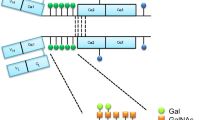
Abstract
Glycosylation abnormalities have been observed in autoimmune diseases and cancer. Here, we compare mechanisms of aberrant O-glycosylation, i.e., formation of Tn and sialyl-Tn structures, on MUC1 in breast cancer, and on IgA1 in an autoimmune disease, IgA nephropathy. The pathways of aberrant O-glycosylation, although different for MUC1 and IgA1, include dysregulation in glycosyltransferase expression, stability, and/or intracellular localization. Moreover, these aberrant glycoproteins are recognized by antibodies, although with different consequences. In breast cancer, elevated levels of antibodies recognizing aberrant MUC1 are associated with better outcome, whereas in IgA nephropathy, the antibodies recognizing aberrant IgA1 are part of the pathogenetic process.


Similar content being viewed by others
References
Brockhausen I (2006) Mucin-type O-glycans in human colon and breast cancer: glycodynamics and functions. EMBO Rep 7:599–604
Chui D et al (2001) Genetic remodeling of protein glycosylation in vivo induces autoimmune disease. Proc Natl Acad Sci USA 98:1142–1147
Kobata A (1998) A retrospective and prospective view of glycopathology. Glycoconj J 15:323–331
Rudd PM, Elliott T, Cresswell P, Wilson IA, Dwek RA (2001) Glycosylation and the immune system. Science 291:2370–2376
Ju T, Cummings RD (2005) Protein glycosylation: chaperone mutation in Tn syndrome. Nature 437:1252
Tabak LA (2010) The role of mucin-type O-glycans in eukaryotic development. Semin Cell Dev Biol 6:616–621
Tomana M et al (1999) Circulating immune complexes in IgA nephropathy consist of IgA1 with galactose-deficient hinge region and antiglycan antibodies. J Clin Invest 104:73–81
Suzuki H et al (2008) IgA1-secreting cell lines from patients with IgA nephropathy produce aberrantly glycosylated IgA1. J Clin Invest 118:629–639
Suzuki H et al (2011) The pathophysiology of IgA nephropathy. J Am Soc Nephrol 22:1795–1803
Suzuki H et al (2009) Aberrantly glycosylated IgA1 in IgA nephropathy patients is recognized by IgG antibodies with restricted heterogeneity. J Clin Invest 119:1668–1677
Singh R, Bandyopadhyay D (2007) MUC1: a target molecule for cancer therapy. Cancer Biol Ther 6:481–486
Lloyd KO, Burchell J, Kudryashov V, Yin BW, Taylor-Papadimitriou J (1996) Comparison of O-linked carbohydrate chains in MUC-1 mucin from normal breast epithelial cell lines and breast carcinoma cell lines. Demonstration of simpler and fewer glycan chains in tumor cells. J Biol Chem 271:33325–33334
Julien S et al (2005) Stable expression of sialyl-Tn antigen in T47-D cells induces a decrease of cell adhesion and an increase of cell migration. Breast Cancer Res Treat 90:77–84
Pinho S et al (2007) Biological significance of cancer-associated sialyl-Tn antigen: modulation of malignant phenotype in gastric carcinoma cells. Cancer Lett 249:157–170
Wandall HH et al (2010) Cancer biomarkers defined by autoantibody signatures to aberrant O-glycopeptide epitopes. Cancer Res 70:1306–1313
Finn OJ (2008) Cancer immunology. N Engl J Med 358:2704–2715
von Mensdorff-Pouilly S et al (1996) Humoral immune response to polymorphic epithelial mucin (MUC-1) in patients with benign and malignant breast tumours. Eur J Cancer 32A:1325–1331
Blixt O et al (2011) Autoantibodies to aberrantly glycosylated MUC1 in early stage breast cancer are associated with a better prognosis. Breast Cancer Res 13:R25
Takahashi K et al (2012) Naturally occurring structural isomers in serum IgA1 O-glycosylation. J Proteome Res 11:692–702
Takahashi K et al (2010) Clustered O-glycans of IgA1: defining macro- and microheterogeneity by use of electron capture/transfer dissociation. Mol Cell Proteomics 9:2545–2557
