Abstract
Background and Objectives: The International Conference of Harmonisation (ICH) E14 guideline for thorough QT studies requires assessing the propensity of new non-antiarrhythmic drugs to affect cardiac repolarization. The present study investigates whether a composite ECG measure of T-wave morphology (Morphology Combination Score [MCS]) can be used together with the heart rate corrected QT interval (QTc) in a fully ICH E14-compliant thorough QT study to exclude clinically relevant repolarization effects of bilastine, a novel antihistamine.
Methods: Thirty participants in this crossover study were randomly assigned to receive placebo, moxifloxacin 400 mg, bilastine at therapeutic and supratherapeutic doses (20 and 100 mg) and bilastine 20 mg co-administered with ketoconazole 400 mg. Resting ECGs recorded at 12 nominal time points before and after treatments were used to determine Fridericia corrected QTc (QTcF) and MCS from the T-wave characteristics: asymmetry, flatness and notching.
Results: There were no effects of bilastine monotherapy (20 and 100 mg) on MCS or QTcF at those study times where the bilastine plasma concentrations were highest. MCS changes for bilastine monotherapy did not exceed the normal intrasubject variance of T-wave shapes for triplicate ECG recordings. Maximum QTcF prolongation for bilastine monotherapy was 5 ms or less: 3.8 ms (90% CI 0.3, 7.3 ms) for bilastine 20 mg and 5.0 ms (90% CI 2.0, 8.0 ms) for bilastine 100 mg. There were no indications of bilastine inducing larger repolarization effects on T-wave morphology as compared with the QTcF interval, as evidenced by the similarity of z-score equivalents for placebo-corrected changes in MCS and QTcF values.
Conclusion: This study shows that bilastine, at therapeutic and supratherapeutic dosages, does not induce any effects on T-wave morphology or QTcF. These results confirm the absence of an effect for bilastine on cardiac repolarization.
Similar content being viewed by others
Introduction
Bilastine, a novel, highly selective non-sedating antihistamine,[1,2] was developed by Faes Farma, S.A., Leioa, Spain, for the symptomatic treatment of allergic rhinoconjunctivitis and urticaria. In clinical trials performed in patients with allergic rhinoconjunctivitis, bilastine 20 mg once daily was effective in relieving symptoms such as sneezing, nasal discharge, nasal congestion and ocular redness, and exhibited a similar or better safety profile than desloratadine, cetirizine and fexofenadine.[3–5] In patients with chronic urticaria, bilastine has been effective in reducing the number and size of wheals as well as relieving the discomfort and intensity of itching, and its efficacy was comparable to that of levocetirizine with a safety profile similar to placebo.[6,7]
The effects of bilastine on corrected QT (QTc) prolongation have recently been investigated in a fully International Conference on Harmonisation (ICH) E14-compliant thorough QTc study.[8] It was concluded that bilastine yields QTc interval changes well below the limits for regulatory concern. The ICH E14 guidance also calls for morphological analyses of ECG waveforms emphasizing that changes in the T-U wave complex should be studied in addition to the QT interval.[9] This aspect of repolarization has not been systematically addressed for bilastine. What is new in this study is our approach to analysing T-wave morphology within the framework of a thorough QT study, which in our opinion is a neglected aspect in the risk assessment for many published thorough QTc studies.
Drug-induced QT-interval prolongation and cardiac risk appear to result from inhibition of the rapidly activating delayed rectifier potassium current, IKr,[10,11] which is encoded by the KCNH2 (human ether-à-go-go [hERG]) gene. Bilastine has shown very low potency for inhibiting the IKr channel[7,12] and would therefore not be expected to have a significant impact on QT-interval prolongation. On the other hand, the QT interval appears to be an imperfect biomarker due to the limited correlation between QT prolongation and the risk of torsades de pointes (TdP).[13] For example, Pratt et al.[14] demonstrated that terfenadine at therapeutic doses induced only a small QT prolongation in healthy subjects (6 ms) but the administration of the drug in patients was associated with several incidents of TdP, whereas amiodarone, a widely used antiarrhythmic, is known to markedly increase the QT interval while rarely causing TdP.[15] Additionally, the QT interval is sensitive to heart rate (HR) and needs to be corrected, which may induce a false QT/QTc signal if the drug being studied is known to markedly change the HR.[16,17] For example, it has been demonstrated that the rate of positive QT studies can be up to 60%, despite no drug effect, and as high as 80% with clinically non-meaningful QTc prolongations (5 ms).[18] Drugs that prolong the QTc interval to the same extent sometimes result in differing incidences of TdP. Moreover, drugs that are associated with a similar incidence of TdP do not necessarily prolong the QTc interval to equivalent extents.[13]
As a consequence of such limitations, several alternative biomarkers focusing on T-wave morphology instead of QT/QTc duration are being developed. Previous work has shown that T-wave morphology biomarkers can be used to discriminate between healthy subjects and long QT syndrome patients[19–21] and to detect drug-induced repolarization changes.[22,23] Logistic regression modeling has shown that T-wave morphology can improve the characterization of patients with a history of TdP[23] and the detection of impaired adaptation of the T-wave amplitude to changes in HR has been useful in identifying drug-induced repolarization changes.[24] In the general population, T-wave morphology parameters, but not the HR-corrected QT interval, provided prognostic information on mortality independent of other clinical risk factors.[25]
It has also been demonstrated that a set of advanced morphology descriptors (asymmetry, flatness, notch and the Morphology Combination Score [MCS]) could be more sensitive to certain drug-induced changes than the QT/QTc measurements[26–29] and that these morphology descriptors are independent of HR.[30] Importantly, using MCS, it was demonstrated that marked drug-induced repolarization abnormalities may be identified in the absence of QTc interval prolongation or even shortening.[27] Recently it was shown that drugs with similar QTc effects may be associated with different T-wave morphology changes, and that their potential to induce TdP may also vary.[29]
Collectively, such findings indicate that careful assessment of T-wave morphology changes could be potentially valuable as an adjunct to QTc analysis with respect to assessing drug effects on cardiac repolarization. They also highlight the obvious challenges in characterizing drug-induced repolarization changes because although the safety profile of a drug is dependent on QTc interval prolongation, this is not the only relevant factor when evaluating risk of arrhythmia.
