Abstract
Epidemic influenza continues to be associated with significant morbidity in the general population, and mortality in the elderly and other high risk patients. Although the case fatality rate averages less than 0.01%, tens of thousands of deaths occur each year. Control through immunisation programmes has not been possible due to incomplete protective efficacy and antigenic variations that occur frequently.
Currently available anti-influenza medications (amantadine and rimantadine) have had limited success due to underutilisation, lack of activity against influenza B, the rapid development of viral resistance to the drugs, and adverse effects. A new class of antiviral agents designed to inhibit influenza neuraminidase, an important surface glycoprotein, is currently under active development for use in the prophylaxis and treatment of influenza A and B infections. Two of these compounds, zanamivir (GG167) and GS4104 (the ethyl ester prodrug of GS4071) have reached clinical trials.
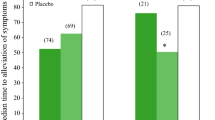
Most studies of zanamivir have involved topical administration by inhalation of dry powder aerosols and/or intranasal doses of aqueous solutions. These routes rapidly provide high local concentrations at the sites of delivery. GS4104 is administered orally, which allows for greater ease of administration, and probably more uniform distribution of the parent compound GS4071 in the respiratory tract. Both have shown potent inhibitory activity against influenza in animal models and experimental human influenza with excellent tolerability profiles. Zanamivir treatment has been shown to reduce the severity and duration of naturally occurring, uncomplicated influenza illness in adults. Clinical resistance to these drugs has not been recognised as a significant problem to date, although strains resistant to each agent have been produced in the laboratory. This class of agents shows considerable promise as a novel approach to prophylaxis and treatment of influenza infections. Ongoing studies should provide the data needed to allow the addition of 1 or more of the neuraminidase inhibitors to the clinician’s anti-influenza armamentarium.
Similar content being viewed by others
References
Colman PM, Varghese JN, Laver WG. Structure of the catalytic and antigenic sites in influenza virus neuraminidase. Nature 1983; 303(5912): 41–4
von Itzstein M, Wu WY, Kok GB, et al. Rational design of potent sialidase-based inhibitors of influenza virus replication. Nature 1993; 363(6428): 418–23
Colman PM. Influenza virus neuraminidase: structure, antibodies, and inhibitors. Protein Sci 1994; 3(10): 1687–96
Schultz-Cherry S, Hinshaw VS. Influenza virus neuraminidase activates latent transforming growth factor beta. J Virol 1996; 70(12): 8624–9
Houde M, Arora DJ. Stimulation of tumor necrosis factor secretion by purified influenza virus neuraminidase. Cell Immunol 1990; 129(1): 104–11
Murphy BR, Kasel JA, Chanock RM. Association of serum anti-neuraminidase antibody with resistance to influenza in man. N Engl J Med 1972; 286(25): 1329–32
Monto AS, Kendal AP. Effect of neuraminidase antibody on Hong Kong influenza. Lancet 1973; 1(7804): 623–5
Couch RB, Kasel JA, Gerin JL, et al. Induction of partial immunity to influenza by a neuraminidase-specific vaccine. J Infect Dis 1974; 129(4): 411–20
Beutner KR, Chow T, Rubi E, et al. Evaluation of a neuraminidase-specific influenza A virus vaccine in children: antibody responses and effects on two successive outbreaks of natural infection. J Infect Dis 1979; 140(6): 844–50
Webster RG, Reay PA, Laver WG. Protection against lethal influenza with neuraminidase. Virology 1988; 164(1): 230–7
Meindl P, Tuppy H. 2-deoxy-2,3-dehydroxialic acids I. Syntheses and properties of 2-deoxy,3dehydro-N-acylneuraminic acids and their methyl esters. Monatsc Chem 1969; 100: 1295–2306
Meindl P, Bodo G, Palese P, et al. Inhibition of neuraminidase activity by derivatives of 2-deoxy-2,3-dehydro-N-acetyl-neuraminic acid. Virology 1974; 58(2): 457–63
Varghese JN, Epa VC, Colman PM. Three-dimensional structure of the complex of 4-guanidino-Neu5Ac2en and influenza virus neuraminidase. Protein Sci 1995; 4(6): 1081–7
Pegg MS, von Itzstein M. Slow-binding inhibition of sialidase from influenza virus. Biochem Mol Biol Int 1994; 32(5): 851–8
