Abstract
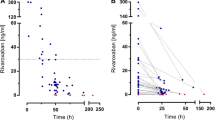
Low-molecular-weight heparins are anticoagulants used in the treatment of thrombosis. They have effectiveness and safety profiles similar to those of unfractionated heparin but are considered an attractive alternative because of their predictable pharmacokinetic characteristics. In this article, we demonstrate the need for therapeutic monitoring of low-molecular-weight heparins by addressing evidence arising from the literature and from modelling and simulation. First, treatment targets for unfractionated heparin and enoxaparin sodium were identified. Then, 10 000 virtual patients were simulated and the rate of treatment success was calculated. Treatment success was found to be similar between the two drugs (48% with unfractionated heparin and 54% with enoxaparin sodium). These results are consistent with empirical evidence from the literature and reflect the inadequacy of current dosing strategies in achieving desired biomarker targets. These results, along with recent advances in Bayesian forecasting techniques, support the need for routine monitoring and dose individualization of enoxaparin sodium in order to improve treatment success.





Similar content being viewed by others
References
Magee KD, Sevcik W, Moher D, et al. Low molecular weight heparins versus unfractionated heparin for acute coronary syndromes. Cochrane Database Syst Rev 2003; (1): CD002132
Murphy SA, Gibson CM, Morrow DA, et al. Efficacy and safety of the low-molecular weight heparin enoxaparin compared with unfractionated heparin across the acute coronary syndrome spectrum: a meta-analysis. Eur Heart J 2007 Sep; 28(17): 2077–86
van Dongen CJ, van den Belt AG, Prins MH, et al. Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for venous thromboembolism. Cochrane Database Syst Rev 2004; (4): CD001100
Turpie AG. Successors to heparin: new antithrombotic agents. Am Heart J 1997 Nov; 134(5 Pt 2): S71–7
Hirsh J, Warkentin TE, Shaughnessy SG, et al. Heparin and low-molecular-weight heparin: mechanisms of action, pharmacokinetics, dosing, monitoring, efficacy, and safety. Chest 2001 Jan; 119 (1 Suppl.): 64S–94S
Antman EM, McCabe CH, Gurfinkel EP, et al. Enoxaparin prevents death and cardiac ischemic events in unstable angina/non-Q-wave myocardial infarction: results of the Thrombolysis in Myocardial Infarction (TIMI) 11B trial. Circulation 1999 Oct 12; 100(15): 1593–601
Cohen M, Demers C, Gurfinkel EP, et al. A comparison of low-molecular-weight heparin with unfractionated heparin for unstable coronary artery disease. Efficacy and Safety of Subcutaneous Enoxaparin in Non-Q-Wave Coronary Events Study Group. N Engl J Med 1997 Aug 14; 337(7): 447–52
Green B, Duffull SB. Development of a dosing strategy for enoxaparin in obese patients. Br J Clin Pharmacol 2003 Jul; 56(1): 96–103
Green B, Greenwood M, Saltissi D, et al. Dosing strategy for enoxaparin in patients with renal impairment presenting with acute coronary syndromes. Br J Clin Pharmacol 2005 Mar; 59(3): 281–90
Begg EJ, Barclay ML. Aminoglycosides: 50 years on. Br J Clin Pharmacol 1995 Jun; 39(6): 597–603
Hirsh J, Raschke R. Heparin and low-molecular-weight heparin: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004 Sep; 126 (3 Suppl.): 188S–203S
Sanofi-Aventis New Zealand Limited. Clexane® and Clexane® Forte: data sheet. Auckland: Sanofi-Aventis New Zealand Limited, 2010 Apr 13 [online]. Available from URL: http://medsafe.govt.nz/profs/Datasheet/c/Clexaneinj.pdf [Accessed 2010 May 25]
The Thrombolysis in Myocardial Infarction (TIMI) 11A Trial Investigators. Dose-ranging trial of enoxaparin for unstable angina: results of TIMI 11A. The Thrombolysis in Myocardial Infarction (TIMI) 11A Trial Investigators. J Am Coll Cardiol 1997 Jun; 29(7): 1474–82
Montalescot G, Collet JP, Tanguy ML, et al. Anti-Xa activity relates to survival and efficacy in unselected acute coronary syndrome patients treated with enoxaparin. Circulation 2004 Jul 27; 110(4): 392–8
Barras MA, Duffull SB, Atherton JJ, et al. Individualized compared with conventional dosing of enoxaparin. Clin Pharmacol Ther 2008 Jun; 83(6): 882–8
Giugliano RP, Rohatagi S, Kastrissios H, et al. The relationship between oral factor Xa (FXa) inhibitor DU-176b pharmacokinetics (PK) and the probability of bleeding events (BE) in patients with atrial fibrillation (AF) [abstract no. OC-WE-003]. XXII Congress, International Society on Thrombosis and Haemostasis; 2009 Jul 11-16; Boston (MA) [online]. Available from URL: http://abstract.mci-group.com/cgi-bin/mc/printabs.pl?APP=ISTH2009ABS-abstract&TEMPLATE=&keyf=2790&showHide=show&client= [Accessed 2010 May 25]
Mungall D, Floyd R. Bayesian forecasting of APTT response to continuously infused heparin with and without warfarin administration. J Clin Pharmacol 1989 Nov; 29(11): 1043–7
Petersen JL, Mahaffey KW, Hasselblad V, et al. Efficacy and bleeding complications among patients randomized to enoxaparin or unfractionated heparin for antithrombin therapy in non-ST-segment elevation acute coronary syndromes: a systematic overview. JAMA 2004 Jul 7; 292(1): 89–96
Ward ML, Duffull SB. Experience with heparin dosing at Christchurch Hospital. N Z Med J 1998 Jun 26; 111(1068): 228–30
Burton ME, Vasko MR, Brater DC. Comparison of drug dosing methods. Clin Pharmacokinet 1985 Jan-Feb; 10(1): 1–37
Duffull SB, van den Berg L, Kirkpatrick C. TCIWorks: target concentration intervention software [online]. Available from URL: http://www.tciworks.info [Accessed 2010 May 25]
Acknowledgements
The authors thank the School of Pharmacy at the University of Otago, Dunedin, New Zealand, for facilitating this research. The authors have no conflicts of interest that are directly relevant to the content of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Sallami, H.S., Barras, M.A., Green, B. et al. Routine Plasma Anti-Xa Monitoring is Required for Low-Molecular-Weight Heparins. Clin Pharmacokinet 49, 567–571 (2010). https://doi.org/10.2165/11532960-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11532960-000000000-00000