This study of bilastine-induced T-wave morphology changes was therefore performed to highlight repolarization aspects of the bilastine thorough QT study,[8] which have not previously been reported.
We hypothesize the combined absence of drug effects on T-wave morphology and the QTc interval to be a characteristic of safe drugs with low proarrhythmic potential and propose here an outline of how MCS may be incorporated into the study design of thorough QT studies. The aim is to determine whether such analysis could be used together with the QTc to exclude clinically relevant repolarization effects of bilastine.
In addition, because the proarrhythmic effects of the antihistamine terfenadine were found to be enhanced after coadministration of a cytochrome P450 (CYP) and P-glycoprotein (P-gp) inhibitor such as ketoconazole,[31] the effects of co-administration of bilastine at a therapeutic dose and ketoconazole are also investigated. Although both in vivo and in vitro preclinical studies and clinical studies (human mass balance and metabolic profiling) have shown that bilastine undergoes negligible metabolic transformation as opposed to terfenadine (parent drug of fexofenadine), it has also been shown that bilastine is a P-gp substrate and thus has drug-drug interactions with P-gp inhibitors such as ketoconazole, erythromycin and diltiazem.[12] Co-administration of bilastine with P-gp inhibitors may thus increase plasma levels of bilastine and therefore increase the risk of adverse effects of bilastine.
Methods
Study Design
This retrospective study of bilastine-induced T-wave morphology was designed to meet ICH E14 standards for QT-interval assessment in thorough QT studies as a randomized, double-blind, multiple-dose, triple-dummy, one-factor, crossover study at five levels.[8] Study participants were randomly assigned to receive a single daily dose of placebo, active control (moxifloxacin 400 mg), bilastine at therapeutic and supratherapeutic doses (bilastine 20 mg and 100 mg) and bilastine 20 mg co-administered with ketoconazole 400 mg. Treatments were preceded by a full baseline day where no drug was given (day −1). All drugs were given for 4 consecutive days (days 1 to 4) with the exception of moxifloxacin, which was given over 3 days (days 2 to 4). There was a minimum 7-day washout between the day 4 dose of a given period and the day 1 dose of the next period.
The study was conducted at MDS Pharma Services (Phoenix, AZ, USA). Further protocol details can be found in the thorough QT study of bilastine.[8] Ethics approval was obtained from the MDS Pharma Services investigational review board, an independent committee, and the study was performed in compliance with current Good Clinical Practice and the Declaration of Helsinki. All subjects gave written informed consent.
Study Population
The study enrolled 30 healthy volunteers (16 males, 14 females) aged from 20 to 44 years (table I). As described previously,[8] eligible subjects had a normal 12-lead ECG at screening and again at check-in with normal sinus rhythm (50 to 100 beats/min), a Bazett QTc <430 ms for males or <450 ms for females, a QRS interval <110 ms, and a PR interval <220 ms, and were taking no medication that would interfere with the study or compromise subject safety. Participants were excluded from the study if any of the following criteria were met: history or presence of ECG abnormalities or history of prolonged QT interval; evidence of clinically significant disease; alcoholism; drug abuse; use of medication within 14 days of first drug administration; pregnant or lactating women.
ECG Acquisition
Standard 12-lead ECGs were recorded at time points corresponding to 0, 0.5, 1, 1.5, 2, 3, 4, 6, 8, 10, 12 and 16 hours after dosing on day 4 and at similar time points on the baseline day (day −1).[8] On days 5 to 7, ECGs were recorded at 24, 36, 48 and 72 hours after dosing. At each nominal time point, triplicate 10-second ECGs were obtained within a 2-minute window using a sample rate of 500 Hz (GE Healthcare Mac 1200, Milwaukee, WI, USA). Subjects were at rest in a fully supine position for at least 5 minutes before recordings were initiated. On day 4, blood samples were drawn immediately following the ECG sampling. Plasma concentrations and ECG recordings on day 4 constitute the data analysed in the present study.
