessment of disease severity in parkinsonism with fluorine-18-fluorodeoxyglucose and PET. J Nucl Med 1995;36:378–83.
Eidelberg D, Moeller JR, Dhawan V, Spetsieris P, Takikawa S, Ishikawa T, et al. The metabolic topography of parkinsonism. J Cereb Blood Flow Metab 1994;14:783–801.
Eidelberg D, Moeller JR, Ishikawa T, Dhawan V, Spetsieris P, Chaly T, et al. Early differential diagnosis of Parkinson’s disease with18F-fluorodeoxyglucose and positron emission tomography. Neurology 1995;45:1995–2004.
Alsop DC, Detre JA, Grossman M. Assessment of cerebral blood flow in Alzheimer’s disease by spin-labeled magnetic resonance imaging. Ann Neurol 2000;47:93–100.
Kobari M, Fukuuchi Y, Shinohara T, Obara K, Nogawa S. Levodopa-induced local cerebral blood flow changes in Parkinson’s disease and related disorders. J Neurol Sci 1995;128:212–8.
Ma Y, Eckert T, Feigin A, Dhawan V, Eidelberg D. Abnormal metabolic topography in patients with multiple systems atrophy: a FDG PET study. Neurology 2005;64(6, suppl 1):A235.
Eidelberg D, Takikawa S, Moeller JR, Dhawan V, Redington K, Chaly T, et al. Striatal hypometabolism distinguishes striatonigral degeneration from Parkinson’s disease. Ann Neurol 1993;33:518–27.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eckert, T., Van Laere, K., Tang, C. et al. Quantification of Parkinson’s disease-related network expression with ECD SPECT. Eur J Nucl Med Mol Imaging 34, 496–501 (2007). https://doi.org/10.1007/s00259-006-0261-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-006-0261-9