Abstract
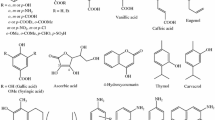
A Quantitative Structure-Activity Relationship (QSAR) of coumarins by genetic algorithms employing physicochemical, topological, lipophilic and electronic descriptors was performed. We have used experimental antioxidant activities of specific coumarin derivatives against the DPPH· radical molecule. Molecular descriptors such as Randic Path/Walk, hydrophilic factor and chemical hardness were selected to propose a mathematical model. We obtained a linear correlation with R2 = 96.65 and Q 2 LOO = 93.14 values. The evaluation of the predictive ability of the model was performed by applying the Q 2 ASYM , \(\hat r^2 \) and Δr 2 m methods. Fukui functions were calculated here for coumarin derivatives in order to delve into the mechanics by which they work as primary antioxidants. We also investigated xanthine oxidase inhibition with these coumarins by molecular docking. Our results show that hydrophobic, electrostatic and hydrogen bond interactions are crucial in the inhibition of xanthine oxidase by coumarins.

Similar content being viewed by others
References
I. Kostova, Curr. Med. Chem. 5, 29 (2005)
D. Berg, M.B.H. Youdim, P. Riederer, Cell Tissue Res. 318, 201 (2004)
M. Garazd, O. Muzychka, A. Vovk, I. Nagorichna, A. Ogorodniichuk, Chem. Nat. Compd. 43, 19 (2007)
A. Kotali, I. Lafazanis, P. Harris, Synthetic Communications 38, 3996 (2008)
M. Traykova, I. Kostova, Int. J. Pharm. 1, 29 (2005)
J.Z. Pedersen, C. Oliveira, S. Incerpi, V. Kumar, A.M. Fiore, P.D Vito, A.K. Prasad, S.V. Malhotra, V.S. Parmar, L. Saso, J. Pharm. Pharmacol. 59, 1721 (2007)
K.C. Fylaktakidou, D.J. Hadjipavlou-Litina, K.E. Litinas, D.N. Nicolaides, Curr. Pharm. Design 10, 3813 (2004)
A.A.H Kadhum, A.A. Al-Amiery, A.Y. Musa, A.B. Mohamad, Int. J. Mol. Sci. 12, 5747 (2011)
J.D. Lambeth, Nat. Rev. Immun. 4, 181 (2004)
T. Inoguchi, T. Sonta, H. Tsubouchi, T. Etoh, M. Kakimoto, N. Sonoda, N. Sato, N. Sekiguchi, K. Kobayashi, H. Sumimoto, H. Utsumi, H. Nawata, J. Am. Soc. Nephro. 14, S227 (2003)
J.S. McNally, M.E. Davis, D.P. Giddens, A. Saha, J. Hwang, S. Dikalov, H. Jo, D.G. Harrison, Am. J. Physio I. 285, H2290 (2003)
Y.C. Chang, F.W. Lee, C.S. Chen, S.T. Huang, S.H. Tsai, S.H. Huang, C.M. Lin, FRBM 43, 1541 (2007)
Jr. Merz, M. Kenneth, D. Ringe, C.H. Reynolds, Drug Design: Structure- and Ligand-Based Approaches (Cambridge University Press, New York, 2010)
R. Benigni, C. Bossa, J. Chem. Inf. Model 48, 971 (2008)
F.A. Pasha, S.J. Cho, Y. Beg, Y.B. Tripathi, Med. Chem. Res. 16, 408 (2007)
Y. Xue, Y. Zheng, L. An, L. Zhang, Y. Qian, D. Yu, X. Gong, Y. Liu, Comp. Theor. Chem. 982, 74 (2012)
R.G. Parr, W.T. Yang, J. Am. Chem. Soc. 106(14), 4049 (1984)
P. Fuentealba, E. Florez, W. Tiznado, J. Chem. Theor. Comp. 6, 1470 (2010)
C. Cardenas, W. Tiznado, P.W. Ayers, P. Fuentealba, J. Phys. Chem. A 115, 2325 (2011)
P. Geerlings, F.D. Proft, W. Langenaeker, Chem. Rev. 103, 1793 (2003)
P.W. Ayers, J. Math. Chem. 43, 285 (2008)
J.P. Perdew, R.G. Parr, M. Levy, J.L.J. Balduz, Phys. Rev. Lett. 49, 1691 (1982)
P.W. Ayers, M. Levy, Theor. Chem. Acc. 103, 353 (2000)
P.W. Ayers, W.T. Yang, L.J. Bartolotti, P.K. Chattaraj, Fukui function chemical reactivity theory: A density functional. Boca Raton: CRC Press, 255 (2009)
W. Kohn, L.J. Sham, Phys. Rev. 140, A1133 (1965)
P. Hohenberg, W. Kohn, Phys. Rev. 136, B864 (1964)
R.G. Parr, W. Yang, Density functional theory of atoms and molecules (Oxford University Press, New York, 1989)
W. Yang, R.G. Parr, R. Pucci, J. Chem. Phys. 81, 2862 (1984)
A. Michalak, F. De Proft, P. Geerlings, R.F. Nalewajski, J. Phys. Chem. A 103(6), 762 (1999)
R. Balawender, P. Geerlings, J. Chem. Phys. 123(12), 124103 (2005)
R. Flores-Moreno, A.M. Köster, J. Chem. Phys. 128(13), 134105 (2008)
R. Flores-Moreno, J. Melin, J.V. Ortiz, G. Merino, J. Chem. Phys. 129(22), 224105 (2008)