ECG Processing
Each extracted ECG segment was used to form a representative median beat in the 8 recorded leads (I, II, V1-V6) using MUSE/Interval Editor software (GE Healthcare). T-wave morphology analysis was performed on a principal component beat that was derived from median beats in the recorded leads.[26] A low-pass Kaiser Window FIR filter with a cutoff frequency of 20 Hz was used on the first principal component beat. This filtered beat was used for analysis of repolarization morphology.
In the original thorough QT study all ECG intervals were measured semi-automatically with adjustments made by cardiologists.[8] In this study fiducial point detection and QT-interval measurements were made automatically using version 21 of the 12SL algorithm (12SL, GE Healthcare).[32] The 12SL algorithm annotates fiducial points on the superimposed representative PQRST complexes from all 12 leads. The algorithm excludes discrete U-waves occurring after the T waves return to baseline, whereas complex multiphasic T waves and T-U complexes are included. QT intervals were corrected for HR with Fridericia’s equation: QTcF = QT/RR1/3.
T-wave Morphology Measures
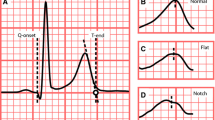
A morphology combination score (MCS) based on asymmetry, flatness and notching was used to identify drug-induced shape changes of the T-wave. These T-wave characteristics have been shown to identify abnormal repolarization in patients with type 2 congenital long QT syndrome (LQT2),[26] to be more sensitive than QTcF to repolarization changes induced by various drugs,[26–29] and to be heart-rate independent.[30] The linear combination of variables comprising MCS (equation 1) effectively gives each variable equal importance in the composite score:
Asymmetry
Asymmetry was defined as the average squared difference between the slopes (first derivatives) of the ascending and descending parts of the T wave.[26–29]
Notches
A curvature signal was calculated from the first and second derivatives of T-waves and used to identify the presence or absence of a notch.[26–29] The magnitude of a notch was measured on a unit amplitude T-wave and assigned to one of three categories as was suggested by Lupoglazoff et al.:[33] no deflection = 0, moderate notch (perceptible bulge) = 0.5, and pronounced notch (distinct protuberance above the apex) = 1.0.
Flatness
Flatness was calculated as a modified version of the standard kurtosis measure, which is used in statistics to describe the peakedness of a probability density function.[26–29]
Statistical Analysis
All statistical analyses were performed using Matlab version 7.11, R2010b (Mathworks Inc, Natick, MA, USA). Paired Student’s t-tests for time-matched changes in the mean for QTcF and MCS were applied with and without placebo adjustment and p < 0.05 was regarded as significant. The baseline value for each post-dose assessment was the time-matched assessment for off-treatment. For all time points the median value of triplicates was used. QTcF and MCS measurements were converted to z-score equivalents by subtraction of the mean and subsequent division by the standard deviation (SD) of placebo values at baseline. z-Score equivalents were used to enable direct comparison of QTcF and MCS on a similar scale. Boundaries representing reliable T-wave morphology changes for individual ECG recordings were determined from the average within-subject standard deviation of triplicate MCS recordings in the placebo group on day −1 and day 4. The corresponding within-subject standard error for triplicate MCS recordings was used to determine boundaries for reliable changes in the mean MCS.
Results
Subject Disposition
Thirty healthy subjects (16 males, 14 females) were enrolled in the study. Two female subjects were discontinued from the study by the investigator due to laboratory results outside the normal range (decreased haemoglobin). The 28 subjects who completed the study had an average age of 32 years (range 20–44 years).
Clinical Safety Assessment
The most commonly observed adverse reactions (>10% of subjects) were: constipation and mild diarrhoea, six subjects each (20%); mild headache, mild dizziness and mild pruritus, five subjects each (17%); mild dyspepsia, four subjects (13%); mild flatulence, mild erythema, mild maculo-papular rash, mild upper abdominal pain and mild eye pain, three subjects each (10%). All remaining treatment-emergent adverse events (TEAEs) were reported by two or fewer subjects each (≤7%). No serious adverse events were reported and the number of subjects reporting TEAEs was evenly distributed between bilastine and placebo treatments: nine subjects (bilastine 20 mg), ten subjects (bilastine 100 mg), ten subjects (bilastine 20 mg + ketoconazole 400 mg) and eight subjects (placebo). There were 16 subjects receiving moxifloxacin who reported at least one TEAE. Administration of bilastine alone or in combination with ketoconazole appeared to be safe and well tolerated.