Novak J, Mestecky J (2009) IgA Immune-complex. In: Lai KN (ed) Recent advances in IgA nephropathy, Imperial College Press and the World Scientific Publisher, Hong Kong, p 177–191
Mattu TS et al (1998) The glycosylation and structure of human serum IgA1, Fab, and Fc regions and the role of N-glycosylation on Fcα receptor interactions. J Biol Chem 273:2260–2272
Sihlbom C et al (2009) Localization of O-glycans in MUC1 glycoproteins using electron-capture dissociation fragmentation mass spectrometry. Glycobiology 19:375–381
Muller S, Hanisch FG (2002) Recombinant MUC1 probe authentically reflects cell-specific O-glycosylation profiles of endogenous breast cancer mucin. High density and prevalent core 2-based glycosylation. J Biol Chem 277:26103–26112
Tarp MA, Clausen H (2008) Mucin-type O-glycosylation and its potential use in drug and vaccine development. Biochim Biophys Acta 1780:546–563
Backstrom M et al (2003) Recombinant MUC1 mucin with a breast cancer-like O-glycosylation produced in large amounts in Chinese-hamster ovary cells. Biochem J 376:677–686
Mall AS (2008) Analysis of mucins: role in laboratory diagnosis. J Clin Pathol 61:1018–1024
Storr SJ et al (2008) The O-linked glycosylation of secretory/shed MUC1 from an advanced breast cancer patient’s serum. Glycobiology 18:456–462
Napoletano C et al (2007) Tumor-associated Tn-MUC1 glycoform is internalized through the macrophage galactose-type C-type lectin and delivered to the HLA class I and II compartments in dendritic cells. Cancer Res 67:8358–8367
Wahrenbrock MG, Varki A (2006) Multiple hepatic receptors cooperate to eliminate secretory mucins aberrantly entering the bloodstream: are circulating cancer mucins the “tip of the iceberg”? Cancer Res 66:2433–2441
Bennett EP et al. (2012) Control of mucin-type O-glycosylation—a classification of the polypeptide GalNAc-transferase gene family. Glycobiology 22:736–756
Gerken TA et al (2011) Emerging paradigms for the initiation of mucin-type protein O-glycosylation by the polypeptide GalNAc transferase family of glycosyltransferases. J Biol Chem 286:14493–14507
Rottger S et al (1998) Localization of three human polypeptide GalNAc-transferases in HeLa cells suggests initiation of O-linked glycosylation throughout the Golgi apparatus. J Cell Sci 111(Pt 1):45–60
Gill DJ, Chia J, Senewiratne J, Bard F (2010) Regulation of O-glycosylation through Golgi-to-ER relocation of initiation enzymes. J Cell Biol 189:843–858
Iwasaki H et al (2003) Initiation of O-glycan synthesis in IgA1 hinge region is determined by a single enzyme, UDP-N-acetyl-α-d-galactosamine:polypeptide N-acetylgalactosaminyltransferase 2. J Biol Chem 278:5613–5621
Wandall HH et al (2007) The lectin domains of polypeptide GalNAc-transferases exhibit carbohydrate-binding specificity for GalNAc: lectin binding to GalNAc-glycopeptide substrates is required for high density GalNAc-O-glycosylation. Glycobiology 17:374–387
Raska M et al (2011) Role of GalNAc-transferases in the synthesis of aberrant IgA1 O-glycans in IgA nephropathy. J Am Soc Nephrol 22:625A
Bennett EP et al (1999) Cloning and characterization of a close homologue of human UDP-N-acetyl-alpha-d-galactosamine:polypeptide N-acetylgalactosaminyltransferase-T3, designated GalNAc-T6. Evidence for genetic but not functional redundancy. J Biol Chem 274:25362–25370
Wandall HH et al (1997) Substrate specificities of three members of the human UDP-N-acetyl-α-d-galactosamine:polypeptide N-acetylgalactosaminyltransferase family, GalNAc-T1, -T2, and -T3. J Biol Chem 272:23503–23514
Schwientek T et al (2002) Functional conservation of subfamilies of putative UDP-N-acetylgalactosamine:polypeptide N-acetylgalactosaminyltransferases in Drosophila, Caenorhabditis elegans, and mammals. One subfamily composed of l(2)35Aa is essential in Drosophila. J Biol Chem 277:22623–22638
