Abstract
The System for Continuous Observation of Rodents in Home-cage Environment (SCORHE) was developed to demonstrate the viability of compact and scalable designs for quantifying activity levels and behavior patterns for mice housed within a commercial ventilated cage rack. The SCORHE in-rack design provides day- and night-time monitoring with the consistency and convenience of the home-cage environment. The dual-video camera custom hardware design makes efficient use of space, does not require home-cage modification, and is animal-facility user-friendly. Given the system’s low cost and suitability for use in existing vivariums without modification to the animal husbandry procedures or housing setup, SCORHE opens up the potential for the wider use of automated video monitoring in animal facilities. SCORHE’s potential uses include day-to-day health monitoring, as well as advanced behavioral screening and ethology experiments, ranging from the assessment of the short- and long-term effects of experimental cancer treatments to the evaluation of mouse models. When used for phenotyping and animal model studies, SCORHE aims to eliminate the concerns often associated with many mouse-monitoring methods, such as circadian rhythm disruption, acclimation periods, lack of night-time measurements, and short monitoring periods. Custom software integrates two video streams to extract several mouse activity and behavior measures. Studies comparing the activity levels of ABCB5 knockout and HMGN1 overexpresser mice with their respective C57BL parental strains demonstrate SCORHE’s efficacy in characterizing the activity profiles for singly- and doubly-housed mice. Another study was conducted to demonstrate the ability of SCORHE to detect a change in activity resulting from administering a sedative.












Similar content being viewed by others
References
Abuhatzira, L., Shamir, A., Schones, D. E., Schäffer, A. A., & Bustin, M. (2011). The chromatin-binding protein hmgn1 regulates the expression of methyl CpG-binding protein 2 (MECP2) and affects the behavior of mice. Journal of Biological Chemistry, 286, 42051–42062. doi:10.1074/jbc.M111.300541
Baker, M. (2011). Animal models: Inside the minds of mice and men. Nature, 475, 123–128. doi:10.1038/475123a
Branson, K. (2007). Tracking multiple mice through severe occlusions. Unpublished Ph.D. dissertation, University of California, San Diego, La Jolla, CA.
Branson, K., & Belongie, S. (2005, June). Tracking multiple mouse contours (without too many samples). Paper presented at the Computer Vision and Pattern Recognition Conference (CVPR 2005), San Diego, CA.
Chu, E. R., Kim, E. K., Gonzalez, J. M., Ko, M. H., Liew, E. C., & Tan, J. C. H. (2014). Intraocular pressure measurement in acepromazine-sedated mice. Clinical & Experimental Ophthalmology. doi:10.1111/ceo.12157
de Chaumont, F., Coura, R. D., Serreau, P., Cressant, A., Chabout, J., Granon, S., & Olivo-Marin, J. C. (2012). Computerized video analysis of social interactions in mice. Nature Methods, 9, 410–417. doi:10.1038/nmeth.1924
Edelman, N. (2011, September). Automated phenotyping of mouse social behavior. Unpublished Master's thesis, Massachusetts Institute of Technology, Cambridge, MA.
Farah, R., Langlois, J. M. P., & Bilodeau, G. (2012). Catching a rat by its edglets. IEEE Transactions on Image Processing, 22, 668–678. doi:10.1109/tip.2012.2221726
Forsyth, D., & Ponce, J. (2003). Computer vision: A modern approach. Upper Saddle River: Prentice Hall.
Gillet, J.-P., & Gottesman, M. M. (2011). [Unpublished data]. Bethesda, MD: National Institutes of Health.
Goulding, E. H., Schenk, A. K., Juneja, P., MacKay, A. W., Wade, J. M., & Tecott, L. H. (2008). A robust automated system elucidates mouse home cage behavioral structure. Proceedings of the National Academy of Sciences, 105, 20575–20582. doi:10.1073/pnas.0809053106
Jhuang, H., Garrote, E., Yu, X., Khilnani, V., Poggio, T., Steele, A. D., & Serre, T. (2010). Automated home-cage behavioural phenotyping of mice. Nature Communications, 1, 68. doi:10.1038/ncomms1064
Kelly, J., & Bianchi, M. (2012). Mammalian sleep genetics. Neurogenetics, 13, 287–326. doi:10.1007/s10048-012-0341-x
Krashes, M. J., Koda, S., Ye, C., Rogan, S. C., Adams, A. C., Cusher, D. S., & Lowell, B. B. (2011). Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. Journal of Clinical Investigation, 121, 1424–1428. doi:10.1172/jci46229
Kravitz, A. V., Freeze, B. S., Parker, P. R. L., Kay, K., Thwin, M. T., Deisseroth, K., & Kreitzer, A. C. (2010). Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature, 466, 622–626. doi:10.1038/nature09159
Kumar, A., Vashist, A., Kumar, P., Kalonia, H., & Mishra, J. (2012). Potential role of licofelone, minocycline and their combination against chronic fatigue stress induced behavioral, biochemical and mitochondrial alterations in mice. Pharmacological Reports, 64, 1105–1115.