Placebo-Unadjusted Changes from Baseline
Relative to baseline, bilastine at therapeutic and supratherapeutic doses (20 mg and 100 mg) did not prolong the QT interval or affect T-wave morphology at any time point after administration. Peak changes from baseline for the mean QTcF without placebo adjustment (ΔQTcF) were less than 1 ms for both 20 mg and 100 mg doses of bilastine, figure 1. Neither did the placebo-unadjusted peak changes from baseline for T-wave morphology (ΔMCS) reach significance for the 20 mg dose (95% CI −0.036, 0.034) or the 100 mg dose (95% CI −0.004, 0.061). For the bilastine 20 mg dose given in combination with ketoconazole 400 mg, ΔQTcF and ΔMCS both reached significance at multiple time points. This was similar for the moxifloxacin 400 mg dose.
Differences between ΔMCS and ΔQTcF
The overall effects of bilastine on T-wave morphology and QTcF were small and comparable in terms of z-scores (figure 2). Following a bilastine 20 mg dose, the mean values of ΔMCS and ΔQTcF (0–16 h post-dose) were shifted by the same number of standard deviations (SDs) relative to placebo: 0.02 SD for ΔMCS and 0.11 SD for ΔQTcF (non-significant difference, p = 0.23). Nor was there any statistically significant difference between the shifting of mean values relative to placebo, for ΔMCS and ΔQTcF for the supratherapeutic bilastine 100 mg dose (p = 0.15) or for the combination therapy with bilastine 20 mg and ketoconazole 400 mg (p = 0.41). Moxifloxacin had a larger overall effect on ΔQTcF compared with ΔMCS: 1.75 SD for ΔQTcF versus 1.11 SD for ΔMCS (p < 0.01).
Changes from baseline (0–16 h post-dose) for T-wave morphology (Morphology Combination Score [MCS]) and Fridericia-corrected QT interval (QTcF). Delta values (Δ) indicate changes from baseline. Double delta values (ΔΔ) and standard deviations (SDs) indicate changes from baseline relative to placebo.
Maximum Effects at Peak Drug Concentrations
There were no effects of bilastine monotherapy (20 mg and 100 mg) on T-wave morphology or QTcF at those study times where the bilastine plasma concentrations were highest (1 hour post-dose) [figure 3]. Moreover, at this time point, no difference could be demonstrated between bilastine 20 mg and 100 mg in terms of ΔΔMCS (95% CI −0.04, 0.06; p = 0.69) or ΔΔQTcF (95% CI −4.2, 9.3 ms; p = 0.45) despite a 7-fold higher plasma concentration for the supratherapeutic 100 mg bilastine dose. The exposure to bilastine for 100 mg monotherapy was three times above that observed following administration of the combination therapy (bilastine 20 mg + ketoconazole 400 mg) [figure 3]. Yet, the impact on T-wave morphology was lower for bilastine 100 mg than for combination therapy (ΔΔMCS 95% CI −0.13, −0.02; p < 0.01) and the QTcF changes for bilastine 100 mg did not exceed those observed for combination therapy (ΔΔQTcF 95% CI −7.3, 1.8; p = 0.22).
Placebo-adjusted changes in T-wave morphology (Morphology Combination Score [MCS]) and Fridericia-corrected QT interval (QTcF) in relation to plasma concentration of bilastine. I-bars indicate two-sided 90% confidence intervals for the mean. Double delta values (ΔΔ) indicate changes from baseline relative to placebo.
Maximum Placebo-Adjusted Effects
The effects of bilastine monotherapy (20 mg and 100 mg) on ΔΔQTcF were 5 ms or less (table II). The upper bound of the two-sided 90% CI for ΔΔQTcF excluded 10 ms at all study times. T-wave morphology changes as assessed by ΔΔMCS did not exceed the normal variance for triplicate recordings. Neither did the QT-interval changes as assessed by ΔΔQTcF. The average within-subject standard error for triplicate MCS recordings in the placebo group was 0.011 at baseline (day −1) and 0.009 on the first study day post-dose (day 4). A reliable increase for ΔΔMCS would therefore be expected to exceed 1.96 × (0.0112 + 0.0092)0.5 = 0.03. However, the largest ΔΔMCS did not exceed this threshold for bilastine 20 mg (95% CI −0.13, 0.06; p = 0.65) or 100 mg (95% CI 0.01, 0.10; p = 0.18). Neither did the maximum ΔΔQTcF values exceed the reliable change threshold of 1.96 × (1.52 + 1.62)0.5 = 4.3 ms for bilastine 20 mg (95% CI −0.4, 8.0; p = 0.8) or 100 mg (95% CI 1.4, 8.6; p = 0.69).
The maximum effects of combination therapy (bilastine 20 mg + ketoconazole 400 mg) on ΔΔMCS and ΔΔQTcF were more than twice the effects of bilastine 100 mg monotherapy, even though the bilastine plasma concentrations were two to three times higher for the monotherapy. There were no indications of bilastine inducing larger repolarization effects on T-wave morphology as compared with the QTcF interval, as evidenced by the similarity of z-score equivalents for ΔΔMCS and ΔΔQTcF (table II).
