Abstract
Introduction
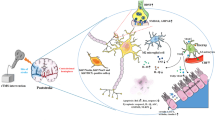
Neurogenic neuroprotection elicited by deep brain stimulation is emerging as a promising approach for treating patients with ischemic brain lesions. In rats, stimulation of the fastigial nucleus, but not dentate nucleus, has been shown to reduce the volume of focal infarction. Protection of neural tissue is a rapid intervention that has a relatively long-lasting effect, rendering fastigial nucleus stimulation (FNS) a potentially valuable method for clinical application.
Methods
We review some of the main findings of animal experimental research from a clinical perspective. Results: Although the complete mechanisms of neuroprotection induced by FNS remain unclear, important data has been presented in the last two decades. The acute effect of electrical stimulation of the fastigial nucleus is likely mediated by a prolonged opening of potassium channels, and the sustained effect appears to be linked to inhibition of the apoptotic cascade.
Conclusion
A better understanding of the cellular and molecular mechanisms underlying neurogenic neuroprotection by stimulation of deep brain nuclei, with special attention to the fastigial nucleus, can contribute toward improving neurological outcomes in ischemic brain insults.
Similar content being viewed by others
References
Alkawi A., Kirmani J.F., Janjua N., Khatri I., Ahmed S., Divani A.A., et al., Advances in thrombolytics and mechanical devices for treatment of acute ischemic stroke, Neurol. Res., 2005, 27(Suppl. 1), S42–49
Chang Y.C., Shyu W.C., Lin S.Z., Li H., Regenerative therapy for stroke, Cell Transplant., 2007, 16, 171–181
Feigin V.L., Findlay M., Advances in subarachnoid hemorrhage, Stroke, 2006, 37, 305–308
Ramani R., Hypothermia for brain protection and resuscitation, Curr. Opin. Anaesthesiol., 2006, 19, 487–491
Fisher M., Henninger N., Translational research in stroke: taking advances in the pathophysiology and treatment of stroke from the experimental setting to clinical trials, Curr. Neurol. Neurosci. Rep., 2007, 7, 35–41
Dorhout S.M., Rinkel G.J., Hop J.W., Algra A., van Gijn J., Antiplatelet therapy in aneurysmal subarachnoid hemorrhage: a systematic review, Stroke, 2003, 34, 285–289
Loch M.R., Management of cerebral vasospasm, Neurosurg. Rev., 2006, 29, 179–193
Roos Y., Rinkel G., Vermeulen M., Algra A., van Gijn J., Antifibrinolytic therapy for aneurysmal subarachnoid hemorrhage: a major update of a Cochrane review, Stroke, 2003, 34, 2308–2309
Mistri A.K., Robinson T.G., Potter J.F., Pressor therapy in acute ischemic stroke: systematic review, Stroke, 2006, 37, 1565–1571
Baron J.C., How healthy is the acutely reperfused ischemic penumbra?, Cerebrovasc. Dis., 2005, 20, S25–S31
Baron J.C., Mapping the ischaemic penumbra with PET: implications for acute stroke treatment, Cerebrovasc. Dis., 1999, 9, 193–201
Ginsberg M.D., Pulsinelli W.A., The ischemic penumbra, injury thresholds, and the therapeutic window for acute stroke, Ann. Neurol., 1994, 36, 553–554
Hakim A.M., The cerebral ischemic penumbra, Can. J. Neurol. Sci., 1987, 14, 557–559
Kidwell C.S., Alger J.R., Saver J.L., Beyond mismatch: evolving paradigms in imaging the ischemic penumbra with multimodal magnetic resonance imaging, Stroke, 2003, 34, 2729–2735
Kidwell C.S., Alger J.R., Saver J.L., Evolving paradigms in neuroimaging of the ischemic penumbra, Stroke, 2004, 35, 2662–2665
Peek K.E., Lockwood A.H., Izumiyama M., Yap E.W., Labove J., Glucose metabolism and acidosis in the metabolic penumbra of rat brain, Metab. Brain Dis., 1989, 4, 261–272
