Abstract
When memory is tested after a delay, performance is typically better if the retention interval includes sleep. However, it is unclear what accounts for this well-established effect. It is possible that sleep enhances the retrieval of information, but it is also possible that sleep protects against memory loss that normally occurs during waking activity. We developed a new research approach to investigate these possibilities. Participants learned a list of paired-associate items and were tested on the items after a 12-h interval that included waking or sleep. We analyzed the number of items gained versus the number of items lost across time. The sleep condition showed more items gained and fewer items lost than did the wake condition. Furthermore, the difference between the conditions (favoring sleep) in lost items was greater than the difference in gain, suggesting that loss prevention may primarily account for the effect of sleep on declarative memory consolidation. This finding may serve as an empirical constraint on theories of memory consolidation.
Similar content being viewed by others
Introduction
Nearly a century ago, Jenkins and Dallenbach (1924) had participants learn lists of nonsense syllables and then tested their memory for the lists at various points. Jenkins and Dallenbach found that memory performance was better after retention intervals that included sleep. This basic pattern of results has since been replicated by a number of researchers. For example, consistent with the results of a number of other studies (Ellenbogen, Hulbert, Jiang & Stickgold, 2009; Ellenbogen, Hulbert, Stickgold, Dinges & Thompson-Schill, 2006; Gais & Born, 2004; Gais, Lucas & Born, 2006; Gais, Molle, Helms & Born, 2002; Molle, Marshall, Gais & Born 2004; Plihal & Born, 1997, 1999), we found that participants’ memory for paired associates was better after a retention interval that included sleep than after one that only including waking activity (Fenn & Hambrick, 2012). Together with findings from research on nondeclarative memory (cf. Brawn, Fenn, Nusbaum & Margoliash, 2010; Fenn, Nusbaum & Margoliash, 2003; Huber, Ghilardi, Massimini & Tononi, 2004; Karni, Tanne, Rubenstein, Askenasy & Sagi, 1994; Stickgold, James & Hobson, 2000), this pattern of results suggests that a critical function of sleep is to consolidate new memories.
While the finding that sleep benefits declarative memory performance is well established, the question of what accounts for this fact remains in dispute in the literature. One possibility, which has been mentioned in media accounts of research on sleep and memory, is that a period of sleep enhances retrieval of information from long-term memory—that information that was not remembered before sleep becomes more accessible during sleep, and thus easier to remember after sleep. We alluded to this possibility to explain our finding that memory for paired associates was better after sleep than after being awake (Fenn & Hambrick, 2012).
Another possibility is that sleep protects against memory loss that normally occurs during waking activity, due to interference. In fact, one prominent theory of forgetting has argued that encoding of information during waking interferes with newly formed memory, and sleep benefits memory by providing a period of time when memory is safe from retroactive interference (Wixted, 2004). Consistent with this possibility, several studies have shown that when participants learn declarative information in the morning and remain awake for a full day prior to sleep, they show more forgetting at a 24-h test than participants who are trained in the evening and go to sleep a few hours after training (Benson & Feinberg, 1977; Gais et al., 2006; Nesca & Koulack, 1994; Payne, Tucker, Ellenbogen, Wamsley, Walker, Schacter & Stickgold, 2012).
Present study
Here, we used a new research approach to investigate these possible explanations for the finding of better memory performance after sleep than waking. The study took place in two sessions, following the procedure in our earlier study (Fenn & Hambrick, 2012). In Session 1, which was conducted in either the morning or the evening, we presented participants with a list of paired associates and tested their memory. In Session 2, which occurred after a 12-h retention interval that included being either awake or asleep, we gave participants a final memory test.
We then used item data to compute subscores reflecting different types of change in memory performance across sessions. Our major analyses focused on two subscores. Memory gain was the number of items that a participant recalled in the final test (in Session 2) that had not been recalled at any point during training (in Session 1), whereas memory loss was the number of items that a participant recalled at the final training test (in Session 1) that were not recalled in the final test (in Session 2).
Our research question was whether the sleep and wake conditions would differ in their average levels of memory gain and memory loss. The finding of a higher level of memory gain in the sleep condition than in the wake condition, combined with no difference between the conditions in memory loss, would suggest that the better memory performance after sleep is due primarily to enhanced retrieval of information. By contrast, the finding of a lower level of memory loss for the sleep than for the wake condition, combined with the lack of a difference between the conditions in memory gain, would suggest that this pattern is due primarily to protection against loss during sleep (see the supplementary online materials for more detailed discussion of this point).