Effects of Sex on ΔΔMCS and ΔΔQTcF
No sex differences were observed for T-wave morphology following bilastine treatment. Bilastine 20 mg: ΔΔMCS = 0.03 (female) versus 0.02 (male), p = 0.85; bilastine 100 mg: ΔΔMCS = 0.08 (female) versus 0.04 (male), p = 0.32; and bilastine 20 mg + ketoconazole 400 mg: ΔΔMCS = 0.16 (female) versus 0.09 (male), p = 0.09.
QTcF prolongations were also similar for males and females. Bilastine 20 mg: ΔΔQTcF = 0.7 ms (female) versus 5.9 ms (male), p = 0.22; bilastine 100 mg: ΔΔQTcF = 6.2 ms (female) versus 4.2 ms (male), p = 0.59; and bilastine 20 mg + ketoconazole 400 mg: ΔΔQTcF = 12.4 ms (female) versus 13.6 ms (male), p = 0.82.
Categorical T-wave Morphology Effects
Categorical analyses did not indicate an increased likelihood of causing noteworthy T-wave morphology changes for bilastine monotherapy, compared with placebo. For individual recordings, a reliable change from baseline would have to exceed the within-subject standard deviation for triplicate MCS recordings in the placebo group on day −1 and day 4 (0.041 and 0.034, respectively) or ±1.96 × (0.0412 + 0.0342)0.5 = ±0.104. The upper and lower limits of these boundaries are shown in figure 4. Measurements displacing more than ±0.104 from the identity line are reliable changes from baseline and any displacement of this magnitude to the left of the identity line would indicate a treatment-related increase in MCS. There was no statistical difference compared with placebo in the distribution of MCS scores inside or outside of the boundaries of reliable change for bilastine 20 mg (chi-squared [χ2] = 1.33; p = 0.25) or bilastine 100 mg (χ2 = 0.01; p = 0.92). For bilastine 20 mg given in combination with ketoconazole 400 mg, the number of recordings that were displaced more than 0.104 from the identity line was higher compared with placebo (χ2 = 11.28; p < 0.01).
Distribution of T-wave morphology scores (Morphology Combination Score [MCS]) along the identity line on day −1 versus day 4: bilastine 20 mg and placebo (a); bilastine 100 mg and placebo (b); bilastine 20 mg + ketoconazole 400 mg and placebo (c). Threshold for reliable change in MCS from the identity line (±0.104, dashed lines) is based on the calculation of the within-subject standard deviation for triplicate MCS recordings in the placebo group. MCS measurements outside these boundaries are regarded as reliable changes in MCS.
Discussion
This thorough QT study with added T-wave morphology analysis has shown that bilastine at therapeutic and supratherapeutic dosages does not induce any effects on the QTcF interval or on the T-wave morphology score (MCS) that would indicate proarrhythmic potential when administered to healthy volunteers.
The finding of an apparently favourable cardiac safety profile for bilastine in the present study is in accordance with the findings from the original thorough QT study[8] and with previous phase I studies where bilastine was administered as single or multiple doses ranging from 2.5 mg to 220 mg or in combination with a metabolic and P-gp inhibitor (erythromycin, ketoconazole or diltiazem).[12] All previous analyses of QTc have failed to demonstrate an effect of bilastine on repolarization that would warrant regulatory concern. This study was designed as a thorough QT study in accordance with the ICH E14 guidance for industry as a randomized, crossover group, multiple-dose study including the active reference moxifloxacin in its own arm to confirm assay sensitivity at the same sampling times as placebo and bilastine. Although bilastine is not metabolized and has no CYP interaction, bilastine is a substrate for P-gp inhibitors, and when co-administered with for example ketoconazole, the bioavailability of bilastine can increase. Co-administration of ketoconazole and terfenadine has been shown to increase the risk of TdP, so a secondary objective of the study was to evaluate the repolarization effects of bilastine co-administered with the P-gp inhibitor, ketoconazole. Essentially, this is the classical framework for evaluating the potential of a drug to prolong the QT interval. In the present study QTc intervals were complemented by quantitative measurements of T-wave morphology to provide an additional and more rigorous assessment of the potential for bilastine to affect ventricular repolarization. Findings from this trial suggest that bilastine is not associated with significant cardiac electrophysiological effects when administered to healthy individuals.
The upper two-sided 90% CI for the maximum time-matched and placebo-corrected mean effect on QTcF did not exceed 10 ms at any timepoint following bilastine monotherapy. As a result, this study meets the definition of a negative QT study in which no clinically meaningful increases in QTcF were seen when bilastine was given as the therapeutic dose (20 mg) or as five times the therapeutic dose (100 mg). The lack of notable QTcF prolongation at the higher dose of bilastine and a lack of QTc prolongation observed in the original thorough QT study[8] supports the expectation that bilastine will not be associated with clinically relevant QTc interval prolongation.