Hossmann K.A., Viability thresholds and the penumbra of focal ischemia, Ann. Neurol., 1994, 36, 557–565
Glickstein S.B., Golanov E.V., Reis D.J., Intrinsic neurons of fastigial nucleus mediate neurogenic neuroprotection against excitotoxic and ischemic neuronal injury in rat, J. Neurosci., 1999, 19, 4142–4154
Golanov E.V., Liu F., Reis D.J., Stimulation of cerebellum protects hippocampal neurons from global ischemia, Neuroreport, 1998, 9, 819–824
Reis D.J., Golanov E.V., Galea E., Feinstein D.L., Central neurogenic neuroprotection: central neural systems that protect the brain from hypoxia and ischemia, Ann. NY Acad. Sci., 1997, 835, 168–186
Reis D.J., Berger S.B., Underwood M.D., Khayata M., Electrical stimulation of cerebellar fastigial nucleus reduces ischemic infarction elicited by middle cerebral artery occlusion in rat, J. Cereb. Blood Flow Metab., 1991, 11, 810–818
Reis D.J., Underwood M.D., Berger S.B., Khayata M., Zaiens N.I., Fastigial nucleus stimulation reduces the volume of cerebral infarction produced by occlusion of the middle cerebral artery in rat, In: Seylaz J., MacKenzie M.T. (Eds.), Neurotransmission and cerebrovascular function I, Elsevier, Amsterdam, 1989, 401–404
Reis D.J., Kobylarz K., Yamamoto S., Golanov E.V., Brief electrical stimulation of cerebellar fastigial nucleus conditions longlasting salvage from focal cerebral ischemia: conditioned central neurogenic neuroprotection, Brain Res., 1998, 780, 159–163
Vieussens R., Neurographia universalis, Lyons, Jean Certe, 1684
Stilling B., Untersuchungen über den Bau des kleinen Gehirns des Menschen, T. Fischer (vol. 3), Cassel, 1864
Achar N.K., Downman C.B., Autonomic responses evoked by stimulation of fastigial nuclei in the anaesthetized cat, J. Physiol. (Lond), 1969, 204, 130
Miura M., Reis D.J., A blood pressure response from fastigial nucleus and its relay pathway in brainstem, Am. J. Physiol., 1970, 219, 1330–1336
Miura M., Reis D.J., Cerebellum: A pressor response elicited from the fastigial nucleus and its efferent pathway in brainstem, Brain. Res., 1969, 13, 595–599
Yamamoto S., Golanov E.V., Reis D.J., Reductions in focal ischemic infarctions elicited from cerebellar fastigial nucleus do not result from elevations in cerebral blood flow, J. Cereb. Blood Flow Metab., 1993, 13, 1020–1024
Golanov E.V., Zhou P., Neurogenic neuroprotection, Cell. Mol. Neurobiol., 2003, 23, 651–663
Golanov E.V., Reis D.J., Cerebral cortical neurons with activity linked to central neurogenic spontaneous and evoked elevations in cerebral blood flow, Neurosci. Lett., 1996, 209, 101–104
Garcia-March G., Sanchez-Ledesma M.J., Anaya J., Broseta J., Cerebral and carotid hemodynamic changes following cervical spinal cord stimulation, An experimental study, Acta Neurochir. Suppl. (Wien), 1989, 46, 102–104
Chida K., Iadecola C., Underwood M.D., Reis D.J., A novel vasodepressor response elicited from the rat cerebellar fastigial nucleus: the fastigial depressor response, Brain Res., 1986, 370, 378–382
Iadecola C., Zhang F., Xu X., Inhibition of inducible nitric oxide synthase ameliorates cerebral ischemic damage, Am. J. Physiol., 1995, 268, 286–292
Nakai M., Iadecola C., Ruggiero D.A., Tucker L.W., Reis D.J., Electrical stimulation of cerebellar fastigial nucleus increases cerebral cortical blood flow without change in local metabolism: evidence for an intrinsic system in brain for primary vasodilation, Brain Res., 1983, 260, 35–49