As a secondary analysis, we tested for correlations between the memory gain and memory loss subscores. A finding that the subscores correlated significantly with each other would suggest that a common mechanism may contribute to both gain and loss. In contrast, a finding that the subscores correlated near zero with each other would suggest that the two subscores may reflect independent mechanisms.
Method
Participants
The participants were 495 native English speakers who reported no history of sleep or memory disorders. A number of the participants were excluded from all analyses because they did not complete the experiment (n = 38), because they reported diagnosed sleep or psychological disorders or habitual difficulty sleeping (n = 10), or because of experimenter error during the test (n = 4). An additional 89 participants were excluded because they reported napping during a waking retention interval;Footnote 1 naps of even very short duration can result in consolidation in this task (Lahl, Wispel, Willigens & Pietrowsky, 2008). The remaining participantsFootnote 2 were 354 undergraduate students between the ages of 18 and 35. Demographic information was not obtained from 72 of the participants; of the 282 who did report this information, 203 were female and 79 were male.
Stimuli
The stimulus set consisted of 48 pairs of semantically related nouns (from Fenn & Hambrick, 2012). The word pairs were adapted from Gais and Born (2004) and were matched for frequency, imagery, and concreteness (Francis & Kučera, 1982).
Procedure
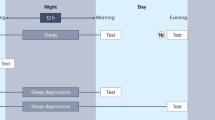
We conducted two experimental sessions, separated by a 12-h retention interval. For the wake condition (n = 165), the first session occurred at 9:00 and the second session occurred at 21:00. For the sleep condition (n = 189), the first session began at 21:00 and the second session began at 9:00 the following morning, after a regular sleep phase. The experimental sessions began within a 30-min window of these times.
In Session 1, participants studied 48 pairs of semantically related words and were tested on the word pairs. The stimuli were presented for 4,000 ms, with a 1,500-ms intertrial interval. Immediately after study, the participants were given a cued-recall test on 40 of the word pairs. The first four and the final four items presented during study never appeared on any of the tests, to control for primacy and recency effects on memory performance, as is standard in the literature. Testing on these items could have artificially increased immediate memory performance, particularly for items that remained in working memory. During the test, the first word of each pair was presented, and participants were given unlimited time to type the second word. After each response, participants were given two forms of feedback: They first were told whether their response was correct or incorrect, and then were shown the correct words in the pair. Participants were trained to a criterion of 60 % correct. If criterion was not met, the entire cued-recall test, including feedback, was repeated, until criterion was achieved. It should be noted that the final test during training always included feedback, so this test was also a learning trial. Thus, we expected performance to improve on the delayed test in Session 2. In Session 2, participants were given a final cued-recall test, without feedback. As in the previous test, the first word in each pair was presented, and the participants had unlimited time to respond. Items were presented randomly in the study phase and in all tests.
Memory measures
As was already mentioned, we computed subscores reflecting change in memory performance from Session 1 to Session 2. The two outcomes that were critical to the present investigation were memory gain and memory loss (Table 1). To reiterate, memory gain was the number of items that a participant recalled in the final test (in Session 2) that had not been recalled at any point during training (in Session 1), whereas memory loss was the number of items that a participant had recalled at the final training test (in Session 1) that were not then recalled in the final test (in Session 2). (Note that lost items may have also been correctly recalled during early training tests.)
We also computed subscores reflecting two other possible outcomes. The recovered subscore was the number of items that had been correctly recalled during any of the early training tests but not on the final training test, and that were then recalled correctly during Session 2. The lost-during-training subscore was the number of items that had been correctly recalled during at least one early training test, but that were not recalled during the final training test (and not recalled during Session 2).
Results
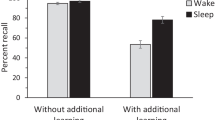
To determine whether the sleep condition showed greater improvement from the final training test (Session 1) to the final test (Session 2) than the wake condition, as in previous studies, we performed a repeated measures analysis of variance (ANOVA) with Condition (wake or sleep) as a between-subjects factor and Test (final training test or final test) as a within-subjects factor. We found main effects of condition [F(1, 352) = 9.46, p < .01] and test [F(1, 352) = 291.13, p < .001], as well as a Test × Condition interaction [F(1, 352) = 28.66, p < .001], indicating that the sleep condition improved more across the retention interval than did the wake condition (Fig. 1).
