Abstract
Purpose
To evaluate whether variability in milrinone blood levels (MBL) occurs during administration to critically ill children after surgical repair of congenital heart disease, and the clinical relevance of this variability.
Methods
Prospective cohort study conducted in the pediatric intensive care unit of a tertiary care teaching and referral hospital. MBL were measured at three time periods after starting milrinone infusion (9–12, 18–24, 40–48 h) and at the end of the infusion. MBL were categorized as within (100–300 ng/ml) or outside the therapeutic range. Low cardiac output syndrome was defined by elevation of either lactate (>2 mmol/l) or arteriovenous oxygen difference (>30 %). Five other clinical outcomes were evaluated. Regression analyses evaluated the relationships between MBL and outcomes.
Results
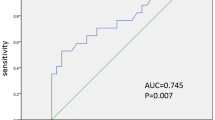
Sixty-three patients were included with a total of 220 MBL. Quantification of MBL was by high-performance liquid chromatography. Overall, 114 (52 %) MBL were outside the therapeutic range: 78 (36 %) subtherapeutic, and 36 (16 %) supratherapeutic. Repeated-measures analysis found a significant association between supratherapeutic MBL and low cardiac output syndrome (p = 0.02), and supratherapeutic MBL were associated with arterial–central venous oxygen saturation difference >30 % at time 3 (p = 0.007).
Conclusions
In this cohort, nontherapeutic MBL were common. Further investigation of milrinone dosing recommendations may improve the postoperative outcomes of children.
Similar content being viewed by others
References
Bishara T, Seto WTW, Trope A, Parshuram Ch (2010) Use of milrinone in critically ill children. Can J Hosp Pharm 63(6):420–428
Hoffman TM, Wernovsky G, Atz AM et al (2003) Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation 107(7):996–1002
Rapundalo ST, Grupp I, Grupp G, Abdul Matlib M, Solaro RJ, Schwartz A (1986) Myocardial actions of milrinone: characterization of its mechanism of action. Circulation 73((3pt-20):III):134–144
Earl CQ, Linden J, Weglicki WB (1986) Inhibition of cyclic AMP-dependent protein kinase activity by the cardiotonic drugs amrinone and milrinone. Life Sci 39(20):1901–1908
Alousi AA, Johnson D (1986) Pharmacology of the bipyridines: amrinone and milrinone. Circulation 73((suppIII)):III10–III24
Sys SU, Goenen MJ, Chalant CH, Brutsaert DL (1986) Inotropic effects of amrinone and milrinone on contraction and relaxation of isolated cardiac muscle. Circulation 73((suppIII)):III25–III35
Honerjäger P (1989) Pharmacology of positive inotropic phosphodiesterase III inhibitors. Eur Heart J 10(Suppl C):25–31
Scholz H, Meyer W (1986) Phosphodiesterase-inhibiting properties of newer inotropic agents. Circulation 73((Supp III)):III99–III108
Endoh M, Yanagisawa T, Taira N, Blinks JR (1986) Effects of new inotropic agents on cyclic nucleotide metabolism and calcium transients in canine ventricular muscle. Circulation 73((3pt2)):III117–III133
Kauffman RF, Schenck KW, Utterback BG, Crowe VG, Cohen ML (1987) In vitro vascular relaxation by new inotropic agents: relationship to phosphodiesterase inhibition and cyclic nucleotides. J Pharmacol Exp Ther 242(3):864–872
Carcillo JA, Fields AI (2002) American College of Critical Care Medicine Task Force Committee M. Clinical practice parameters for hemodynamic support of pediatric and neonatal patients in septic shock. Crit Care Med 30(6):1365–1378
Vogt W, Läer S (2011) Prevention for pediatric low cardiac output syndrome: results from the European survey EuLoCOS-Paed. Pediatric Anesthesia 21:1176–1184
De Luca L, Proietti P, Palombaro GL, Battagliese A, Celotto A, Bucciarelli Ducci C, Fedele F (1986) New positive inotropic agents for congestive heart failure. N Engl J Med 315(6):396–397
Rettig GF, Schieffer HJ (1989) Acute effects of intravenous milrinone in heart failure. Eur Heart J 10(Suppl C):39–43
Young RA, Ward A (1988) Milrinone. A preliminary review of its pharmacological properties and therapeutic use. Drugs 36(2):158–192
Bailey JM, Miller BE, Lu W, Tosone SR, Kanter KR, Tam VK (1999) The pharmacokinetics of milrinone in pediatric patients after cardiac surgery. Anesthesiology 90(4):1012–1018
Bailey JM, Levy JH, Kikura M, Szlam F, Hug CC Jr (1994) Pharmacokinetics of intravenous milrinone in patients undergoing cardiac surgery. Anesthesiology 81(3):616–622
Prielipp RC, MacGregor DA, Butterworth JFt et al (1996) Pharmacodynamics and pharmacokinetics of milrinone administration to increase oxygen delivery in critically ill patients. Chest 109(5):1291–1301