Bradley D.J., Pascoe J.P., Paton J.F., Spyer K.M., Cardiovascular and respiratory responses evoked from the posterior cerebellar cortex and fastigial nucleus in the cat, J. Physiol. (Lond.), 1987, 393, 107–121
Henry R.T., Connor J.D., Axons of passage may be responsible for fastigial nucleus pressor response, Am. J. Physiol., 1989, 257, 1436–1440
Haroian A.J., Massopust L.C., Young P.A., Cerebellothalamic projections in the rat: an autoradiographic and degeneration study, J. Comp. Neurol., 1981, 197, 217–236
Batton R.R., Jayaraman A., Ruggiero B., Carpenter M.B., Fastigial efferent projections in the monkey: an autoradiographic study, J. Comp. Neurol., 1977, 174, 281–305
Snider R.S., Maiti A., Cerebellar contributions to the Papez circuit, J. Neurosci. Res., 1976, 2, 133–146
Dietrichs E., Haines D.E., Do the same hypothalamic neurons project to both amygdala and cerebellum?, Brain Res., 1986, 364, 241–248
Glickstein S.B., Ilch C.P., Reis D.J., Golanov E.V., Stimulation of the subthalamic vasodilator area and fastigial nucleus independently protects the brain against focal ischemia, Brain Res., 2001, 912, 47–59
Glickstein S.B., Ilch C.P., Golanov E.V., Electrical stimulation of the dorsal periaqueductal gray decreases volume of the brain infarction independently of accompanying hypertension and cerebrovasodilation, Brain Res., 2003, 994, 135–145
Person R.J., Andrezik J.A., Dormer K.J., Foreman R.D., Fastigial nucleus projections in the midbrain and thalamus in dogs, Neuroscience, 1986, 18, 105–120
Noda H., Sugita S., Ikeda Y., Afferent and efferent connections of the oculomotor region of the fastigial nucleus in the macaque monkey, J. Comp. Neurol., 1990, 302, 330–348
Beitz A.J., The organization of afferent projections to the midbrain periaqueductal gray of the rat, Neuroscience, 1982, 7, 133–159
Meller S.T., Dennis B.J., Efferent projections of the periaqueductal gray in the rabbit, Neuroscience, 1991, 40, 191–216
Reichling D.B., Basbaum A.I., Collateralization of periaqueductal gray neurons to forebrain or diencephalon and to the medullary nucleus raphe magnus in the rat, Neuroscience, 1991, 42, 183–200
Rinvik E., Wiberg M., Demonstration of a reciprocal connection between the periaqueductal gray matter and the reticular nucleus of the thalamus, Anat. Embryol., 1990, 181, 577–584
Blix A.S., Cardiovascular adjustments to diving in mammals and birds, In: Renkin E.M., Michel C.C., The cardiovascular system, Bethesda, American Physiological Society, 1983, 917–945
Norris D.G., Principles of magnetic resonance assessment of brain function, J. Magn. Reson. Imaging, 2006, 23, 794–807
Toma K., Nakai T., Functional MRI in human motor control studies and clinical applications, Magn. Reson. Med. Sci., 2002, 1, 109–120
Golanov E.V., Reis D.J., Contribution of oxygen-sensitive neurons of the rostral ventrolateral medulla to hypoxic cerebral vasodilatation in the rat, J. Physiol. (Lond.), 1996, 495, 201–216
Hamer J., Hoyer S., Alberti E., Weinhardt F., Cerebral blood flow and oxidative brain metabolism during and after moderate and profound arterial hypoxaemia, Acta Neurochir. (Wien), 1976, 33, 141–150
Sun M.K., Reis D.J., Differential responses of barosensitive neurons of rostral ventrolateral medulla to hypoxia in rats, Brain Res., 1993, 609, 333–337
Underwood M.D., Iadecola D., Reis D.J., Lesions of the rostral ventrolateral medulla reduce the cerebrovascular response to hypoxia, Brain Res., 1994, 635, 217–223
Sun M.K., Reis D.J., Differential responses of barosensitive neurons of rostral ventrolateral medulla to hypoxia in rats, Brain Res., 1993, 609, 333–337
