Abstract
Purpose
Hippocampal-avoidance whole brain radiotherapy (HA-WBRT) for multiple brain metastases may prevent treatment-related cognitive decline, compared to standard WBRT. Additionally, simultaneous integrated boost (SIB) on individual metastases may further improve the outcome. Here, we present initial data concerning local tumour control (LTC), intracranial progression-free survival (PFS), overall survival (OS), toxicity and safety for this new irradiation technique.
Methods and materials
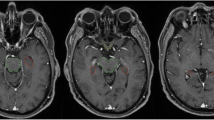
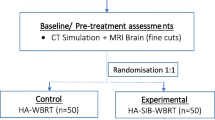
Twenty patients, enrolled between 2011 and 2013, were treated with HA-WBRT (30 Gy in 12 fractions, D98 % to hippocampus ≤ 9 Gy) and a SIB (51 Gy) on multiple (2–13) metastases using a volumetric modulated arc therapy (VMAT) approach based on 2–4 arcs. Metastases were evaluated bidimensionally along the two largest diameters in contrast-enhanced three-dimensional T1-weighed MRI.
Results
Median follow-up was 40 weeks. The median time to progression of boosted metastases has not been reached yet, corresponding to a LTC rate of 73 %. Median intracranial PFS was 40 weeks, corresponding to a 1-year PFS of 45.3 %. Median OS was 71.5 weeks, corresponding to a 1-year OS of 60 %. No obvious acute or late toxicities grade > 2 (NCI CTCAE v4.03) were observed. Dmean to the bilateral hippocampi was 6.585 Gy ± 0.847 (α/β = 2 Gy). Two patients developed a new metastasis in the area of hippocampal avoidance.
Conclusion
HA-WBRT (simultaneous integrated protection, SIP) with SIB to metastases is a safe and tolerable regime that shows favorable LTC for patients with multiple brain metastases, while it has the potential to minimize the side-effect of cognitive deterioration.
Zusammenfassung
Hintergrund
Die Hippocampus-schonende Ganzhirnbestrahlung (HS-GHB) kann im Vergleich zur Standard-GHB die Verschlechterung der neurokognitiven Funktion verhindern. Zusätzlich vermag ein simultan integrierter Boost (SIB) auf die Metastasen die Prognose der betroffenen Patienten weiter zu verbessern. In dieser Studie präsentieren wir erste Ergebnisse hinsichtlich lokaler Tumorkontrolle, des intrakraniellen progressionsfreien Überlebens (PFS), des Gesamtüberlebens (OS), der Toxizität und der Sicherheit dieser neuen Bestrahlungstechnik.
Methoden und Material
Es wurden 20 Patienten zwischen 2011 und 2013 mit einer HS-GHB (30 Gy in 12 Fraktionen, D98 % am Hippocampus ≤ 9 Gy) und simultan integriertem Boost (51 Gy) auf multiple (2 bis 13) Metastasen behandelt. Eingesetzt wurde eine individuell geplante VMAT („volumetric modulated arc therapy“)-Technik mit 2 bis 4 Bögen. Die Größe der Metastasen wurde bidimensional entlang der 2 größten Durchmesser in kontrastmittelverstärkten, T1-gewichteten dreidimensionalen MRT-Aufnahmen evaluiert.
Ergebnisse
Die mediane Nachbeobachtungszeit betrug 40 Wochen. Die mediane Zeit zur Progression der geboosteten Metastasen wurde nicht erreicht, was mit einer lokalen Tumorkontrolle von 73 % einhergeht. Das mediane PFS betrug 40 Wochen (1-Jahres-PFS 45,3 %). Das mediane OS betrug 71,5 Wochen (1-Jahres-OS 60 %). Es traten keine Toxizitäten > Grad 2 (NCI CTCAE v4.03) auf. Die Dmean auf die Hippocampi betrug 6,585 Gy ± 0,847 (α/β = 2 Gy). Zwei Patienten entwickelten je eine neue Metastase im Bereich der Hippocampusschonung.
