Abstract
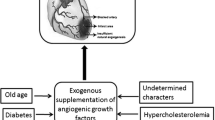
Currently available approaches for treating human coronary heart disease aim to relieve symptoms and the risk of myocardial infarction by reducing myocardial oxygen demand (drugs), preventing further disease progression (drugs), restoring coronary blood flow either pharmacologically (thrombolysis) or mechanically (angioplasty), or bypassing the stenotic lesions and obstructed coronary artery segments (surgery). Direct gene therapy, as well as gene-derived therapy, especially by angiogenic growth factors, is emerging as a potential new treatment for cardiovascular disease. After extensive experimental research on angiogenic growth factors, the first clinical studies on patients with coronary heart disease or peripheral vascular lesions are being performed.
The polypeptides fibroblast growth factor (FGF) and vascular endothelial growth factor seem to be effective in initiating neovascularisation (neo-angiogenesis) in hypoxic or ischaemic tissues. The first clinical study on patients with coronary heart disease treated by local injection of FGF-1 into the compromised underperfused myocardial tissue showed a 3-fold increase of capillary density mediated by the growth factor.
Angiogenic therapy of the human myocardium introduces a new modality of treatment for coronary heart disease in terms of regulation of blood vessel growth. Beyond drug therapy, angioplasty and bypass surgery, this therapy may evolve to be a fourth principle of treatment of atherosclerotic cardiovascular disease.
Similar content being viewed by others
References
Loop FD. Coronary artery surgery: the end of the beginning. Eur J Cardiothorac Surg 1998; 14: 554–71
Folkman J, Klagsbrun M. Angiogenic factors. Science 1987; 235: 442–7
Folkman J, Shing Y. Angiogenesis. J Biol Chem 1992; 267: 10931–4
Folkman J. Clinical applications of research on angiogenesis. N Engl J Med 1995; 333: 1757–63
Burgess WH, Mehrman T, Friesel R, et al. Multiple forms of endothelial cell growth factor. Rapid isolation and biological and chemical characterization. J Biol Chem 1985; 260: 11389–92
Thomas KA. Vascular endothelial growth factor, a potent and selective angiogenetic agent. J Biol Chem 1996; 271: 603–6
Thompson JA, Haudenschild CC, Anderson KD, et al. Heparinbinding growth factor induces the formation of organoid neovascular structures in vivo. Proc Natl Acad Sci U S A 1989; 86: 7928–32
Dvorak HF, Brown LF, Detmar M, et al. Vascular permeability factor/vascular endothelial growth factor, micro-vascular hyperpermeability and angiogenesis. Am J Pathol 1995; 146: 1029–39
Friesel R, Burgess WH, Mehrman T, et al. The characterization of the receptor for endothelial cell growth factor by covalent ligand attachment. J Biol Chem 1986; 261: 7581–4
Ware JA, Simons M. Angiogenesis in ischemic heart disease. Nature Med 1997; 3: 158–64
Slavin J. Fibroblast growth factors: at the heart of angiogenesis. Cell Biol In 1995; 19: 431–44
Kuwabara K, Ogawa S, Matsumoto M, et al. Hypoxia-mediated induction of acidic/basic fibroblast growth factor and platelet-derived growth factor in mononuclear phagocytes stimulates growth of hypoxic endothelial cells. Proc Natl Acad Sci U S A 1995; 92: 4606–10
Shweiki D, Itin A, Soffer D, et al. Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 1992; 359: 843–5
Ferrara N, Carrer-Moore K, Chen H, et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 1996; 380: 439–42
Folkman J. Tumor angiogenesis: therapeutic implications. N Engl J Med 1971; 285: 1182–6
Gimbrone MA Jr, Cotran RS, Folkman J. Endothelial regeneration and turnover: studies with human endothelial cell cultures. Ser Haematol 1973; 6: 453–5
Shing Y, Folkman J, Sullivan R, et al. Heparin affinity: purification of a tumor-derived capillary endothelial cell growth factor. Science 1984; 223: 1296–8
Maciag T, Mehrman T, Friesel R, et al. Heparin binds endothelial cell growth factor, the principal mitogen in the bovine brain. Science 1984; 225: 932–5
Baird A, Bohlen P, Esch F, et al. Acidic fibroblast growth factor (FGF) from bovine brain: amino-terminal sequence and comparison with basic FGF. EMBO J 1985; 4(8): 1951–6
Taylor S, Folkman J. Protamine is an inhibitor of angiogenesis. Nature 1982; 297: 307–12
Crum R, Szabo S, Folkman J. A new class of steroids inhibits angiogenesis in the presence of heparin or a heparin fragment. Science 1985; 230: 1375–8
White CW, Sondheimer HM, Crouch EC, et al. Treatment of pulmonary hemangiomatosis with recombinant interferon alfa-2a. N Engl J Med 1989; 320: 1197–200
Sorelle R. Two sides of the same coin. Circulation 1998; 98: 383–4
Isner JM, Pieczek A, Schainfeld R, et al. Clinical evidence of angiogenesis after arterial gene transfer of ph VEGF165 in patient with ischemic limb. Lancet 1996; 348: 370–4
Baumgartner J, Pieczek A, Manor O, et al. Constitutive expression of ph VEGF165 after intramuscular gene transfer promotes collateral vessel development in patients with critical limb ischemia. Circulation 1998; 97: 1114–23