Cheng L et al (2004) Characterization of a novel human UDP-GalNAc transferase, pp-GalNAc-T15. FEBS Lett 566:17–24
Zhang Y et al (2003) Cloning and characterization of a new human UDP-N-acetyl-α-d-galactosamine:polypeptide N-acetylgalactosaminyltransferase, designated pp-GalNAc-T13, that is specifically expressed in neurons and synthesizes GalNAc α-serine/threonine antigen. J Biol Chem 278:573–584
Wang H et al (2003) Cloning and characterization of a novel UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferase, pp-GalNAc-T14. Biochem Biophys Res Commun 300:738–744
Hassan H et al (2000) The lectin domain of UDP-N-acetyl-d-galactosamine: polypeptide N-acetylgalactosaminyltransferase-T4 directs its glycopeptide specificities. J Biol Chem 275:38197–38205
Brooks SA, Carter TM, Bennett EP, Clausen H, Mandel U (2007) Immunolocalisation of members of the polypeptide N-acetylgalactosaminyl transferase (ppGalNAc-T) family is consistent with biologically relevant altered cell surface glycosylation in breast cancer. Acta Histochem 109:273–284
Mandel U et al (1999) Expression of polypeptide GalNAc-transferases in stratified epithelia and squamous cell carcinomas: immunohistological evaluation using monoclonal antibodies to three members of the GalNAc-transferase family. Glycobiology 9:43–52
Marcos NT et al (2003) Polypeptide GalNAc-transferases, ST6GalNAc-transferase I, and ST3Gal-transferase I expression in gastric carcinoma cell lines. J Histochem Cytochem 51:761–771
Cooper LS et al (1999) Expression of GalNAc transferases in breast tissues and cell lines. J Pathol 187:26A–26A
Berois N et al (2006) UDP-N-acetyl-d-galactosamine: polypeptide N-acetylgalactosaminyltransferase-6 as a new immunohistochemical breast cancer marker. J Histochem Cytochem 54:317–328
Freire T et al (2006) UDP-N-acetyl-d-galactosamine:polypeptide N-acetylgalactosaminyltransferase 6 (ppGalNAc-T6) mRNA as a potential new marker for detection of bone marrow-disseminated breast cancer cells. Int J Cancer 119:1383–1388
Park JH et al (2010) Critical roles of mucin 1 glycosylation by transactivated polypeptide N-acetylgalactosaminyltransferase 6 in mammary carcinogenesis. Cancer Res 70:2759–2769
Wu C et al (2010) N-Acetylgalactosaminyltransferase-14 as a potential biomarker for breast cancer by immunohistochemistry. BMC Cancer 10:123
Wagner KW et al (2007) Death-receptor O-glycosylation controls tumor-cell sensitivity to the proapoptotic ligand Apo2L/TRAIL. Nat Med 13:1070–1077
Gray-McGuire C et al (2010) Confirmation of linkage to and localization of familial colon cancer risk haplotype on chromosome 9q22. Cancer Res 70:5409–5418
Guda K et al (2009) Inactivating germ-line and somatic mutations in polypeptide N-acetylgalactosaminyltransferase 12 in human colon cancers. Proc Natl Acad Sci USA 106:12921–12925
Wada Y et al (2010) Comparison of methods for profiling O-glycosylation: Human Proteome Organisation Human Disease Glycomics/Proteome Initiative multi-institutional study of IgA1. Mol Cell Proteomics 9:719–727
Ju T, Brewer K, D’Souza A, Cummings RD, Canfield WM (2002) Cloning and expression of human core 1 β1,3-galactosyltransferase. J Biol Chem 277:178–186
Wang Y et al (2010) Cosmc is an essential chaperone for correct protein O-glycosylation. Proc Natl Acad Sci USA 107:9228–9233
Ju T, Cummings RD (2002) A unique molecular chaperone Cosmc required for activity of the mammalian core 1 β 3-galactosyltransferase. Proc Natl Acad Sci USA 99:16613–16618
Charlier E et al (2010) SHIP-1 inhibits CD95/APO-1/Fas-induced apoptosis in primary T lymphocytes and T leukemic cells by promoting CD95 glycosylation independently of its phosphatase activity. Leukemia 24:821–832
Dall’Olio F, Chiricolo M (2001) Sialyltransferases in cancer. Glycoconj J 18:841–850
