Summary
-
1.
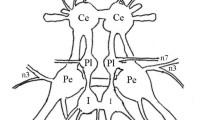
The pleural D gp. neurones ofLymnaea can be identified readily on the bases of size, colour, position and their characteristic background activity of axon spikes that fail to propagate actively into the soma.
-
2.
Injection of Lucifer Yellow shows that pleural D gp. neurones have three radial axons arising from, or close to, a centrally located soma. Action potential collision experiments show that each different amplitude of blocked axon spike originates from a different radial axon.
-
3.
Because axon spikes do not necessarily cause somatic action potentials, each of the radial axons can function independently. Synaptic input to excite the soma acts as a gate causing the radial axons to act in concert.
Similar content being viewed by others
References
Benjamin PR, Winlow W (1981) The distribution of three wide- acting synaptic inputs to identified neurons in the isolated brain ofLymnaea stagnalis (L.). Comp Biochem Physiol [A] 70:293–307
Calabrese RL (1980) Control of multiple impulse-initiation sites in a leech interneurone. J Neurophysiol 44:878–896
Calabrese RL, Kennedy D (1974) Multiple sites of spike initiation in a single dendritic system. Brain Res 82:316–321
Essen DC Van (1973) The contribution of membrane hyperpolarization to adaptation and conduction block in sensory neurones of the leech. J Physiol (Lond) 230:509–534
Friesen WO (1975) Physiological anatomy and burst pattern in the cardiac ganglion of the spiny lobster,Panulirus interruptus. J Comp Physiol 101:173–189
Getting PA, Willows AOD (1974) Modification of neuron properties by electrotonic synapses. II. Burst formation by electrotonic synapses. J Neurophysiol 37:858–868
Grossman Y, Parnas I, Spira ME (1979) Ionic mechanisms involved in differential conduction of action potentials at high frequency in a branching axon. J Physiol (Lond) 295:307–322
Haydon PG, Winlow W (1981) Morphology of the giant dopamine- containing neurone, R.Pe.D.1, inLymnaea stagnalis (L.) revealed by Lucifer Yellow CH. J Exp Biol 94:149–157
Heitler WJ, Goodman CS (1978) Multiple sites of spike initiation in a bifurcating locust neurone. J Exp Biol 76:63–84
Holden AV, Yoda M (1981) The effects of ionic channel density on neuronal function. J Theor Neurobiol 1:60–81
Korn H, Bennett MVL (1975) Vestibular nystagmus and teleost oculomotor neurons: Functions of electrotonic coupling and dendritic impulse initiation. J Neurophysiol 38:430–451
Llinás R, Nicholson C (1971) Electrophysiological properties of dendrites and somata in alligator Purkinje cells. J Neurophysiol 34:532–551
Moulins M, Nagy F (1981) Participation of an unpaired motor neurone in the bilaterally organized oesophageal rhythm in the lobstersJasus lalandii andPalinurus vulgaris. J Exp Biol 90:205–230
Nagy F, Dickinson PS, Moulins M (1981) Rhythmical synaptic control of axonal conduction in a lobster motor neuron. J Neurophysiol 45:1109–1124
Slade CT, Mills J, Winlow W (1981) The neuronal organization of the paired pedal ganglia ofLymnaea stagnalis (L.). Comp Biochem Physiol [A] 69:789–803
Spencer AN, Satterlie RA (1980) Electrical and dye coupling in an identified group of neurons in a coelenterate. J Neurobiol 11:13–19
Spencer WA, Kandel ER (1961) Electrophysiology of hippocampal neurons. IV. Fast prepotentials. J Neurophysiol 24:272–285
Spira ME, Yarom Y, Parnas I (1976) Modulation of spike frequency by regions of special axonal geometry and by synaptic inputs. J Neurophysiol 39:882–899
Stewart WW (1978) Functional connections between cells as revealed by dye coupling with a highly fluorescent naphthalamide tracer. Cell 14:741–759
Swadlow HA, Kocsis JD, Waxman SG (1980) Modulation of impulse conduction along the axonal tree. Annu Rev Biophys Bioeng 9:143–179
Tauc L, Hughes GM (1963) Modes of initiation and propagation of spikes in the branching axons of molluscan central neurons. J Gen Physiol 46:533–549
Vedel JP, Moulins M (1978) A motor neuron involved in two centrally generated motor patterns by means of two different spike initiating sites. Brain Res 138:347–352
Winlow W, Benjamin PR (1976) Neuronal mapping of the brain of the pond-snailLymnaea stagnalis (L.). In: Salanki J (ed) Neurobiology of invertebrates, gastropoda brain. Academiai Kiado, Budapest, pp 41–59
Winlow W, Kandel ER (1976) The morphology of identified neurones in the abdominal ganglia ofAplysia californica. Brain Res 112:221–249
Winlow W, Haydon PG, Benjamin PR (1981) Multiple postsynaptic actions of the giant dopamine-containing neurone R.Pe.D.1 ofLymnaea stagnalis. J Exp Biol 94:137–148
Wood JD, Mayer CJ (1979) Intracellular study of tonic-type enteric neurons in guinea-pig small intestine. J Neurophysiol 42:569–581
Wong RKS, Pearson KG (1975) Limitations on impulse conduction in the terminal branches of insect sensory nerve fibers. Brain Res 100:431–436
Wong RKS, Prince DA, Basbaum AI (1979) Intradendritic recordings from hippocampal neurons. Proc Natl Acad Sci USA 76:986–990
Yau KW (1976) Receptive fields, geometry and conduction block of sensory neurones in the central nervous system of the leech. J Physiol (Lond) 263:513–538
Zucker RS (1972) Crayfish escape behaviour and central synapses. III. Electrical junctions and dendrite spikes in fast flexor motoneurons. J Neurophysiol 35:638–651
Author information
Authors and Affiliations
Additional information
Abbreviation: D gp. see Introduction (Para 3)
Emma and Leslie Reid Scholar
Rights and permissions
About this article
Cite this article
Haydon, P.G., Winlow, W. Multipolar neurones ofLymnaea stagnalis . J. Comp. Physiol. 147, 503–510 (1982). https://doi.org/10.1007/BF00612016
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00612016