Schlussfolgerung
HS-GHB (simultane integrierte Protektion, SIP) mit SIB auf die Metastasen ist ein sicheres und tolerables Therapiekonzept mit einer vorteilhaften lokalen Tumorkontrolle bei Patienten mit multiplen Hirnmetastasen bei gleichzeitiger Möglichkeit, die behandlungsbedingten neurokognitiven Einschränkungen von Lernen und Gedächtnis zu verhindern.


Similar content being viewed by others
References
Cairncross JG, Kim JH, Posner JB (1980) Radiation therapy for brain metastases. Ann Neurol 7:529–541
Weissman DE (1988) Glucocorticoid treatment for brain metastases and epidural spinal cord compression: a review. J Clin Oncol 6:543–551
Diener-West M, Dobbins TW, Phillips TL, Nelson DF (1989) Identification of an optimal subgroup for treatment evaluation of patients with brain metastases using RTOG study 7916. Int J Radiat Oncol Biol Phys 16:669–673
Kondziolka D, Patel A, Lunsford LD et al (1999) Stereotactic radiosurgery plus whole brain radiotherapy versus radiotherapy alone for patients with multiple brain metastases. Int J Radiat Oncol Biol Phys 45:427–434
Kocher M, Soffietti R, Abacioglu U et al (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952–26001 study. J Clin Oncol 29:134–141
Li J, Bentzen SM, Li J, Renschler M, Mehta MP (2008) Relationship between neurocognitive function and quality of life after whole-brain radiotherapy in patients with brain metastasis. Int J Radiat Oncol Biol Phys 71:64–70
Soffietti R, Kocher M, Abacioglu UM et al (2013) A European Organisation for Research and Treatment of Cancer phase III trial of adjuvant whole-brain radiotherapy versus observation in patients with one to three brain metastases from solid tumors after surgical resection or radiosurgery: quality-of-life results. J Clin Oncol 31:65–72
Roman DD, Sperduto PW (1995) Neuropsychological effects of cranial radiation: current knowledge and future directions. Int J Radiat Oncol Biol Phys 31:983–998
Welzel G, Fleckenstein K, Schaefer J et al (2008) Memory function before and after whole brain radiotherapy in patients with and without brain metastases. Int J Radiat Oncol Biol Phys 72:1311–1318
Chang EL, Wefel JS, Hess KR et al (2009) Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol 10:1037–1044
Taphoorn MJ, Klein M (2004) Cognitive deficits in adult patients with brain tumours. Lancet Neurol 3:159–168
Milner B, Squire LR, Kandel ER (1998) Cognitive neuroscience and the study of memory. Neuron 20:445–468
Dickerson BC, Eichenbaum H (2010) The episodic memory system: neurocircuitry and disorders. Neuropsychopharmacology 35:86–104
Scoville WB, Milner B (1957) Loss of recent memory after bilateral hippocampal lesions. J Neurol Neurosurg Psychiatry 20:11–21
Shors TJ, Miesegaes G, Beylin A et al (2001) Neurogenesis in the adult is involved in the formation of trace memories. Nature 410:372–376
Deng W, Aimone JB, Gage FH (2010) New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat Rev Neurosci 11:339–350
Small SA, Schobel SA, Buxton RB et al (2011) A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nat Rev Neurosci 12:585–601
Gondi V, Tome WA, Marsh J et al (2010) Estimated risk of perihippocampal disease progression after hippocampal avoidance during whole-brain radiotherapy: safety profile for RTOG 0933. Radiother Oncol 95:327–331
Oskan F, Ganswindt U, Schwarz SB et al (2014) Hippocampus sparing in whole-brain radiotherapy. A review. Strahlenther Onkol 190:337–341
Assouline A, Levy A, Chargari C et al (2011) Whole brain radiotherapy: prognostic factors and results of a radiation boost delivered through a conventional linear accelerator. Radiother Oncol 99:214–217
Casanova N, Mazouni Z, Bieri S et al (2010) Whole brain radiotherapy with a conformational external beam radiation boost for lung cancer patients with 1–3 brain metastasis: a multi institutional study. Radiat Oncol 5:13