The MCS data in this study also showed that bilastine 20 mg and 100 mg had negligible effects on T-wave morphology. The placebo-adjusted morphology changes for both doses of bilastine did not exceed the normal variance of T-wave shapes for triplicate ECGs taken less than 2 minutes apart. Thus, relative to an individual’s short-term fluctuation there appear to be no effects of clinical concern for bilastine on T-wave morphology. Moreover, because bilastine did not raise regulatory flags based on ΔΔQTcF, the similarity of z-score equivalents for ΔΔMCS and ΔΔQTcF presents strong evidence that bilastine monotherapy has minimal effects on cardiac repolarization.
Ketoconazole was found to potentiate the effects of terfenadine by pharmacokinetic actions to elevate plasma levels and by pharmacodynamic action on hERG currents.[34] Ketoconazole (400 mg) by itself has also been reported to increase the mean QTc from baseline by 7.0 ms and 7.5 ms.[35] Such prolongation closely corresponds to the added QTcF effect of ketoconazole observed in the present study when the drug was given in combination with bilastine 20 mg. The efflux transporter interaction by P-gp inhibition of ketoconazole increased the bilastine bioavailability for combination therapy in this study to the equivalent of increasing bilastine dose up to three times. However, our results showed that, even at five times the therapeutic dose, bilastine monotherapy did not induce repolarization effects above those observed for a therapeutic dose. At the same time, bilastine plasma concentrations for the combination therapy of bilastine 20 mg and ketoconazole 400 mg were lower at all nominal time points, compared with the concentrations for bilastine 100 mg alone. Yet the repolarization effects of combination therapy were larger. In the recently performed thorough QT study of bilastine,[8] a pharmacokinetic/pharmacodynamic relationship showed no correlation between bilastine plasma concentration and QTc intervals and it was also found that QTc prolongation for combination therapy correlated with the maximum plasma concentration (Cmax) for ketoconazole, but not bilastine. Together these results suggest that the repolarization effects of combination therapy in this study are almost exclusively related to the repolarization effects of ketoconazole and not to those of bilastine.
Regulatory guidance notes for drug trials mandate an integrated risk assessment. This includes a full description of all ECG changes, including T-wave morphology. However, because there is no standard approach to assessing abnormal T-wave morphology on the ECG, such assessment is left to the reading cardiologist. For most studies, therefore, the interpretation and reporting of drug-induced changes in the morphology of the T wave is subjective and unsystematic. In contrast, the MCS approach to analysing T-wave morphology offers the advantages of being systematic and repeatable while at the same time enabling a thorough statistical analysis of T-wave morphology changes. Computerized analysis of the T-wave morphology of ECG tracings may also provide an opportunity to identify effects on cardiac repolarization that cannot be described by the QT interval alone.
Different numerical quantifiers of the beat-to-beat QT variability have also been proposed as adjuncts to the QT interval, and perhaps most frequently the QT variability index (QTvi) has been used.[36] Using this index, it was found that patients with the LQT2 phenotype exhibit greater temporal QT variability than controls and that patients with an IKs mutation and a prolonged QT interval may have a normal QT variability index.[37] Such findings support the conclusion that an increased variability of repolarization may also provide information complementary to the QT interval about the degree to which ion channel function is disrupted. There are now many studies that have shown the QT variability to be a valuable predictor of cardiac mortality and it is therefore likely that the QTvi may also play a role in the assessment of repolarization changes in drug trials in the future.
Drugs that prolong the QTc interval to clinically relevant magnitudes are likely to be torsadogenic if they also produce relevant changes in the morphology of repolarization waveforms. The antiarrhythmic class III drug d,l-sotalol has a reported incidence of causing proarrhythmia in 1.8% of patients when dosages up to 320 mg/day are used.[38,39] This risk profile is indicated not only by the QTc prolonging property of d,l-sotalol, but even more so by the T-wave characteristics represented by MCS.[26,29] The antipsychotic drug sertindole is associated with a five-fold greater risk of sudden cardiac death compared with risperidone.[40] Sertindole prolongs QTcF to over 500 ms in 1.9% of patients (20 mg daily dose)[40] but the proarrhythmic potential of the drug is more clearly revealed by distinct changes in the T-wave characteristics comprising MCS.[27] For compounds with less pronounced effects on repolarization such as those associated with Lu 35-138, an investigational IKr-inhibiting antipsychotic compound that was terminated during development, the MCS measure of T-wave morphology was used to support the evidence of abnormal repolarization, which was indicated by prolongation of the QTc interval.[28] Collectively such findings suggest that abnormal repolarization is reflected in the shape of the electrocardiographic T-wave. On the other hand, this study has shown that IKr inhibition does necessarily translate into marked changes in T-wave morphology that could potentially present an obstacle for the further development and approval process of a drug. Moreover, the QTc interval produces both false-positive and false-negative results, and a careful assessment of MCS could therefore be used to complement QTc interval measurements for the assessment of a drug’s potential to affect cardiac repolarization. The MCS offers a predefined quantitative assessment of T-wave morphology using the same 10-second ECG segment (XML file) that is extracted for the processing of QTc intervals. Therefore, MCS can easily be added as an additional module in the already existing software systems used by many central ECG labs.