Schumacher B, Pecher P, von Specht BU, et al. Induction of neoangiogenesis in ischemic myocardium by human growth factors. First clinical results of a new treatment of coronary heart disease. Circulation 1998; 97(7): 645–50
Tuder RM, Flook BE, Voelkel NF. Increased gene expression for VEGF and the VEGF receptors KDR/Flk and Flt in lungs exposed to acute or to chronic hypoxia. Modulation of gene expression by nitric oxide. J Clin Invest 1995; 95: 1798–807
Nomura M, Yamagishi S, Havada S, et al. Possible participation of autocrine and paracrine vascular endothelial growth factors in hypoxia-induced proliferation of endothelial cells and pericytes. J Biol Chem 1995; 270: 28316–24
Westernacher D, Schaper W. A novel heart derived inhibitor of vascular cell proliferation. Purification and biological activity. J Mol Cell Cardiol 1995; 27: 1535–43
Yanagisawa-Miwa A, Uchida Y, Nakamura F, et al. Salvage of infarcted myocardium by angiogenic action of basic fibroblast growth factor. Science 1992: 257: 1401–3
Battler A, Scheinowitz M, Bor A, et al. Intracoronary injection of basic fibroblast growth factor enhances angiogenesis in infarcted swine myocardium. J Am Coll Cardiol 1993; 22: 2001–6
Giordano FJ, Ping P, McKirnan MD, et al. Intracoronary transfer of fibroblast growth factor-5 increases blood flow and contractile function in an ischemic region of the heart. Nature Med 1996; 2: 534–9
Lazarous DF, Scheinowitz M, Shou M, et al. Effects of chronic systemic administration of basic fibroblast growth factor on collateral development in the canine heart. Circulation 1995; 91: 145–53
Unger EF, Banai S, Shou M, et al. Basic fibroblast growth factor enhances myocardial collateral flow in a canine model. Am J Physiol 1994; 266: 1588–95
Harada K, Grossman W, Friedman M, et al. Basic fibroblast growth factor improves myocardial function in chronically ischemic porcine hearts. J Clin Invest 1994; 94: 623–30
Banai S, Jaklitsch MT, Casscells W, et al. Effects of acidic fibroblast growth factor on normal and ischemic myocardium. Circulation Res 1991; 69: 76–85
Fasol R, Schumacher B, Schlaudraff K, et al. Experimental use of a modified fibrin-glue to induce site-directed angiogenesis from the aorta to the heart. J Thorac Cardiovasc Surg 1994; 107(6): 1432–9
Schlaudraff K, Schumacher B, von Specht BU, et al. Growth of ‘new’ coronary vascular structures by angiogenetic growth factor. Eur J Cardiothorac Surg 1993; 7(12): 637–44
Sellke FW, Wang SY, Stamler A, et al. Angiogenesis induced by acidic fibroblast growth factor as an alternative method of revascularization for chronic myocardial ischemia. Surgery 1996; 120: 182–8
Pearlman JD, Hibberd MG, Chuang ML, et al. Magnetic resonance mapping demonstrates benefits of VEGF-induced myocardial angiogenesis. Nature Med 1995; 1: 1085–9
Banai S, Jaklitsch MT, Shou M, et al. Angiogenic-induced enhancement of collateral flow to ischemic myocardium by vascular endothelial growth factor in dogs. Circulation 1994; 89: 2183–9
Baffour R, Berman J, Garb JL, et al. Enhanced angiogenesis and growth of collaterals by in vivo administration of recombinant basic fibroblast growth factor in a rabbit model of acute lower limb ischemia: dose-response effect of basic fibroblast growth factor. J Vasc Surg 1992; 16: 181–91
Mack CA, Magovern CJ, Budenbender KT, et al. Salvage angiogenesis induced by adenovirus-mediated gene transfer of vascular endothelial growth factor protects against ischemic vascular occlusion. J Vasc Surg 1998; 27: 699–709
Takeshita S, Zheng LP, Brogi E, et al. Therapeutic angiogenesis. Single intraarterial bolus of vascular endothelial growth factor augments revascularization in a rabbit ischemic hind limb model. J Clin Invest 1994; 93: 662–70
Pelletier MP, Giaid A, Sivaraman S, et al. Angiogenesis and growth factor expression in a model of transmyocardial revascularization. Ann Thorac Surg 1998; 66: 12–8
Sellke FW, Laham RJ, Edelman R, et al. Therapeutic angiogenesis with basic fibroblast growth factor: technique and early results. Ann Thorac Surg 1998; 65: 1540–4
Kuzuya M, Satake S, Esaki T, et al. Induction of angiogenesis by smooth muscle cell-derived factor: possible role in neovascularization in atherosclerotic plaque. J Cell Physiol 1995; 164: 658–67
Folkman J. Therapeutic angiogenesis in ischemic limbs [editorial]. Circulation 1998; 97: 1108–10
Folkman J. Angiogenic therapy of the human heart [editorial]. Circulation 1998; 97(7): 628–9
Schumacher B, von Specht BU, Haberstroh J, et al. The stimulation of neo-angiogenesis in the ischemic heart by the human growth factor FGF. J Cardiovasc Surg 1998; 39: 445–53
Schumacher B, Stegmann T, Pecher P. The stimulation of neoangiogenesis in the ischemic human heart by the growth factor FGF: first clinical results. J Cardiovasc Surg 1998; 39(6): 783–9
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stegmann, T.J. New Approaches to Coronary Heart Disease. BioDrugs 11, 301–308 (1999). https://doi.org/10.2165/00063030-199911050-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-199911050-00002