Prokic V, Wiedenmann N, Fels F et al (2013) Whole brain irradiation with hippocampal sparing and dose escalation on multiple brain metastases: a planning study on treatment concepts. Int J Radiat Oncol Biol Phys 85:264-270
Broemme J, Abu-Isa J, Kottke R et al (2013) Adjuvant therapy after resection of brain metastases. Frameless image-guided LINAC-based radiosurgery and stereotactic hypofractionated radiotherapy. Strahlenther Onkol 189:765–770
Brant-Zawadzki M, Gillan GD, Nitz WR (1992) MP RAGE: a three-dimensional, T1-weighted, gradient-echo sequence–initial experience in the brain. Radiology 182:769–775
Lin NU, Lee EQ, Aoyama H et al (2013) Challenges relating to solid tumour brain metastases in clinical trials, part 1: patient population, response, and progression. A report from the RANO group. Lancet Oncol 14:e396–e406
Monje ML, Mizumatsu S, Fike JR, Palmer TD (2002) Irradiation induces neural precursor-cell dysfunction. Nat Med 8:955–962
Dietrich J, Monje M, Wefel J, Meyers C (2008) Clinical patterns and biological correlates of cognitive dysfunction associated with cancer therapy. Oncologist 13:1285–1295
Gondi V, Pugh SL, Tome WA et al (2014) Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J Clin Oncol 32:3810–3816
Harth S, Abo-Madyan Y, Zheng L et al (2013) Estimation of intracranial failure risk following hippocampal-sparing whole brain radiotherapy. Radiother Oncol 109:152–158
Gondi V, Hermann BP, Mehta MP, Tomé WA (2013) Hippocampal dosimetry predicts neurocognitive function impairment after fractionated stereotactic radiotherapy for benign or low-grade adult brain tumors. Int J Radiat Oncol Biol Phys 85:348–354
Rades D, Pluemer A, Veninga T et al (2007) A boost in addition to whole-brain radiotherapy improves patient outcome after resection of 1 or 2 brain metastases in recursive partitioning analysis class 1 and 2 patients. Cancer 110:1551–1559
Andrews DW, Scott CB, Sperduto PW et al (2004) Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet 363:1665–1672
Yamamoto M, Serizawa T, Shuto T et al (2014) Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901): a multi-institutional prospective observational study. Lancet Oncol 15:387–395
Wiggenraad R, Verbeek-de Kanter A, Mast M et al (2012) Local progression and pseudo progression after single fraction or fractionated stereotactic radiotherapy for large brain metastases. A single centre study. Strahlenther Onkol 188:696–701
Lawrence YR, Li XA, el Naqa I et al (2010) Radiation dose–volume effects in the brain. Int J Radiat Oncol Biol Phys 76:S20–S27
Awad R, Fogarty G, Hong A et al (2013) Hippocampal avoidance with volumetric modulated arc therapy in melanoma brain metastases—the first Australian experience. Radiat Oncol 8:62
Rades D, Gerdan L, Segedin B et al (2013) Brain metastasis. Prognostic value of the number of involved extracranial organs. Strahlenther Onkol 189:996–1000
Gaspar L, Scott C, Rotman M et al (1997) Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys 37:745–751
Acknowledgments
The present study was supported by the German Cancer Consortium (DKTK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
O. Oehlke, D. Wucherpfennig, F. Fels, L. Frings, K. Egger, A. Weyerbrock, V. Prokic, C. Nieder and A.-L. Grosu state that there are no conflicts of interest.
All studies on humans described in the present manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current, revised form). Informed consent was obtained from all patients included in studies.
Rights and permissions
About this article
Cite this article
Oehlke, O., Wucherpfennig, D., Fels, F. et al. Whole brain irradiation with hippocampal sparing and dose escalation on multiple brain metastases. Strahlenther Onkol 191, 461–469 (2015). https://doi.org/10.1007/s00066-014-0808-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-014-0808-9
Keywords
- Neoplasm metastases
- Whole brain irradiation
- Hippocampal sparing
- Simultaneous integrated protection
- Neurocognition