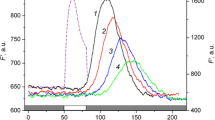
Abstract
Emerging evidence suggests that cytoplasmic streaming can regulate the plasma-membrane H+ transport and photosynthetic electron flow. Microfluorometric and surface pH measurements on Chara corallina internodes revealed the transmission of photoinduced signals by the cytoplasmic flow for a distance of few millimeters from the site of stimulus application. When a 30-s pulse of bright light was locally applied, the downstream cell regions responded with either release or enhancement of non-photochemical quenching of chlorophyll fluorescence, depending on the background irradiance of the analyzed cell area. Under dim background irradiance (<20 μmol m−2 s−1), the arrival of the distant signal from the brightly illuminated 400-μm-wide zone elevated the maximal fluorescence F ′m in the analyzed downstream area, whereas at higher background irradiances it induced strong quenching of F ′m . At intermediate irradiances the increase and decrease in F ′m appeared as two successive waves. The transition between the F ′m responses of opposite polarities occurred at a narrow threshold range of irradiances. This indicates that inevitable slight variations in irradiance at the bottom chloroplast layer combined with the cyclosis-transmitted signals may contribute to the formation of a photosynthetic activity pattern. The rapid cyclosis-mediated release of non-photochemical quenching, unlike the delayed response of opposite polarity, was associated with opening of H+ (OH−)-conducting plasma membrane channels, as evidenced by the concurrent alkaline pH shift on the cell surface. It is proposed that the initial increase in F ′m after application of a distant photostimulus is determined, among other factors, by the wave of alkaline cytoplasmic pH.









Similar content being viewed by others
Abbreviations
- AOI:
-
Area of inspection
- F ′m :
-
The maximum chlorophyll fluorescence induced by saturating light pulse under actinic light
- NPQ:
-
Non-photochemical quenching
- PFD:
-
Photon flux density
- PSII:
-
Photosystem II
References
Allen NS, Allen RD (1978) Cytoplasmic streaming in green plants. Annu Rev Biophys Bioenerg 7:497–526
Alova AV, Bulychev AA, Cherkashin AA (2012) Longitudinal profiles of the vacuolar pH in internally perfused cells of Characean alga. Biochem (Mosc) Suppl Ser A Membr Cell Biol 6:227–234
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59:89–113
Beilby MJ, Bisson MA (2012) pH banding in charophyte algae. In: Volkov AG (ed) Plant electrophysiology: methods and cell electrophysiology. Springer, Berlin, pp 247–271
Berestovsky GN, Kataev AA (2005) Voltage-gated calcium and Ca2+-activated chloride channels and Ca2+ transients: voltage-clamp studies of perfused and intact cells of Chara. Eur Biophys J 34:973–986
Bulychev AA (2012) Membrane excitation and cytoplasmic streaming as modulators of photosynthesis and proton flows in Characean cells. In: Volkov AG (ed) Plant electrophysiology: methods and cell electrophysiology. Springer, Berlin, pp 273–300
Bulychev AA, Dodonova SO (2011) Effects of cyclosis on chloroplast–cytoplasm interactions revealed with localized lighting in Characean cells at rest and after electrical excitation. Biochim Biophys Acta 1807:1221–1230
Bulychev AA, Polezhaev AA, Zykov SV, Pljusnina TY, Riznichenko GY, Rubin AB, Jantoss W, Zykov VS, Mueller SC (2001) Light-triggered pH banding profile in Chara cells revealed with a scanning pH microprobe and its relation to self-organization phenomena. J Theor Biol 212:275–294
Dodonova SO, Bulychev AA (2011) Cyclosis-related asymmetry of chloroplast–plasma membrane interactions at the margins of illuminated area in Chara corallina cells. Protoplasma 248:737–749
Feild TS, Nedbal L, Ort DR (1998) Nonphotochemical reduction of the plastoquinone pool in sunflower leaves originates from chlororespiration. Plant Physiol 116:1209–1218
Felle H, Bertl A (1986) Light-induced cytoplasmic pH changes and their interrelation to the activity of the electrogenic proton pump in Riccia fluitans. Biochim Biophys Acta 848:176–182
Foissner I (2004) Microfilaments and microtubules control the shape, motility, and subcellular distribution of cortical mitochondria in characean internodal cells. Protoplasma 224:145–157
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875
Foyer CH, Noctor G (2009) Redox regulation in photosynthetic organisms: signaling, acclimation, and practical implications. Antioxid Redox Signal 11:861–905