Sun M.K., Reis D.J., Hypoxic excitation of medullary vasomotor neurons in rats are not mediated by glutamate or nitric oxide, Neurosci. Lett., 1993, 157, 219–222
Abols I.A., Basbaum A.I., Afferent connections of the rostral medulla of the cat: a neural substrate for midbrain — medullary interactions in the modulation of pain, J. Comp. Neurol., 1981, 201, 285–297
Golanov E.V., Ruggiero D.A., Reis D.J., A brainstem area mediating cerebrovascular and EEG responses to hypoxic excitation of rostral ventrolateral medulla in rat, J. Physiol., 2000, 529, 413–429
Lovick T.A., Midbrain influences on ventrolateralmedullo-spinal neurones in the rat, Exp. Brain Res., 1992, 90, 147–152
Lovick T.A., The periaqueductal gray -rostral medulla connection in the defense reaction-efferent pathways and descending control mechanisms, Behav. Brain Res., 1993, 58, 19–25
Van Bockstaele E.J., Aston-Jones G., Pieribone V.A., Ennis M., Shipley M.T., Subregions of the periaqueductal gray topographically innervate the rostral ventral medulla in the rat, J. Comp. Neurol., 1991, 309, 305–327
Chida K., Underwood M.D., Miyagawha M., Participation of the rostral ventrolateral medulla in the cerebral blood flow of rats: Effects of stimulation and lesions on systemic and cerebral circulations, Ther. Res., 1990, 11, 77–85
Chida K., Iadecola C., Reis D.J., Lesions of rostral ventrolateral medulla abolish some cardio- and cerebrovascular components of the cerebellar fastigialpressor and depressor responses, Brain Res., 1990, 508, 93–104
Ruggiero D.A., Anware M., Golanov E.V., Reis D.J., The pedunculopontine tegmental nucleus issues collaterals to the fastigial nucleus and rostral ventrolateral reticular nucleus in the rat, Brain Res., 1997, 760, 272–276
Sun M.K., Reis D.J., Hypoxia-activated Ca2+currents in pacemaker neurones of rat rostral ventrolateral medulla in vitro, J. Physiol., 1994, 476, 101–116
Golanov E.V., Christensen J., Reis D.J., Neurons of a subthalamiccerebrovasodilator area (SCA) mediate elevations in cerebral flood flow (rCBF) evoked by excitation of neurons of rostral ventrolateral medulla, Soc. Neurosci. Abstr., 1999, 25, 12
Golanov E.V., Reis D.J., Vasodilation evoked from medulla and cerebellum is coupled to bursts of cortical EEG activity in rats, Am. J. Physiol., 1995, 268, 454–467
Dormer K.J., Foreman R.D., Ohata C.A., Fastigial nucleus stimulation and excitatory spinal sympathetic activity in dog, Am. J. Physiol., 1982, 243, 25–33
Nakai M., Iadecola C., Reis D.J., Global cerebral vasodilation by stimulation of rat fastigial cerebellar nucleus, Am. J. Physiol., 1982, 243, 226–235
Baron J.C., Bousser M.G., Rey A., Guillard A., Comar D., Castaigne P., Reversal of focal “misery-perfusion syndrome” by extra-intracranial arterial bypass in hemodynamic cerebral ischemia. A case study with 15O positron emission tomography, Stroke, 1981, 12, 454–459
Chida K., Iadecola C., Reis D.J., Global reduction in cerebral blood flow and metabolism elicited from intrinsic neurons of fastigial nucleus, Brain Res., 1989, 500, 177–192
Mraovitch S., Iadecola C., Ruggiero D.A., Reis D.J., Widespread reductions in cerebral blood flow and metabolism elicited by electrical stimulation of the parabrachial nucleus in rat, Brain Res., 1985, 341, 283–296
Leao A.A.P., Spreading depression of activity in the cerebral cortex, J. Neurophysiol., 1944, 4, 359–390
Golanov E.V., Reis D.J., Neuroprotective electrical stimulation of cerebellar fastigial nucleus attenuates expression of periinfarction depolarizing waves (PIDs) and inhibits cortical spreading depression, Brain Res., 1999, 818, 304–315