Harduin-Lepers A et al (2001) The human sialyltransferase family. Biochimie 83:727–737
Gharavi AG et al (2008) Aberrant IgA1 glycosylation is inherited in familial and sporadic IgA nephropathy. J Am Soc Nephrol 19:1008–1014
Raska M et al (2007) Identification and characterization of CMP-NeuAc:GalNAc-IgA1 α2,6-sialyltransferase in IgA1-producing cells. J Mol Biol 369:69–78
Marcos NT et al (2004) Role of the human ST6GalNAc-I and ST6GalNAc-II in the synthesis of the cancer-associated sialyl-Tn antigen. Cancer Res 64:7050–7057
Suzuki H et al (2009) Mechanisms of aberrant glycosylation of IgA1 in patients with IgA nephropathy. J Am Soc Nephrol 20:301A
Dalziel M et al (2001) The relative activities of the C2GnT1 and ST3Gal-I glycosyltransferases determine O-glycan structure and expression of a tumor-associated epitope on MUC1. J Biol Chem 276:11007–11015
Brockhausen I, Yang JM, Burchell J, Whitehouse C, Taylor-Papadimitriou J (1995) Mechanisms underlying aberrant glycosylation of MUC1 mucin in breast cancer cells. Eur J Biochem 233:607–617
Hanisch FG, Stadie TR, Deutzmann F, Peter-Katalinic J (1996) MUC1 glycoforms in breast cancer—cell line T47D as a model for carcinoma-associated alterations of O-glycosylation. Eur J Biochem 236:318–327
Sewell R et al (2006) The ST6GalNAc-I sialyltransferase localizes throughout the Golgi and is responsible for the synthesis of the tumor-associated sialyl-Tn O-glycan in human breast cancer. J Biol Chem 281:3586–3594
Ju T et al (2008) Human tumor antigens Tn and sialyl Tn arise from mutations in Cosmc. Cancer Res 68:1636–1646
Litvinov SV, Hilkens J (1993) The epithelial sialomucin, episialin, is sialylated during recycling. J Biol Chem 268:21364–21371
Avrameas S, Ternynck T (1998) Natural antibodies. In: Delves PJ, Roitt IM (eds) Encyclopedia of immunology. Academic Press, San Diego, p 1806–1809
Brandlein S et al (2003) Cysteine-rich fibroblast growth factor receptor 1, a new marker for precancerous epithelial lesions defined by the human monoclonal antibody PAM-1. Cancer Res 63:2052–2061
Coutinho A, Kazatchkine MD, Avrameas S (1995) Natural autoantibodies. Curr Opin Immunol 7:812–818
Avrameas S, Ternynck T, Tsonis IA, Lymberi P (2007) Naturally occurring B-cell autoreactivity: a critical overview. J Autoimmun 29:213–218
Vollmers HP, Brandlein S (2007) Natural antibodies and cancer. J Autoimmun 29:295–302
Brandlein S et al (2003) Natural IgM antibodies and immunosurveillance mechanisms against epithelial cancer cells in humans. Cancer Res 63:7995–8005
Mouthon L et al (1995) Analysis of the normal human IgG antibody repertoire. Evidence that IgG autoantibodies of healthy adults recognize a limited and conserved set of protein antigens in homologous tissues. J Immunol 154:5769–5778
Springer GF (1997) Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy. J Mol Med 75:594–602
Bray J, MacLean GD, Dusel FJ, McPherson TA (1982) Decreased levels of circulating lytic anti-T in the serum of patients with metastatic gastrointestinal cancer: a correlation with disease burden. Clin Exp Immunol 47:176–182
Macher BA, Galili U (2008) The Galα1,3Galβ1,4GlcNAc-R (α-Gal) epitope: a carbohydrate of unique evolution and clinical relevance. Biochim Biophys Acta 1780:75–88
Galili U, Mandrell RE, Hamadeh RM, Shohet SB, Griffiss JM (1988) Interaction between human natural anti-α-galactosyl immunoglobulin G and bacteria of the human flora. Infect Immun 56:1730–1737
Baumgarth N, Tung JW, Herzenberg LA (2005) Inherent specificities in natural antibodies: a key to immune defense against pathogen invasion. Springer Semin Immunopathol 26:347–362
Gharavi AG et al (2011) Genome-wide association study identifies susceptibility loci for IgA nephropathy. Nat Genet 43:321–327