Grams TEE, Lautner S, Felle HH, Matyssek R, Fromm J (2009) Heat-induced electrical signals affect cytoplasmic and apoplastic pH as well as photosynthesis during propagation through the maize leaf. Plant, Cell Environ 32:319–326
Hansen U-P, Moldaenke C, Tabrizi H, Ramm D (1993) The effect of transthylakoid proton uptake on cytosolic pH and the imbalance of ATP and NADPH/H+ production as measured by CO2- and light-induced depolarisation of the plasmalemma. Plant Cell Physiol 34:681–695
Harada A, Shimazaki K (2009) Measurement of changes in cytosolic Ca2+ in Arabidopsis guard cells and mesophyll cells in response to blue light. Plant Cell Physiol 50:360–373
Hauser M, Eichelmann H, Oja V, Heber U, Laisk A (1995) Stimulation by light of rapid pH regulation in the chloroplast stroma in vivo as indicated by CO2 solubilization in leaves. Plant Physiol 108:1059–1066
Islam MS, Niwa Y, Takagi S (2009) Light-dependent intracellular positioning of mitochondria in Arabidopsis thaliana mesophyll cells. Plant Cell Physiol 50:1032–1040
Johnson MP, Ruban AV (2011) Restoration of rapidly reversible photoprotective energy dissipation in the absence of PsbS protein by enhanced ΔpH. J Biol Chem 286:19973–19981
Kamiya N (1959) Protplasmic streaming. Springer, Wien
Krupenina NA, Bulychev AA, Schreiber U (2011) Chlorophyll fluorescence images demonstrate variable pathways in the effects of plasma membrane excitation on electron flow in chloroplasts of Chara cells. Protoplasma 248:513–522
Lucas WJ, Dainty J (1977) Spatial distribution of functional OH− carriers along a Characean internodal cell: determined by the effect of cytochalasin B on H14CO3 − assimilation. J Membr Biol 32:75–92
Naydov IA, Mubarakshina MM, Ivanov BN (2012) Formation kinetics and H2O2 distribution in chloroplasts and protoplasts of photosynthetic leaf cells of higher plants under illumination. Biochemistry (Moscow) 77:143–151
Nixon PJ (2000) Chlororespiration. Philos Trans R Soc B 355:1541–1547
Okazaki Y, Tazawa M, Iwasaki N (1994) Light-induced changes in cytosolic pH in leaf cells of Egeria densa: measurements with pH-sensitive microelectrodes. Plant Cell Physiol 35:943–950
Pickard WF (2003) The role of cytoplasmic streaming in symplastic transport. Plant, Cell Environ 26:1–15
Raven JA, Hurd CL (2012) Ecophysiology of photosynthesis in macroalgae. Photosynth Res 113:105–125
Remis D, Bulychev AA, Kurella GA (1988) Photo-induced pH changes in the vicinity of isolated Peperomia metallica chloroplasts. J Exp Bot 39:633–640
Rochaix J-D (2011) Reprint of: regulation of photosynthetic electron transport. Biochim Biophys Acta 1807:878–886
Shimmen T, Yokota E (2004) Cytoplasmic streaming in plants. Curr Opin Cell Biol 16:68–72
Takeshige K, Tazawa M (1989) Measurement of the cytoplasmic and vacuolar buffer capacities in Chara corallina. Plant Physiol 89:1049–1052
Taniguchi M, Miyake H (2012) Redox-shuttling between chloroplast and cytosol: integration of intra-chloroplast and extra-chloroplast metabolism. Curr Opin Plant Biol 15:1–9
Tazawa M (2003) Cell physiological aspects of the plasma membrane electrogenic H+ pump. J Plant Res 116:419–442
van de Meent JW, Sederman AJ, Gladden LF, Goldstein RE (2010) Measurement of cytoplasmic streaming in single plant cells by magnetic resonance velocimetry. J Fluid Mech 642:5–14
Verchot-Lubicz J, Goldstein RE (2010) Cytoplasmic streaming enables the distribution of molecules and vesicles in large plant cells. Protoplasma 240:99–107
Weber APM (2007) Synthesis, export and partitioning of the end products of photosynthesis. In: Wise RR, Hoober JK (eds) The structure and function of plastids. Springer, Dordrecht, pp 273–292
Woodrow IE (1999) Metabolite flux and organelle transporters. In: Atwell BJ, Kriedemann PE, Turnbull CGN (eds) Plants in action. Chapter 2.1.8. Macmillan Education Australia, Melbourne
Acknowledgments
This work was supported by the Russian Foundation for Basic Research (Project No. 10-04-00968-a).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bulychev, A.A., Alova, A.V. & Rubin, A.B. Propagation of photoinduced signals with the cytoplasmic flow along Characean internodes: evidence from changes in chloroplast fluorescence and surface pH. Eur Biophys J 42, 441–453 (2013). https://doi.org/10.1007/s00249-013-0895-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-013-0895-z