Thus, we replicated the finding of greater improvement in memory performance after a retention interval that includes sleep than after one that includes only waking activity. With this established, we performed analyses on the subscores to better understand the source of this effect. First, we performed a repeated measures ANOVA with Condition (wake or sleep) as a between-subjects factor and Subscore Type (gain or loss) as a within-subjects factor.Footnote 3 Main effects emerged of both condition, F(1, 352) = 24.73, p < .001, and subscore type, F(1, 352) = 1,721.86, p < .001. The effect of subscore type indicates a larger amount of gain than of loss across the conditions. We also found a significant Condition × Subscore Type interaction, F(1, 352) = 7.26, p < .01. The difference between the sleep and wake conditions in loss was greater than the difference between the conditions in gain (Fig. 2). Indeed, the sleep-versus-wake effect size was more than four times larger for loss (d = 0.69) than for gain (d = 0.17).Footnote 4
We also compared the conditions in average number of items recovered and lost during training for participants who took two or more tests to reach criterion (n = 222; 62.7 % of our sample). These values were quite low for both conditions (Table 2) and were not significantly different across conditions (ts < 1, ps > .4).
Correlations
The correlation between the gain and loss subscores approached statistical significance in the wake condition but was quite small (r = .14, p = .07), and was near zero in the sleep condition (r = .03, p = .42). Thus, there was some evidence that memory gain and memory loss may reflect independent mechanisms. Considering all four subscores, the average correlations were r = −.07 in the wake condition and r = .06 in the sleep condition (see the supplementary online materials).
Circadian and sleep analyses
Because participants in the wake condition were trained in the morning and tested in the evening, while participants in the sleep condition were trained in the evening and tested in the morning, it is possible that diurnal or circadian differences might explain our finding of less loss and greater gain in the sleep condition. To investigate this possibility, we compared the wake and sleep conditions in terms of performance on both the initial and the final training test, as well as on the number of tests to reach criterion. If one of our conditions performed better on these measures than the other did, this might suggest that paired-associate learning was better in either the morning or the evening. However, we found no evidence of circadian variation in this experiment: Recall on the first training test was not significantly different between the two conditions, t(352) = 0.09, p = .92, and the conditions also showed similar performance on their final training tests, t(352) = 0.62, p = .53 (Table 3). Furthermore, no significant difference was apparent in the numbers of tests to reach criterion, t(352) = 1.4, p = .14. Thus, our results cannot be attributed to performance differences based on time of day.
We also compared the amounts of self-reported sleep on the night of the study between our conditions. The wake condition reported an average of 6.96 ± 1.3 h (mean ± SD) of sleep, whereas the sleep condition reported an average of 6.79 ± 1.6 h.Footnote 5 These values were not significantly different, t(345) = 1.14, p = .25, and the trend was for the wake condition to report having slept more than the sleep condition. Thus, longer duration of sleep cannot explain the superior performance in the sleep condition.
Discussion
The finding that memory performance improves more after a retention interval that includes sleep than after one that includes only waking activity is well replicated, but still not well understood. Participants studied paired associates and were tested immediately after training and after a 12-h interval that was composed either entirely of waking or included sleep. We analyzed the numbers of items gained and lost across time and found that the sleep condition showed both greater gain of individual items and less loss of individual items than did the wake condition. The difference between the conditions in the numbers of items lost was greater than the difference in the numbers of items gained.
Although we have discussed gain in terms of an improvement in memory performance, it is important to note that participants received feedback on their final training test. That is, on each trial, they were given the correct word pair after they had responded. Thus, we can assume that memory improved as a result of the feedback given on this test. However, we did not test the participants after this criterion test. Therefore, our measure of memory performance on the final training test likely underestimated performance. This means that our measure of memory gain may not reflect actual memory improvement. Regardless, using the exact same task and same design that has been used to show memory enhancement across sleep, we have shown that the strongest effect of sleep on declarative memory is in protection against loss, not in memory gain.
