Summary
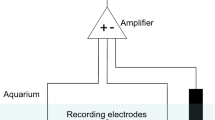
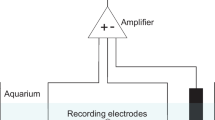
The mantis shrimpSquilla mantis responds to an electrical shock with a quick and violent flexure of its body (Fig. 1 and 2). The reaction time of this defensive response was measured for each experimental shrimp and the minimal current that elicited a reaction time equal to or lesser than 0.22 s was considered as its intensity threshold (Fig. 3). Different doses of morphine-HCl were injected and results showed that this drug produces a dose-related analgesia by increasing the intensity threshold. A concentration of morphine equal to 30.0 μg/mm of animal length (about 91.0 μg/g) causes a 50% inhibition in the sensitivity to the electrical stimulus, but the effect is fully blocked by naloxone (Table 2).
The present study represents the first behavioral analysis of opiate effects in invertebrates. However, the effective dose reported in this experiment is far greater than that used with vertebrates. Some speculative arguments are mentioned in order to account for a such large difference.
Similar content being viewed by others
References
Alumets J, Hakanson R, Sundler F, Thorell J (1979) Neuronal localisation of immunoreactive enkephalin and B-endorphin in the earthworm. Nature 279:805–806
Ankier SI (1974) New hot plate test to quantify antinociceptive and narcotic antagonist activities. Eur J Pharmacol 27:1–4
Babbini M, Gaiaroli M, Bartolett M (1979) Stimulus-response relationships in a quickly learned escape from shock: effects of morphine. Pharmacol Biochem Behav 11:155–158
Cox BM, Padhya R (1977) Opiate binding and effect in ileum preparations from normal and morphine pretreated guinea-pigs. Br J Pharmacol 61:271–278
D'Amour FE, Smith DL (1941) A method for determining loss of pain sensation. J Pharmacol Exp Ther 72:74–79
Davis M (1979) Morphine and naloxone: effects on conditioned fear as measured with potentiated startle paradigm. Eur J Pharmacol 54:341–347
Henderson G, Hughes J, Kosterlitz HW (1972) A new example of a morphine-sensitive neuro-effector junction: adrenergic transmission in the mouse vas deferens. Br J Pharmacol 46:764–766
Hughes J, Kosterlitz HW, Leslies FM (1975) Effect of morphine on adrenergic transmission in the mouse vas deferens. Assessment of agonist and antagonist potencies of narcotic analgesics. Br J Pharmacol 53:371–381
Kobayashi RM, Palkovits M, Miller RJ, Chang K, Cuatrecasas P (1978) Brain enkephalin unaltered by hypophysectomy. Life Sci 22:527–530
Kream RM, Zukin RS, Stefano GB (1980) Demonstration of two classes of opiate binding sites in the nervous tissue of the marine molluscMytilus edulis. J Biol Chem 255:9218–9224
Liebman FM, Klinger G (1980) In vivo hamster skeletal muscle preparation: morphine sensitive — naloxone insensitive. Eur J Pharmacol 62:167–175
Lineberry CG, Kulics AT (1980) Morphine effects on escape in the rhesus monkey. Neuropharmacology 19:107–110
Markowitz R, Jacobson J, Bain G, Kornetsky G (1976) Naloxone blockade of morphine analgesia: a dose-effect study of duration and magnitude. J Pharmacol Exp Ther 199:385–388
Miller RJ, Cuatrecasas P (1978) Enkephalins and endorphins. Vitam Horm 36:297–382
Mucha RF (1980) Effect of naloxone and morphine on guinea pig tonic immobility. Behav Neurol Biol 28:111–115
Pearse AGE (1980) The phylogeny of the common peptides. Proc R Soc Lond [Biol] 210:61–62
Pert CB, Aposhian D, Snyder SH (1974) Phylogenetic distribution of opiate receptor binding. Brain Res 75:356–361
Peters RH, Hughes RA (1978) Naloxone interactions with morphine and shock-potentiated tonic immobility in chickens. Pharmacol Biochem Behav 9:153–156
Simantov R, Goodman R, Aposhian D, Solomon H (1976) Phylogenetic distribution of morphine-like peptide “enkephalin”. Brain Res 111:204–211
Smits SE, Takemori AE (1970) Quantitative studies on the antagonism by naloxone of some narcotic and narcotic-antagonist analgesics. Br J Pharmacol 39:627–638
Stefano GB, Catapane EJ (1979) Enkephalins increase dopamine levels in the CNS of a marine mollusc. Life Sci 24:1617–1622
Stefano GB, Hiripi L (1979) Methionine enkephalin and morphine alter monoamine and cyclic nucleotide levels in the central ganglia of the freshwater bivalveAnodonta cygnea. Life Sci 25:291–298
Stefano GB, Kream RM, Zukin RS (1980) Demonstration of stereospecific opiate binding in the nervous tissue of the marine molluscMytilus edulis. Brain Res 181:440–445
Stretch R, Sanchez-Ramos JR (1979) Effect of morphine, naloxone and diallylnormorphine upon behaviour maintained by electricshock postponement in squirrel monkeys. Can J Physiol Pharmacol 57:98–105
Takemori AE, Kupferberg HJ, Miller JW (1969) Quantitative studies of the antagonism of morphine by nalorphine and naloxone. J Pharmacol Exp Ther 169:39–45
Wallnau LB, Gallup GG (1979) Morphine potentiation of tonic immobility: effects of naloxone, PCPA, and 5,6-DHT. Pharmacol Biochem Behav 10:499–504
Watson JS, Akil H, Sullivan S, Barchas JD (1977) Immunocyto- chemical localisation of methionine enkephalin: preliminary observations. Life Sci 21:733–738
Wei E, Sigel S, Way EL (1975) Regional sensitivity of the rat brain to the inhibitory effects of morphine on wet shake behaviour. J Pharmacol Exp Ther 193:56–63
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maldonado, H., Miralto, A. Effect of morphine and naloxone on a defensive response of the mantis shrimp (Squilla mantis). J. Comp. Physiol. 147, 455–459 (1982). https://doi.org/10.1007/BF00612010
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00612010