O’Boyle KP et al (1992) Immunization of colorectal cancer patients with modified ovine submaxillary gland mucin and adjuvants induces IgM and IgG antibodies to sialylated Tn. Cancer Res 52:5663–5667
MacLean GD et al (1993) Immunization of breast cancer patients using a synthetic sialyl-Tn glycoconjugate plus Detox adjuvant. Cancer Immunol Immunother 36:215–222
Longenecker BM, Reddish M, Koganty R, MacLean GD (1993) Immune responses of mice and human breast cancer patients following immunization with synthetic sialyl-Tn conjugated to KLH plus detox adjuvant. Ann N Y Acad Sci 690:276–291
Longenecker BM, Reddish M, Koganty R, MacLean GD (1994) Specificity of the IgG response in mice and human breast cancer patients following immunization against synthetic sialyl-Tn, an epitope with possible functional significance in metastasis. Adv Exp Med Biol 353:105–124
MacLean GD, Miles DW, Rubens RD, Reddish MA, Longenecker BM (1996) Enhancing the effect of THERATOPE STn-KLH cancer vaccine in patients with metastatic breast cancer by pretreatment with low-dose intravenous cyclophosphamide. J Immunother Emphasis Tumor Immunol 19:309–316
Guo Z, Wang Q (2009) Recent development in carbohydrate-based cancer vaccines. Curr Opin Chem Biol 13:608–617
Gilewski TA et al (2007) Immunization of high-risk breast cancer patients with clustered STn-KLH conjugate plus the immunologic adjuvant QS-21. Clin Cancer Res 13:2977–2985
Slovin SF et al (2003) Fully synthetic carbohydrate-based vaccines in biochemically relapsed prostate cancer: clinical trial results with α-N-acetylgalactosamine-O-serine/threonine conjugate vaccine. J Clin Oncol 21:4292–4298
Slovin SF et al (2005) A bivalent conjugate vaccine in the treatment of biochemically relapsed prostate cancer: a study of glycosylated MUC-2-KLH and Globo H-KLH conjugate vaccines given with the new semi-synthetic saponin immunological adjuvant GPI-0100 OR QS-21. Vaccine 23:3114–3122
Elkon K, Casali P (2008) Nature and functions of autoantibodies. Nat Clin Pract Rheumatol 4:491–498
Ni J, Song H, Wang Y, Stamatos NM, Wang LX (2006) Toward a carbohydrate-based HIV-1 vaccine: synthesis and immunological studies of oligomannose-containing glycoconjugates. Bioconjug Chem 17:493–500
Dziadek S, Hobel A, Schmitt E, Kunz H (2005) A fully synthetic vaccine consisting of a tumor-associated glycopeptide antigen and a T-cell epitope for the induction of a highly specific humoral immune response. Angew Chem Int Ed Engl 44:7630–7635
Cremer GA et al (2006) Synthesis and biological evaluation of a multiantigenic Tn/TF-containing glycopeptide mimic of the tumor-related MUC1 glycoprotein. ChemMedChem 1:965–968
Lo-Man R et al (2004) A fully synthetic therapeutic vaccine candidate targeting carcinoma-associated Tn carbohydrate antigen induces tumor-specific antibodies in nonhuman primates. Cancer Res 64:4987–4994
Ingale S, Wolfert MA, Gaekwad J, Buskas T, Boons GJ (2007) Robust immune responses elicited by a fully synthetic three-component vaccine. Nat Chem Biol 3:663–667
Acknowledgments
This work was supported in part by grants from the National Institutes of Health DK082753, DK078244, DK083663, DK075868, DK077279, and GM098539, a grant from the IGA Nephropathy Foundation of America, and by grants and LH11046 Ministry of School, Youth, and Sport, GAP302/10/1055 Czech Science Foundation, NT11081 Grant Agency of the Ministry of the Health, Czech Republic.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Stuchlová Horynová, M., Raška, M., Clausen, H. et al. Aberrant O-glycosylation and anti-glycan antibodies in an autoimmune disease IgA nephropathy and breast adenocarcinoma. Cell. Mol. Life Sci. 70, 829–839 (2013). https://doi.org/10.1007/s00018-012-1082-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-012-1082-6