Conclusion
In this study it was found that bilastine monotherapy is devoid of any effects of clinical concern on the QTc interval and the composite score of asymmetry, notches and flatness of the T-wave (MCS). It was further found that the effects of bilastine on cardiac repolarization are unlikely to increase when given in combination with a metabolic and P-gp inhibitor such as ketoconazole. We believe this concurrent analysis of QTc and T-wave morphology has general validity for the safety evaluation of drugs and the risk of bilastine-induced TdP is therefore expected to be minimal.
References
Corcóstegui R, Labeaga L, Innerárity A, et al. Preclinical pharmacology of bilastine, a new selective histamine H1 receptor antagonist: receptor selectivity and in vitro antihistaminic activity. Drugs R D 2005; 6(6): 371–84
García-Gea C, Martínez-Colomer J, Antonijoan RM, et al. Comparison of peripheral and central effects of single and repeated oral dose administrations of bilastine, a new H1 antihistamine: a dose-range study in healthy volunteers with hydroxyzine and placebo as control treatments. J Clin Psychopharmacol 2008; 28(6): 675–85
Horak F, Zieglmayer P, Zieglmayer R, et al. The effects of bilastine compared to cetirizine, fexofenadine, and placebo on allergen-induced nasal and occular symptoms in patients exposed to aeroallergen in the Vienne Challenge Chamber. Inflamm Res 2010; 59(5): 391–8
Kuna P, Bachert C, Nowacki Z, et al. Efficacy and safety of bilastine 20mg compared with cetirizine 10mg and placebo for the symptomatic treatment of seasonal allergic rhinitis: a randomized, double-blind, parallel-group study. Clin Exp Allergy 2009; 39(9): 1338–47
Bachert C, Kuna P, Sanquer F, et al. Comparison of the effacacy and safety of bilastine 20 mg vs desloratadine 5 mg in seasonal allergic rhinitis patients. Allergy 2009; 64(1): 158–65
Zuberbier T, Oanta A, Bogacka E, et al. Comparison of the effacacy and safety of bilastine 20mg vs levocetirizine 5mg for the treatment of chronic idiopathic urticaria: a multicentre, double-blind, randomized, placebo-controlled study. Allergy 2010; 65(4): 516–28
Bachert C, Kuna P, Zuberbier T. Bilastine in allergic rhinoconjunctivitis and urticaria. Allergy 2010; 65Suppl. 93: 1–13
Tyl B, Kabbaj M, Azzam S, et al. Lack of significant effect of bilastine administered alone and with ketoconazole on ventricular repolarisation: results of a thorough QT study (TQTS) with QT-concentration analysis. J Clin Pharmacol 2011. Epub ahead of print
Food and Drug Administration (FDA). Guidance for Industry: E14 clinical evaluation of QT/QTc interval prolongation and proarrhythmic potential for non-antiarrhythmic drugs. October 2005 [online]. Available from URL: http://www.fda.gov/downloads/RegulatoryInformation/Guidances/UCM129357.pdf [Accessed 2012 Feb 1]
Haverkamp W, Breithart G, Camm AJ, et al. The potential for QT prolongation and proarrhythmia by non-antiarrhythmic drugs: clinical and regulatory implications. Eur Heart J 2000; 21(15); 1216–31
Hoffmann P, Warner B. Are hERG channel inhibition and QT interval prolongation all there is in drug-induced torsadogenesis? A review of emerging trends. J Pharmacol Toxicol Methods 2006; 53(2): 87–105
Faes Farma S.A. Bilastine Summary. Data on file [online]. Available from URL: http://www.faes.es [Accessed 2011 Jan 10]
Roden DM. Drug-induced prolongation of the QT interval. N Engl J Med 2004; 350(10): 1013–22
Pratt CM, Ruberg S, Morganroth J, et al. Dose-response relation between terfenadine (Seldane) and the QTc interval on the scalar electrocardiogram: distinguishing a drug effect from spontaneous variability. Am Heart J 1996; 131(3): 472–80
Yoshida H, Sugiyama A, Satoh Y, et al. Comparison of the in vivo electrophysiological and proarrhythmic effects of amiodarone with those of a selective class III drug, sematilide, using a canine chronic atrioventricular block model. Circ J 2002; 66(8): 758–62
Malik M. Problems of heart rate correction in assessment of drug-induced QT interval prolongation. J Cardiovasc Electrophysiol 2001; 12(4): 411–20
Desai M, Li L, Desta Z, et al. Variability of heart rate correction methods for the QT interval. Br J Clin Pharmacol 2003; 55(6): 511–7
Hutmacher MM, Chapel S, Agin MA, et al. Performance characteristics for some typical QT study designs under the ICH E-14 guidance. J Clin Pharmacol 2008; 48(2): 215–24
Struijk JJ, Kanters JK, Andersen MP, et al. Classification of the long-QT syndrome based on discriminant analysis of T-wave morphology. Med Biol Eng Comput 2006; 44(7): 543–9