The finding that sleep both increases gain and reduces loss raises the question of whether these two effects reflect the same underlying mechanism. One possibility is that both effects reflect protection against loss in the sleep condition. The smaller number of items lost in the sleep condition, as compared to the wake condition, likely reflects protection from interference and loss of memory. The increased gain in the sleep condition, however, may also reflect protection against loss. Both conditions were given feedback on the final training test. We therefore expected both conditions to improve between the final training test and the final test. If both groups acquired the same number of new items on the final training test, the reduced gain in the wake condition may actually reflect loss of some of the training-related gain in memory. For example, both groups may have acquired five new word pairs on the final training test. However, the wake group may have lost some of this initial gain, potentially due to interference. Therefore, the gain observed in the sleep group may reflect maintenance of memory from the final training test, whereas the lower amount of gain in the wake group may reflect some loss of memory that had been acquired on the final training test. Thus, it is possible that both the gain and loss scores reflect the same underlying mechanism: prevention against loss. This is consistent with early accounts of the role of sleep in memory performance (cf. Jenkins & Dallenbach, 1924) and with a more recent theory that has argued that sleep benefits memory by protecting it against retroactive interference, and thus, forgetting (Wixted, 2004).
If the gain and loss subscores do in fact reflect the same underlying mechanism, we would expect the subscores to be correlated with each other. However, this correlation was nonsignificant in both conditions. This finding may suggest that two mechanisms contribute to consolidation. We speculate that the mechanism underlying the reduced memory loss in the sleep condition is protection against interference. By contrast, the mechanism underlying increased memory gain in the sleep condition may be enhanced retrieval. Several explanations are possible for how sleep might enhance retrieval ability. For example, several studies have shown that neurons that are active during task activity are subsequently reactivated during sleep (Dave & Margoliash, 2000; Ji & Wilson, 2007; Louie & Wilson, 2001; O’Neill, Senior, Allen, Huxter & Csicsvari, 2008; Wilson & McNaughton, 1994), and this reactivation of a memory trace may enhance retrieval processes. Thus, it is possible that two separate consolidation mechanisms operate during sleep: a passive mechanism of protection from interference, and an active mechanism of information processing. While we cannot definitively argue for an active consolidation process, it is clear that even if sleep works to actively enhance retrieval processing, the strongest effects in this task are in protection against loss.
In conclusion, we have adopted a new approach to the study of declarative memory consolidation. To better understand the exact effect that sleep has on memory, future studies would benefit from employing the analytic approach described here. The pattern of results that we report also represents an important empirical constraint on theorization about sleep-related effects on declarative memory, in that proposed explanations for consolidation must be able to account not only for greater gain following sleep, but also for diminished loss.
Notes
The participants who napped also showed significantly less sleep on the night of the study than did either the wake or the sleep condition. A one-way ANOVA revealed a significant effect of condition (wake, sleep, or nap) on sleep duration F(2, 433) = 11.41, p < .001, and Scheffe’s post-hoc tests revealed that participants who napped showed significantly less sleep on average than did either the wake (p < .001) or the sleep (p < .001) condition. We therefore excluded participants who napped from all of the analyses.
All participants were recruited for an experiment requiring three separate sessions. In the third session, the participants returned to complete a cognitive test battery, but the results concerning those tests are to be described in a separate report.
We transformed the loss variable by multiplying each value by −1, so that better performance would be in the same direction for this subscore and the gain subscore.
One potential concern about this finding is that some participants scored near ceiling on the final training test, and therefore had little room for change in memory performance across the retention interval, thereby artificially producing this interaction. However, when participants who were at ceiling on the final training test were removed from the analysis, the interaction remained significant (see the supplemental online materials).
Four participants in the wake condition and three participants in the sleep condition did not report sleep duration.
References
Benson, K., & Feinberg, I. (1977). Beneficial effect of sleep in an extended Jenkins and Dallenbach paradigm. Psychophysiology, 14, 375–384.
Brawn, T. P., Fenn, K. M., Nusbaum, H. C., & Margoliash, D. (2010). Consolidating the effects of waking and sleep on motor-sequence learning. Journal of Neuroscience, 30, 13977–13982.
Dave, A. S., & Margoliash, D. (2000). Song replay during sleep and computational rules for sensorimotor vocal learning. Science, 290, 812–816.
Ellenbogen, J. M., Hulbert, J. C., Jiang, Y., & Stickgold, R. (2009). The sleeping brain’s influence on verbal memory: Boosting resistance to interference. PLoS ONE, 4, e4117. doi:10.1371/journal.pone.0004117
Ellenbogen, J. M., Hulbert, J. C., Stickgold, R., Dinges, D. F., & Thompson-Schill, S. L. (2006). Interfering with theories of sleep and memory: Sleep, declarative memory, and associative interference. Current Biology, 16, 1290–1294.