R. Flores-Moreno, J. Chem. Theor. Comp. 6(1), 48 (2010)
W. Yang, A.J. Cohen, F. De Proft, P. Geerlings, J. Chem. Phys. 136(14), 144110 (2012)
H.C. Lin, S.H. Tsai, C.S. Chen, Y.C. Chang, C.M. Lee, Z.Y. Lai, C.M. Lin, Biochem. Pharm. 75, 1416 (2008)
F.J. Martinez-Martinez, R.S. Razo-Hernandez, A.L. Peraza-Campos, M. Villanueva-Garcia, M.T. Sumaya-Martinez, D. Jaramillo-Cano, Z. Gomez-Sandoval, Molecules 17, 14882 (2012)
J.J.P. Stewart, J. Comput. Chem. 10, 209 (1989)
W.J. Hehre, Guide to Molecular Mechanics and Quantum Chemical Calculations, (Wavefunction Inc. Irvine, California, 2003)
A.D. Becke, J. Chem. Phys. 98, 5648 (1993)
G.A. Petersson, M.A. Al-Laham, J. Chem. Phys. 94, 6081 (1991)
Gaussian 09, Revision A.1, M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, Ö. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox (Gaussian, Inc., Wallingford CT, 2009)
F.A. Pasha, S.J. Cho, Y. Beg, Y.B. Tripathi, Med. Chem. Res. 408 (2007)
R. Todeschini, M. Vighi, A. Finizio and P. Gramatica, SAR QSAR Envir. Res. 7(1–4) 173 (1997)
R.G. Parr, W. Yang, Chemical potential derivatives. Density-Functional Theory of Atoms and Molecules (Oxford University Press, New York, 1989) 87–95
P.K. Chattaraj, D.R. Roy, Chem. Rev. 107(9), PR46 (2007)
A. Sakar, T.R. Middya, A.D. Jana, J. Mol. Model 18, 2621 (2012)
M. Randic, J. Chem. Inf. Model 41, 607 (2001)
A.K. Ghose, V.N. Viswanadhan, J.J. Wendoloski, J. Phys. Chem. A 102, 3762 (1998)
A.K. Ghose, G.M. Crippen, J. Chem. Infor. Comp. Sci. 27, 21 (1987)
DRAGON for Windows, Version 5.4. (TALETE srl, Milano, Italy, 2006)
R. Thomsen, M.H. Christensen, J. Med. Chem. 49, 3315 (2006)
A.M. Köster, G. Geudtner, P. Calaminici, M.E. Casida, V.D. Dominguez, R. Flores-Moreno, G.U. Gamboa, A. Goursot, T. Heine, A. Ipatov, F. Janetzko, J.M. del Campo, J.U. Reveles, A. Vela, B. Zuñiga-Gutierrez, D.R. Salahub, deMon2k version 3. [Online] Cinvestav, (2011), http://www.demon-software.com
P.A.M. Dirac, P. Roy, Soc. London A 117, 610 (1928)
S.H. Vosko, L. Wilk, M. Nusair, Can. J. Phys. 58, 1200 (1980)
N. Godbout, D.R. Salahub, J. Andzelm, E. Wimmer, Can. J. Phys. 70, 560 (1992)
J. Andzelm, E. Radzio, D.R. Salahub, J. Comput. Chem. 6, 520 (1985)
J. Andzelm, N. Russo, D.R. Salahub, J. Chem. Phys. 87, 6562 (1987)
R. Flores-Moreno, K. Pineda-Urbina, Z. Gomez-Sandoval, Sinapsis Version XII-V. Sinapsis developers. [Online] http://sinapsis.sourceforge.net.
R. Leardi, in: R. Todeschini, V. Consonni, A. Mauri, M. Pavan (Eds.), MobyDigs: software for regression and classification models by genetic algorithms. Nature-inspired Methods in Chemometrics: Genetic Algorithms and Artificial Neural Networks (Elsevier, Amsterdam, 2003) 141–167
R. Todeschini, D. Ballabio, V. Consonni, A. Mauri, M. Pavan, MobyDigs-Version 1.0. s.l: TALETE srl, (2004)
I.E. Frank, R. Todeschini, The Data Analysis Handbook (Elsevier, Amsterdam, Netherlands, 1994)
R. Todeschini, V. Consonni, A. Maiocchi, Chemometrics Intell. Lab. Sys. 46(1), 13 (1999)
R. Todeschini, Anal. Chim. Acta 348, 419 (1997)
R. Todeschini, V. Consonni, A. Mauri, M. Pavan, Anal. Chim. Acta 515, 199 (2004)
G. Cruciani, M. Baroni, S. Clementi, G. Costantino, D. Riganelli, B. Skagerberg, J. Chemometr. 6, 347 (1992).
U. Wählby, E.N. Jonsson, M.O. Karlsson, J. Am. Assoc. Pharm. Sci. 4, 68 (2002)
P. K. Ojha, I. Mitra, R.N. Das, K. Roy, Chemometrics Intell. Lab. Sys. 107(1), 194 (2011)
K. Roy, I. Mitra, S. Kar, P.K. Ojha, R.N. Das, H. Kabir, J. Chem. Inf. Model. 52(2), 396 (2012)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
About this article
Cite this article
Razo-Hernández, R.S., Pineda-Urbina, K., Velazco-Medel, M.A. et al. QSAR study of the DPPH· radical scavenging activity of coumarin derivatives and xanthine oxidase inhibition by molecular docking. cent.eur.j.chem. 12, 1067–1080 (2014). https://doi.org/10.2478/s11532-014-0555-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11532-014-0555-x