Couderc JP, McNitt S, Xia J, et al. Repolarization morphology in adult LQT2 carriers with borderline prolonged QTc interval. Heart Rhythm 2006; 3(12): 1460–6
Kanters JK, Graff C, Andersen MP, et al. Long QT syndrome genotyping by electrocardiography: fact, fiction, or something in between? J Electrocardiol 2006; 39Suppl. 4: S1 19–22
Couderc JP, McNitt S, Hyrien O, et al. Improving the detection of subtle IKr-inhibition: assessing electrocardiographic abnormalities of repolarization induced by moxifloxacin. Drug Saf 2008; 31(3): 249–60
Couderc JP, Kaab S, Hinterseer M, et al. Baseline values and sotalol-induced changes of ventricular repolarization duration, heterogeneity, and instability in patients with a history of drug-induced torsades de pointes. J Clin Pharmacol 2009; 49(1): 6–16
Couderc JP, Vaglio M, Xia X, et al. Impaired T-amplitude adaptation to heart rate characterizes IKr inhibition in the congenital and acquired forms of the long QT syndrome. J Cardiovasc Electrophysiol 2007; 18(12): 1299–305
Porthan K, Viitasalo M, Jula A, et al. Predictive value of electrocardiographic QT interval and T-wave morphology parameters for all-cause and cardiovascular mortality in a general population. Heart Rhythm 2009; 6(8): 1202–8
Graff C, Andersen MP, Xue JQ, et al. Identifying drug-induced repolarization abnormalities from distinct ECG patterns in congenital long QT syndrome: a study of sotalol effects on T-wave morphology. Drug Saf 2009; 32(7): 599–611
Nielsen J, Graff C, Hardahl T, et al. Sertindole causes distinct electrocardiographic T-wave morphology changes. Eur Neuropsychopharmacol 2009; 19(10): 702–7
Graff C, Matz J, Christensen EB, et al. Quantitative analysis of T-wave morphology increases confidence in drug-induced cardiac repolarization abnormalities: evidence from the investigational IKr inhibitor Lu 35-138. J Clin Pharmacol 2009; 49(11): 1331–42
Graff C, Struijk JJ, Matz J, et al. Covariate analysis of QTc and T-wave morphology: new possibilities in the evaluation of drugs that affect cardiac repolarization. Clin Pharmacol Ther 2010; 88(1): 88–94
Andersen MP, Xue JQ, Graff C, et al. New descriptors of T-wave morphology are independent of heart rate. J Electrocardiol 2008; 41(6): 557–61
Pratt CM, Hertz RP, Ellis BE, et al. Risk of developing life-threatening ventricular arrhythmia associated with terfenadine in comparison with over-the-counter antihistamines, ibuprofen and clemastine. Am J Cardiol 1994; 73(5): 346–52
Xue JQ. Robust QT interval estimation: from algorithm to validation. Ann Noninvasive Electrocardiol 2009; 14Suppl. 1: S35–41
Lupoglazoff JM, Denjoy I, Berthet M, et al. Notched T waves on Holter recordings enhance detection of patients with LQT2 (HERG) mutations. Circulation 2001; 103(8): 1095–101
Dumaine R, Roy ML, Brown AM. Blockade of HERG and Kv1.5 by ketoconazole. J Pharmacol Exp Ther 1998; 286(2): 727–35
Chaikin P, Gillen MS, Malik M, et al. Co-administration of ketoconazole with H1-antagonists ebastine and loratadine in healthy subjects: pharmacokinetic and pharmacodynamic effects. Br J Clin Pharmacol 2005; 59(3): 346–54
Berger RD, Kasper EK, Baughman KL, et al. Beat-to-beat QT interval variability: novel evidence for repoalrization lability in ischemic and nonischemic dilated cardiomyopathy. Circulation 1997; 96(5): 1557–65
Bilchick K, Viitasalo M, Oikarinen L, et al. Temporal repolarization lability differences among genotyped patients with the long QT syndrome. Am J Cardiol 2004; 94(10): 1312–6
Haverkamp W, Martinez-Rubio A, Hief C, et al. Efficacy and safety of d,l-sotalol in patients with ventricular tachycardia and in survivors of cardiac arrest. J Am Coll Cardiol 1997; 30(2): 487–95
Hohnloser SH. Proarrhythmia with class III antiarrhythmic drugs: types, risks, and management. Am J Cardiol 1997; 80(8A): 82G–9G
Kronstein P. Briefing book, April 7, 2009 PDAC serdolect (sertindole) tablets [online]. Available from URL: http://www.fda.gov [Accessed 2011 Jan 10]
Acknowledgements
No sources of funding were used in the preparation of this manuscript. CG, JJS, JKK, MPA and ET are authors of two patents describing the T-wave morphology method. BT has no disclosures. The authors thank Nathalie Blanco for providing data for the bilastine study and FAES.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graff, C., Struijk, J.J., Kanters, J.K. et al. Effects of Bilastine on T-wave Morphology and the QTc Interval. Clin Drug Investig 32, 339–351 (2012). https://doi.org/10.2165/11599270-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11599270-000000000-00000