Malley, J. D., Malley, K. G., & Pajevic, S. (2011). Statistical learning for biomedical data. Cambridge, UK: Cambridge University Press.
Noldus, L. P. J. J., Spink, A. J., & Tegelenbosch, R. A. J. (2001). EthoVision: A versatile video tracking system for automation of behavioral experiments. Behavior Research Methods, Instruments, & Computers, 33, 398–414. doi:10.3758/BF03195394
Novak, C. M., Burghardt, P. R., & Levine, J. A. (2012). The use of a running wheel to measure activity in rodents: Relationship to energy balance, general activity, and reward. Neuroscience & Biobehavioral Reviews, 36, 1001–1014. doi:10.1016/j.neubiorev.2011.12.012
Ohayon, S., Avni, O., Taylor, A. L., Perona, P., & Roian Egnor, S. E. (2013). Automated multi-day tracking of marked mice for the analysis of social behaviour. Journal of Neuroscience Methods, 219, 10–19. doi:10.1016/j.jneumeth.2013.05.013
Portal, E., Riess, O., & Nguyen, H. P. (2013). Automated home cage assessment shows behavioral changes in a transgenic mouse model of spinocerebellar ataxia type 17. Behavioural Brain Research, 250, 157–165. doi:10.1016/j.bbr.2013.04.042
Schaefer, A. T., & Claridge-Chang, A. (2012). The surveillance state of behavioral automation. Current Opinion in Neurobiology, 22, 170–176. doi:10.1016/j.conb.2011.11.004
Spruijt, B. M., & DeVisser, L. (2006). Advanced behavioural screening: Automated home cage ethology. Drug Discovery Today: Technologies, 3, 231–237. doi:10.1016/j.ddtec.2006.06.010
Steele, A. D., Jackson, W. S., King, O. D., & Lindquist, S. (2007). The power of automated high-resolution behavior analysis revealed by its application to mouse models of Huntington’s and prion diseases. Proceedings of the National Academy of Sciences, 104, 1983–1988. doi:10.1073/pnas.0610779104
Weissbrod, A., Shapiro, A., Vasserman, G., Edry, L., Dayan, M., Yitzhaky, A., & Kimchi, T. (2013). Automated long-term tracking and social behavioural phenotyping of animal colonies within a semi-natural environment. Nature Communications, 4, 2018. doi:10.1038/ncomms3018
Zarringhalam, K., Ka, M. H., Kook, Y. H., Terranova, J. I., Suh, Y., King, O. D., & Um, M. (2012). An open system for automatic home-cage behavioral analysis and its application to male and female mouse models of Huntington’s disease. Behavioural Brain Research, 229, 216–225. doi:10.1016/j.bbr.2012.01.015
Zubidat, A. E., Nelson, R. J., & Haim, A. (2009). Photosensitivity to different light intensities in blind and sighted rodents. Journal of Experimental Biology, 212, 3857–3864. doi:10.1242/jeb.033969
Zubidat, A. E., Nelson, R. J., & Haim, A. (2010). Photoentrainment in blind and sighted rodent species: responses to photophase light with different wavelengths. The Journal of Experimental Biology, 213, 4213–4222. doi:10.1242/jeb.048629
Zurn, J. B., Hohmann, D., Dworkin, S. I., & Motai, Y. (2005, January). A real-time rodent tracking system for both light and dark cycle behavior analysis. Paper presented at the Seventh IEEE Workshop on Application of Computer Vision (WACV/MOTIONS’05), Breckenridge, CO.
Zurn, J. B., Xianhua, J., & Motai, Y. (2005, May). Video-based rodent activity measurement using near-infrared illumination. Paper presented at the IEEE Instrumentation and Measurement Technology Conference (IMTC 2005), Ottawa, ON, Canada.
Author note
The authors wish to thank Cumhur Demirkale for his assistance with the statistical analysis. G.H.S. wishes to thank Kristin Branson and Lex Kravitz for helpful discussions and input.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
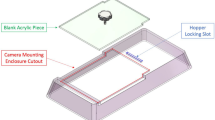
Dimension drawings of the SCORHE unit. a SCORHE unit in rack, with components color-coded for clarity. b Top view of the SCORHE unit out of the rack. c Front view of the SCORHE unit in rack. The shelf rail on which cages are suspended while in the rack is shown in more detail. d Side view of the SCORHE unit in the rack. Measurements of all color-coded elements are also included.
Rights and permissions
About this article
Cite this article
Salem, G.H., Dennis, J.U., Krynitsky, J. et al. SCORHE: A novel and practical approach to video monitoring of laboratory mice housed in vivarium cage racks. Behav Res 47, 235–250 (2015). https://doi.org/10.3758/s13428-014-0451-5
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13428-014-0451-5