Mies G., Kohno K., Hossmann K.A., Prevention of periinfarct direct current shifts with glutamate antagonist NBQX following occlusion of the middle cerebral artery in the rat, J. Cereb. Blood Flow Metab., 1994, 14, 802–807
Ochs S., The nature of spreading depression in neural networks, Int. Rev. Neurobiol., 1962, 4, 1–69
Golanov E.V., Christensen J.D., Reis J.D., Role of potassium channels in the central neurogenic neuroprotection elicited by cerebellar stimulation in rat, Brain Res., 1999, 842, 496–500
Shimizu K., Bari F., Busija D.W., Glibenclamide enhances cortical spreading depression-associated hyperemia in the rat, Neuroreport, 2000, 11, 2103–2106
Lassmann H., Zimprich F., Rossler K., Vass K., Inflammation in the nervous system. Basic mechanisms and immunological concepts, Rev. Neurol., 1991, 147, 763–781
Heneka M.T., Galea E., Gavriluyk V., Dumitrescu-Ozimek L., Daeschner J., O’Banion M.K., et al., Noradrenergic depletion potentiates betaamyloid induced cortical inflammation: Implications for Alzheimer’s disease, J. Neurosci., 2002, 22, 2434–2442
Heneka M.T., Wiesinger H., Dumitrescu-Ozimek L., Riederer P., Feinstein D.L., Klockgether T., Neuronal and glial coexpression of argininosuccinatesynthetase and inducible nitric oxide synthase in Alzheimer disease, J. Neuropathol. Exp. Neurol., 2001, 60, 906–916
Galea E., Golanov E.V., Feinstein D.L., Kobylarz K.A., Glickstein S.B., Reis D.J., Cerebellar stimulation reduces inducible nitric oxide synthase expression and protects brain from ischemia, Am. J. Physiol., 1998, 274, 2035–2045
Galea E., Glickstein S.B., Feinstein D.L., Golanov E.V., Reis D.J., Stimulation of cerebellar fastigial nucleus inhibits interleukin-1[beta]-induced cerebrovascular inflammation, Am. J. Physiol., 1998, 275, 2053–2063
Rothwell N.J., Luheshi G.N., Interleukin 1 in the brain: Biology, pathology and therapeutic target, Trends Neurosci., 2000, 23, 618–625
del Zoppo G., Ginis I., Hallenbeck J.M., Iadecola C., Wang X., Feuerstein G.Z., Inflammation and stroke: Putative role for cytokines, adhesion molecules and iNOS in brain response to ischemia, Brain Pathol., 2000, 10, 95–112
Forster C., Clark H.B., Ross M.E., Iadecola C., Inducible nitric oxide synthase expression in human cerebral infarcts, ActaNeuropathol., 1999, 97, 215–220
Iadecola C., Zhang F., Xu S., Casey R., Ross M.E., Inducible nitric oxide synthase gene expression in brain following cerebral ischemia, J. Cereb. Blood Flow Metab., 1999, 15, 378–384
Jander S., Kraemer M., Schroeter M., Witte O.W., Stoll G., Lymphocytic infiltration and expression of intercellular adhesion molecule-1 in photochemically induced ischemia of the rat cortex, J. Cereb. Blood Flow Metab., 1995, 15, 42–51
Zhang R.L., Chopp M., Jiang N., Tangb W.X., Prostak J., Manning A.M., et al., Anti-intercellular adhesion molecule-1 antibody reduces ischemic cell damage after transient but not permanent middle cerebral artery occlusion in the Wistar rat, Stroke, 1995, 26, 1438–1442
Stroemer R.P., Rothwell N.J., Cortical protection by localized striatal injection of IL-1ra following cerebral ischemia in the rat, J. Cereb. Blood Flow Metab., 1997, 17, 597–604
Choi D.W., Ischemia-induced neuronal apoptosis, Curr. Opin. Neurobiol., 1996, 6, 667–672
Gillardon F., Klimaschewski L., Wickert H., Krajewski S., Reed J.C., Zimmermann M., Expression pattern of candidate cell death effector proteins Bax, Bcl-2, Bcl-X, and c-Jun in sensory and motor neurons following sciatic nerve transection in the rat, Brain Res., 1996, 739, 244–250