De Hert SG, Moens MM, Jorens PG, Delrue GL, DePaep RJ, Vermeyen KM (1995) Comparison of two different loading doses of milrinone for weaning from cardiopulmonary bypass. J Cardiothorac Vasc Anesth 9(3):264–271
Wernovsky G, Wypij D, Jonas RA (1995) Postoperative course and hemodynamic profile after the arterial switch operation in neonates and infants: a comparison of low-flow cardiopulmonary bypass and circulatory arrest. Circulation 92:2226–2235
Lindsay CA, Barton P, Lawless S et al (1998) Pharmacokinetics and pharmacodynamics of milrinone lactate in pediatric patients with septic shock. J Pediatr 132(2):329–334
Ramamoorthy C, Anderson GD, Williams GD, Lynn AM (1998) Pharmacokinetics and side effects of milrinone in infants and children after open heart surgery. Anesth Analg 86(2):283–289
Bailey JM, Hoffman TM, Wessel DL et al (2004) A population pharmacokinetic analysis of milrinone in pediatric patients after cardiac surgery. J Pharmacokinet Pharmacodyn 31(1):43–59
Edelson J, Koss RF, Baker JF, Park GB (1983) High-performance liquid chromatographic analysis of milrinone in plasma and urine. Intravenous pharmacokinetics in the dog. J Chromatogr 276(2):456–462
Paradisis M, Jiang X, McLachlan AJ et al (2007) Population pharmacokinetics and dosing regimen design of milrinone in preterm infants. Arch Dis Child Fetal Neonatal 92(3):F204–F209
Cheung PY, Chui N, Joffe AR, Rebeyka IM, Robertson CM (2005) Postoperative lactate concentrations predict the outcome of infants aged 6 weeks or less after intracardiac surgery: a cohort follow-up to 18 months. J Thorac Cardiovasc Surg 130(3):837–843
Munoz R, Laussen PC, Palacio G, Zienko L, Piercey G, Wessel DL (2000) Changes in whole blood lactate levels during cardiopulmonary bypass for surgery for congenital cardiac disease: an early indicator of morbidity and mortality. J Thorac Cardiovasc Surg 119(1):155–162
Takami Y, Masumoto H (2005) Mixed venous-arterial CO2 tension gradient after cardiopulmonary bypass. Asian Cardiovasc Thorac Ann 13(3):255–260
Hatherill M, Sajjanhar T, Tibby SM et al (1997) Serum lactate as a predictor of mortality after paediatric cardiac surgery. Arch Dis Child 77(3):235–238
Tibby SM, Murdoch IA (2003) Monitoring cardiac function in intensive care. Arch Dis Child 88(1):46–52
Tibby SM, Murdoch IA (2002) Measurement of cardiac output and tissue perfusion. Curr Opin Pediatr 14(3):303–309
Tibby SM, Hatherill M, Marsh MJ, Murdoch IA (1997) Clinicians’ abilities to estimate cardiac index in ventilated children and infants. Arch Dis Child 77(6):516–518
Romagnoli C, De Carolis MP, Muzii U et al (1992) Effectiveness and side effects of two different doses of caffeine in preventing apnea in premature infants. Ther Drug Monit 14(1):14–19
Olson KR, Benowitz NL, Woo OF, Pond SM (1985) Theophylline overdose: acute single ingestion versus chronic repeated overmedication. Am J Emerg Med 3(5):386–394
Dager WE, Albertson TE (1992) Impact of therapeutic drug monitoring of intravenous theophylline regimens on serum theophylline concentrations in the medical intensive care unit. Ann Pharmacother 26(10):1287–1291
Paloucek FP, Rodvold KA (1988) Evaluation of theophylline overdoses and toxicities. Ann Emerg Med 17(2):135–144
Smith AH, Owen J, Borgman KY, Fish FA, Kannankeril PJ (2011) Relation of milrinone after surgery for congenital heart disease to significant postoperative tachyarrhythmias. Am J Cardiol 108:1620–1624
Baer AB, Holstege CP (2002) Milrinone overdose induced hypotension reversed by vasopressin and norepinephrine infusions. J Toxicol Clin Toxicol 40(5):690
Choong K, Bohn D, Fraser DD, Gaboury I, Hutchison JS, Joffe AR, Litalien C, Menon K, McNamara P, Ward RE (2009) Canadian Critical Care Trials Group. Vasopressin in pediatric vasodilatory shock: a multicenter randomized controlled trial. Am J Respir Crit Care Med 180(7):632–639
Plötz FB, Bouma AB, van Wijk JA, Kneyber MC, Bökenkamp A (2008) Pediatric acute kidney injury in the ICU: an independent evaluation of pRIFLE criteria. Intensive Care Med 34(9):1713–1717
Acknowledgments
Financial support was provided by the Department of Pediatrics (University of Alberta) and Pediatric Critical Care Associates.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Appendix
Rights and permissions
About this article
Cite this article
Garcia Guerra, G., Joffe, A.R., Senthilselvan, A. et al. Incidence of milrinone blood levels outside the therapeutic range and their relevance in children after cardiac surgery for congenital heart disease. Intensive Care Med 39, 951–957 (2013). https://doi.org/10.1007/s00134-013-2858-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-013-2858-3