Abstract
Objective: To assess the cost effectiveness of aspirin 25mg plus dipyridamole 200mg twice daily in the secondary prevention of ischaemic stroke, according to the French social security perspective, using efficacy data from the second European Stroke Prevention Study (ESPS-2). The ESPS-2 was a double-blind, placebo-controlled clinical trial which assessed the efficacy of four secondary prevention strategies: (i) placebo; (ii) aspirin (acetylsalicylic acid) 25mg twice daily; (iii) dipyridamole 200mg twice daily; and (iv) aspirin 25mg plus dipyridamole 200mg twice daily.
Method: We performed a cost-effectiveness analysis with Monte Carlo simulations to compute confidence intervals. We combined data from various sources including the Dijon Stroke Registry, Institut National de la Statistique et des Etudes Economiques, Etude du Coût de l’Infarctus Cérébral (Study of the Cost of Cerebral Infarction [ECIC]) study and the ESPS-2 trial.
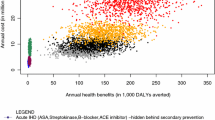
Results: According to our findings, a preventive strategy with aspirin 25mg plus dipyridamole 200mg twice daily is associated with net benefits per avoided stroke recurrence amounting to $US23 932 (95% CI -$US32 609, $US35 772) compared with aspirin 25mg twice daily alone, and $US31 555 (95% CI $US4921, $US74 515) compared with dipyridamole alone (1997 values). Sensitivity analysis demonstrated that dipyridamole plus aspirin was still cost effective when the average cost of adverse effects per episode (ignored in the original estimation of the cost-effectiveness ratios due to a lack of data) was assumed to be $US8600 (50 000 French francs); this cost is unlikely as most of the adverse effects associated with aspirin plus dipyridamole are only slight to moderate in severity.
Conclusions: In the secondary prevention of stroke in France, this study suggests, given its underlying assumptions and data, that aspirin 25mg plus dipyridamole 200mg twice daily is likely to be a cost-effective strategy from the social security perspective, when compared with other relevant strategies that were evaluated in the ESPS-2 trial.







Similar content being viewed by others
Notes
The use of trade names is for product identification only and does not imply endorsement.
A French government official publication compiling all drugs reimbursed by the French Social Security, indicating the price and reimbursement rate.
References
Van Bergen PFMM, Jonker JJC, van Hout BA, et al. Costs and effects of long-term oral anticoagulant treatment after myocardial infarction. JAMA 1995; 273 (12): 925–8
Scott G, Scott HM. Application of the findings of the European Stroke Prevention Study 2 (ESPS-2) to a New Zealand ischaemic stroke cost analysis. Pharmacoeconomics 1997; 12 (6): 667–74
Chambers M, Hutton J, Gladman J. Cost-effectiveness analysis of antiplatelet therapy in the prevention of recurrent stroke in the UK. Pharmacoeconomics 1999; 16 (5 Pt 2): 577–93
Sarasin FP, Gaspoz JM, Bounameaux H. Cost-effectiveness of new antiplatelet regimens used as secondary prevention of stroke or transient ischemic attack. Arch Intern Med 2000; 160: 2773–8
Marissal JP, Selke B, Lebrun T. Economic assessment of the secondary prevention of ischaemic events with lysine acetylsalicylate. Pharmacoeconomics 2000; 18 (2): 185–200
Giroud M, Milan C, Beuriat P, et al. Incidence and survival rates during a two-year period of intracerebral and subarachnoid haemorrhages, cortical infarcts, lacunes and transient ischaemic attacks. The Stroke Registry of Dijon: 1985–1989. Int J Epidemiol 1991; 20 (4): 892–9
Lemesle M, Milan C, Faivre J, et al. Incidence trends of ischemic stroke and transient ischemic attacks in a well-defined French population from 1985 through 1994. Stroke 1999; 30: 371–7
INSEE. Emploi. In: Tableaux de l’éeconomie fran¸caise. Paris: INSEE, 2000: 72–3
Mercier MA, Lee P. Le marcheé du travail en France et au Royaume-Uni. Paris: INSEE Première n°670, 1999: 4
Spieler JF, Lanoe¨ JL, Amarenco P. La prise en charge des patients atteints d’infarctus céeréebral en France: analyse des fili`eres de soins et éevaluation de leur coˆut. L’éetude ECIC: 88 pages, Paris [unpublished paper]
Spieler JF, Lano¨e JL, Amarenco P. Socioeconomic aspects of postacute care for patients with brain infarction in France. Cerebrovasc Dis 2002; 13: 132–41
Diener HC, Cunha L, Forbes C, et al. European Stroke Prevention Study: II. Dipyridamole and aspirin in the secondary prevention of stroke. J Neurol Sci 1996; 143: 1–13
Garber AM, Phelps CE. Economic foundations of cost-effectiveness analysis. J Health Econ 1997; 16: 1–31
CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 1996; 348: 1329–39
Antiplatelet Trialists’ Collaboration. Collaborative overview of randomised trials of antiplatelet therapy: I. prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. BMJ 1994; 308: 81–106
Patrono C, Coller B, Dalen JE, et al. Platelet-active drugs: the relationships among dose, effectiveness and side effects. Chest 2001; 119: 39S-63S
Acknowledgements
This study received a grant from Boehringer Ingelheim, France. The authors have provided no information on conflicts of interest directly relevant to the content of this study.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marissal, JP., Selke, B. Economic Assessment of the Secondary Prevention of Ischaemic Stroke with Dipyridamole plus Aspirin (Aggrenox®/Asasantin®) in France. PharmacoEconomics 22, 661–670 (2004). https://doi.org/10.2165/00019053-200422100-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-200422100-00004