Guegan B., Sola C., Early and sequential recruitment of apoptotic effectors after focal permanent ischemia in mice, Brain Res., 2000, 856, 93–100
Vaux D. L., Korsmeyer S., Cell death in development, Cell, 1999, 96, 245–254
Earnshaw W.C., Martins L.M., Kaufmann S.H., Mammalian caspases: structure, activation, substrates, and functions during apoptosis, Annu. Rev. Biochem., 1999, 68, 383–424
Reed J., Mechanisms of apoptosis, Am. J. Pathol., 2000, 157, 1415–1430
Zhou P., Qian L., Glickstein S.B., Golanov E.V., Pickel V.M., Reis D.J., Electrical stimulation of cerebellar fastigial nucleus protects rat brain, in vitro, from staurosporine-induced apoptosis, J. Neurochem., 2001, 79, 328–338
Prehn J.H., Jordán J., Ghadge G.D., Preis E., Galindo M.F., Roos R.P., et al., Ca2+ and reactive oxygen species in staurosporine-induced neuronal apoptosis, J. Neurochem., 1997, 68, 1679–1685
Kruman I., Guo Q., Mattson M. P., Calcium and reactive oxygen species mediate staurosporine-induced mitochondrial dysfunction and apoptosis in PC12 cells, J. Neurosci. Res., 1998, 51, 293–308
Krajewski S., Krajewska M., Ellerby L.M., Welsh K., Xie Z., Deveraux Q.L., et al. Release of caspase-9 from mitochondria during neuronal apoptosis and cerebral ischemia, Proc. Natl. Acad. Sci. USA, 1999, 96, 5752–5757
Krohn A.J., Preis E., Prehn J.H., Staurosporine-induced apoptosis of cultured rat hippocampal neurons involves caspase-1-like proteases as upstream initiators and increased production of superoxide as a main downstream effector, J. Neurosci., 1998, 18, 8186–8197
Velier J.J., Ellison J.A., Kikly K.K., Spera P.A., Barone F.C., Feuerstein G.Z., Caspase-8 and caspase-3 are expressed by different populations of cortical neurons undergoing delayed cell death after focal stroke in the rat, J. Neurosci., 1999, 19, 5932–5941
Zhou P., Qian L., Zhou T., Iadecola C., Mitochondria are involved in the neurogenic neuroprotection conferred by stimulation of cerebellar fastigial nucleus, J. Neurochem., 2005, 95, 221–229
Antonsson B., Montessuit S., Sanchez B., Martinou J.C., Bax is present as a high molecular weight oligomer/complex in the mitochondrial membrane of apoptotic cells, J. Biol. Chem., 2001, 276, 615–623
Antonsson B., Montessuit S., Lauper S., Eskes R., Martinou J.C., Bax oligomerization is required for channel-forming activity in liposomes and to trigger cytochrome c release from mitochondria, Biochem. J., 2000, 345, 271–278
Newmeyer S.D., Ferguson-Miller D., Mitochondria: releasing power for life and unleashing the machineries of death, Cell, 2003, 112, 481–490
Silver I.A., Erecinska M., Intracellular and extracellular changes of [Ca2+]_in hypoxia and ischemia in rat brain in vivo, J. Gen. Physiol., 1990, 95, 837–866
Kristian T., Katsura K., Gido G., Siesjo B.K., The influence of pH on cellular calcium influx during ischemia, Brain Res., 1994, 641, 295–302
Schinder A.F., Olson E.C., Spitzer N.C., Montal M., Mitochondrial dysfunction is a primary event in glutamate neurotoxicity, J. Neurosci., 1996, 16, 6125–6133
Reynolds I.J., Mitochondrial membrane potential and the permeability transition in excitotoxicity, Ann. NY Acad. Sci., 1999, 893, 33–43
Kroemer G., Reed J., Mitochondrial control of cell death, Nat. Med., 2000, 6, 513–519
Pfeiffer D.R., Gudz T.I., Novgorodov S.A., Erdahl W.L., The peptide mastoparan is a potent facilitator of the mitochondrial permeability transition, J. Biol. Chem. 1995, 270, 4923–4932