Fenn, K. M., & Hambrick, D. Z. (2012). Individual differences in working memory capacity predict sleep-dependent memory consolidation. Journal of Experimental Psychology: General, 141, 404–410.
Fenn, K. M., Nusbaum, H. C., & Margoliash, D. (2003). Consolidation during sleep of perceptual learning of spoken language. Nature, 425, 614–616.
Francis, W. N., & Kučera, H. (1982). Frequency analysis of English usage: Lexicon and grammar. Boston, MA: Houghton Mifflin.
Gais, S., & Born, J. (2004). Low acetylcholine during slow-wave sleep is critical for declarative memory consolidation. Proceedings of the National Academy of Sciences, 101, 2140–2144.
Gais, S., Lucas, B., & Born, J. (2006). Sleep after learning aids memory recall. Learning and Memory, 13, 259–262.
Gais, S., Molle, M., Helms, K., & Born, J. (2002). Learning-dependent increases in sleep spindle density. Journal of Neuroscience, 22, 6830–6834.
Huber, R., Ghilardi, M. F., Massimini, M., & Tononi, G. (2004). Local sleep and learning. Nature, 430, 78–81.
Jenkins, J. M., & Dallenbach, K. M. (1924). Obliviscence during sleep and waking. American Journal of Psychology, 35, 605–612.
Ji, D., & Wilson, M. A. (2007). Coordinated memory replay in the visual cortex and hippocamus during sleep. Nature Neuroscience, 10, 100–107.
Karni, A., Tanne, D., Rubenstein, B. S., Askenasy, J. J. M., & Sagi, D. (1994). Dependence on REM sleep of overnight perceptual skill. Science, 265, 679–682.
Lahl, O., Wispel, C., Willigens, B., & Pietrowsky, R. (2008). An ultra short episode of sleep is sufficient to promote declarative memory performance. Journal of Sleep Research, 17, 3–10.
Louie, K., & Wilson, M. A. (2001). Temporally structured replay of awake hippocampal ensemble activity during rapid eye movement sleep. Neuron, 29, 145–156.
Molle, M., Marshall, L., Gais, S., & Born, J. (2004). Learning increases human electroencephalographic coherence during subsequent slow sleep oscillations. Proceedings of the National Academy of Sciences, 101, 13963–13968.
Nesca, M., & Koulack, D. (1994). Recognition memory, sleep and circadian rhythms. Canadian Journal of Experimental Psychology, 48, 359–379.
O’Neill, J., Senior, T. J., Allen, K., Huxter, J. R., & Csicsvari, J. (2008). Reactivation of experience-dependent cell assembly patterns in the hippocampus. Nature Neuroscience, 11, 209–215.
Payne, J. D., Tucker, M. A., Ellenbogen, J. M., Wamsley, E. J., Walker, M. P., Schacter, D. L., & Stickgold, R. (2012). Memory for semantically related and unrelated declarative information: The benefit of sleep, the cost of wake. PLoS ONE, 7, e33079. doi:10.1371/journal.pone.0033079
Plihal, W., & Born, J. (1997). Effects of early and late nocturnal sleep on declarative and procedural memory. Journal of Cognitive Neuroscience, 9, 534–547.
Plihal, W., & Born, J. (1999). Memory consolidation in human sleep depends on inhibition of glucocorticoid release. Learning and Memory, 10, 2741–2747.
Stickgold, R., James, L., & Hobson, J. A. (2000). Visual discrimination learning requires sleep after training. Nature Neuroscience, 3, 1237–1238.
Wilson, M. A., & McNaughton, B. L. (1994). Reactivation of hippocampal ensemble memories during sleep. Science, 265, 676–679.
Wixted, J. T. (2004). The psychology and neuroscience of forgetting. Annual Review of Psychology, 55, 235–269. doi:10.1146/annurev.psych.55.090902.141555
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 163 kb)
Rights and permissions
About this article
Cite this article
Fenn, K.M., Hambrick, D.Z. What drives sleep-dependent memory consolidation: Greater gain or less loss?. Psychon Bull Rev 20, 501–506 (2013). https://doi.org/10.3758/s13423-012-0366-z
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13423-012-0366-z