Nicolay K., Laterveer F.D., van Heerde W.L., Effects of amphipathic peptides, including presequences, on the functional integrity of rat liver mitochondrial membranes, J. Bioenerg. Biomembr., 1994, 26, 327–334
Stein S.C., Levine J.M., Nagpal S., LeRoux P.D., Vasospasm as the sole cause of cerebral ischemia: how strong is the evidence?, Neurosurg. Focus, 2006, 21, 22–25
Feigin V.L., Rinkel G.J., Lawes C.M., Algra A., Bennett D.A., van Gijn J., et al., Risk factors for subarachnoid hemorrhage: an updated systematic review of epidemiological studies, Stroke, 2005, 36, 2773–2780
Lee K.H., Lukovits T., Friedman J.A., “Triple-H” therapy for cerebral vasospasm following subarachnoid hemorrhage, Neurocrit. Care, 2006, 4, 68–76
Prevedello D.M., Cordeiro J.G., de Morais A.L., Saucedo N.S.Jr., Chen I.B., Araújo J.C., Magnesium sulfate: role as possible attenuating factor in vasospasm morbidity, Surg. Neurol., 2006, 65, 1–20
Cecon A.D., Figueiredo E.G., Bor-Seng-Shu E., Scaff M., Teixeira M.J., Extremely delayed cerebral vasospasm after subarachnoid hemorrhage, Arq. Neuropsiquiatr., 2008, 66, 554–556
Fisher C.M., Kistler J.P., Davis J.M., Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by CT scanning, Neurosurgery, 1980, 6, 1–9
Hijdra A., Brouwers P.J., Vermeulen M., van Gijn J., Grading the amount of blood on computed tomograms after subarachnoid hemorrhage, Stroke, 1990, 21, 1156–1161
Claassen J., Bernardini G.L., Kreiter K., Bates J., Du Y.E., Copeland D., Effect of cisternal and ventricular blood on risk of delayed cerebral ischemia after subarachnoid hemorrhage. The Fisher Scale revisited, Stroke, 2001, 32, 2012–2020
Dupont S.A., Wijdicks E.F., Manno E.M., Lanzino G., Brown R.D.Jr., Rabinstein A.A., Timing of computed tomography and prediction of vasospasm after aneurysmal subarachnoid hemorrhage, Neurocrit. Care, 2009, 11, 71–75
Cameron T., Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20-year literature review, J. Neurosurg., 2004, 100, 254–267
de Jongtse M.J., Haaksma J., Hautvast R.W., Hillege H.L., Meyle P.W., Staal M.J., Effects of spinal cord stimulation on myocardial ischaemia during daily life in patients with severe coronary artery disease. A prospective ambulatory electrocardiographic study, Br. Heart J., 1994, 71, 413–418
Gersbach P., Hasdemir M.G., Stevens R.D., Nachbur B., Mahler F., Discriminative microcirculatory screening of patients with refractory limb ischaemia for dorsal column stimulation, Eur. J. Vasc. Endovasc. Surg., 1997, 13, 464–471
Hosobuchi Y., Electrical stimulation of cervical spinal cord increases cerebral blood flow in humans, Appl. Neurophysiol., 1985, 48, 327–376
Matsui T., Hosobuchi Y., The effects of cervical spinal cord stimulation (cSCS) on rCBF, Pacing Clin. Electrophysiol., 1989, 12, 830–835
Matsui T., Hosobuchi Y., The effects of the cervical spinal cord stimulation (CSCS) on experimental stroke, Pacing Clin. Electrophysiol., 1989, 12, 726–732
Visocchi M., Argiolas L., Meglio M., Cioni B., Dal Basso P., Rollo M., Cabezas D., Spinal cord stimulation and early experimental cerebral spasm: The functional monitoring and preventing effect, Acta Neurochir. (Wien), 2001, 143, 177–185
Shinonaga M., Takanashi Y., Vasodilating effect of spinal cord stimulation for cerebral vasospasm, Acta Neurochir. Suppl., 2001, 77, 229–230
Takanashi Y., Shinonaga M., Spinal cord stimulation for cerebral vasospasm as prophylaxis, Neurol. Med. Chir. (Tokyo), 2000, 40, 352–357
Visocchi M., Cioni B., Pentimalli L., Meglio M., Increase of cerebral blood flow and improvement of brain motor control following spinal cord stimulation in ischemic spastic hemiparesis, Stereotact. Funct. Neurosurg., 1994, 62, 103–107
Visocchi M., Giordano A., Calcagni M., Cioni B., Di Rocco F., Meglio M., Spinal cord stimulation and cerebral blood flow in stroke: personal experience, Stereotact. Funct. Neurosurg., 2001, 76, 262–268
Broseta J., García-March G., Sánchez-Ledesma M.J., Gonçalves J., Silva I., Barcia J.A., et al., High-cervical spinal cord electrical stimulation in brain low perfusion syndromes: Experimental basis and preliminary clinical report, Stereotact. Funct. Neurosurg., 1994, 62, 171–178
Sagher O., Huang D.L., Mechanisms of spinal cord stimulation in ischemia, Neurosurg. Focus, 2006, 21, 2
Göksel H.M., Karadag O.O., Turaçlar U., Taş F., Oztoprak I., Nitric oxide synthase inhibition attenuates vasoactive response to spinal cord stimulation in an experimental cerebral vasospasm model, Acta Neurochir. (Wien), 2001, 143, 383–391
Linderoth B., Gunasekera L., Meyerson B.A., Effects of sympathectomy on skin and muscle microcirculation during dorsal column stimulation: animal studies, Neurosurgery, 1991, 29, 874–879
Linderoth B., Gherardini G., Ren B., Lundeberg T., Preemptive spinal cord stimulation reduces ischemia in an animal model of vasospasm, Neurosurgery, 1995, 37, 266–272
Treggiari M.M., Romand J.A., Martin J.B., Reverdin A., Rüfenacht D.A., de Tribolet N., Cervical sympathetic block to reverse delayed ischemic neurological deficits after aneurysmal subarachnoid hemorrhage, Stroke, 2003, 34, 961–967
Patel S., Huang D.L., Sagher O., Evidence for a central pathway in the cerebrovascular effect of spinal stimulation, Neurosurgery, 2004, 55, 201–206
Reis D.J., Golanov E.V., Rugier A., Sun M.K., Sympatho-excitatory neurons of the rostral ventrolateral medulla are oxygen sensors and essential elements in the tonic and reflex control of the systemic and cerebral circulations, J. Hypertens., 1994, 12, 159–180
Sun M.K., Reis D.J., Cyanide excites medullary sympathoexcitatory neurons in rats, Am. J. Physiol., 1992, 262, 182–189
Golanov E.V., Reis D.J., Synchronization of EEG activity in cerebral cortex by stimulation of nucleus tractus solitarii (NTS) in rat: Coupling to cerebral blood flow and mediation by rostral ventrolateral medulla (RVL), Soc. Neurosci. Abstr., 1995, 21, 1669
Vahedi K., Vicaut E., Mateo J., Kurtz A., Orabi M., Guichard J.P., et al., Sequential-design multicenter randomized controlled trial of early decompressive craniectomy in malignant middle cerebral artery infarction (DECIMAL Trial), Stroke, 2007, 38, 2506–2517
The National Institute of Neurological Disorder, Tissue plasminogen activator for acute ischemic stroke, N. Engl. J Med., 1995, 333, 1581–1587
Molina C.A., Saver J.L., Extending reperfusion therapy for acute ischemic stroke: emerging pharmacological, mechanical, and imaging strategies, Stroke, 2005, 36, 2311–2320
Reeves M.J., Arora S., Broderick J.P., Acute stroke care in the US: results from the 4 pilot prototypes of the Paul Coverdell National Acute Stroke Registry, Stroke, 2005, 36, 1232–1240
Micieli G., Marcheselli S., Tosi P.A., Safety and efficacy of alteplase in the treatment of acute ischemic stroke, Vasc. Health Risk Manag., 2009, 5, 397–409
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Mandel, M., Fonoff, E.T., Bor-Seng-Shu, E. et al. Neurogenic neuroprotection: Future perspectives. Translat.Neurosci. 3, 399–412 (2012). https://doi.org/10.2478/s13380-012-0047-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s13380-012-0047-4