Hart GJ, Bethell RC. 2,3-didehydro-2,4-dideoxy-4-guanidino-N-acetyl-D-neuraminic acid (4-guanidino-Neu5Ac2en) is a slow-binding inhibitor of sialidase from both influenza A virus and influenza B virus. Biochem Mol Biol Int 1995; 36(4): 695–703
Woods JM, Bethell RC, Coates JA, et al. 4-Guanidino-2,4-dideoxy-2,3-dehydro-N-acetylneuraminic acid is a highly effective inhibitor both of the sialidase (neuraminidase) and of growth of a wide range of influenza A and B viruses in. vitro. Antimicrob Agents Chemother 1993; 37(7): 1473–9
Thomas GP, Forsyth M, Penn CR, et al. Inhibition of the growth of influenza viruses in vitro by 4-guanidino-2,4-dideoxy-N-acetylneuraminic acid. Antiviral Res 1994; 24(4): 351–6
Gubareva LV, Penn CR, Webster RG. Inhibition of replication of avian influenza viruses by the neuraminidase inhibitor 4-guanidino-2,4-dideoxy-2,3-dehydro-N-acetylneuraminic acid. Virology 1995; 212(2): 323–30
Hayden FG, Rollins BS, Madren LK. Anti-influenza virus activity of the neuraminidase inhibitor 4-guanidino-Neu5Ac2en in cell culture and in human respiratory epithelium [erratum in Antiviral Res 1994 Dec; 25 (3–4): 287]. Antiviral Res 1994; 25(2): 123–31
Ryan DM, Ticehurst J, Dempsey MH, et al. Inhibition of influenza virus replication in mice by GG167 (4-guanidino-2,4-dideoxy-2,3-dehydro-N-acetylneuraminic acid) is consistent with extracellular activity of viral neuraminidase (sialidase). Antimicrob Agents Chemother 1994; 38(10): 2270–5
Ryan DM, Ticehurst J, Dempsey MH. GG167 (4-guanidino-2,4-dideoxy-2,3-dehydro-N-acetylneuraminic acid) is a potent inhibitor of influenza virus in ferrets. Antimicrob Agents Chemother 1995; 39(11): 2583–4
GG167 Clinical investigator’s brochure. 3-97. Greenford, Middlesex, England: Glaxo Wellcome Research and Development Ltd., March 1997
McCauley JW, Pullen LA, Forsyth M, et al. 4-Guanidino-Neu5Ac2en fails to protect chickens from infection with highly pathogenic avian influenza virus. Antiviral Res 1995; 27(1–2): 179–86
Sidwell RW, Huffman JH, Barnard DL, et al. Inhibition of influenza virus infections in mice by GS4104, an orally effective influenza virus neuraminidase inhibitor. Antiviral Res 1998; 37: 107–20
Efthymiopoulos C, Barrington P, Patel JA. Pharmacokinetics of the neuraminidase inhibitor 4-guanidino Neu5Ac2en (GG167) following intravenous, intranasal, and inhaled administration in man [abstract no. H70]. 34th Interscience Conference on Antimicrobial Agents and Chemotherapy, American Society of Microbiology; 1994 Oct 4–7; Orlando (FL), 265
Newman SP, Brown J, Pickford M, et al. Deposition pattern in the respiratory tract of the neuraminidase inhibitor zanamivir; a gamma scintigraphic study [abstract no. H-134]. 37th Inter-science Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 237
Hayden FG, Treanor JJ, Betts RF, et al. Safety and efficacy of the neuraminidase inhibitor GG167 in experimental human influenza. JAMA 1996; 275(4): 295–9
Walker JB, Hussey EK, Treanor JJ, et al. Effects of the neuraminidase inhibitor zanamivir on otologic manifestations of experimental human influenza. J Infect Dis 1997; 176: 1417–22
Hayden FG, Lobo M, Hussey EK, et al. Efficacy of intranasal GG167 in experimental human influenza A and B virus infection. In: Brown LE, Hampson AW, Webster RG, editors. Options for the control of influenza III. Amsterdam: Elsevier Science B.V, 1996: 718–25
Hayden FG, Osterhaus ADME, Treanor JJ, et al. Efficacy and safety of the neuraminidase inhibitor zanamivir in the treatment of influenza virus infections. N Engl J Med 1997; 337(13): 874–9
Matsumoto K, Nerome K, Numasaki Y. Inhaled and intranasal GG167 in the treatment of influenza A and B: preliminary results. In: Brown LE, Hampson AW, Webster RG, editors. Options for the control of influenza III. Amsterdam: Elsevier Science; 1996: 713–7
Aoki FY, Fleming DM, Lacey L, et al. Impact of treatment of influenza with zanamivir on patients’ health status, sleep quality, productivity and healthcare use [abstract no. N-15]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 384
Schilling M, Povinelli L, Krause P, et al. Efficacy of zanamivir of chemoprophylaxis of nursing home influenza A outbreaks [abstract no. H-92]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 230
Kim CU, Lew W, Williams MA, et al. Influenza neuraminidase inhibitors possessing a novel hydrophobic interaction in the enzyme active site: design, synthesis, and structural analysis of carbocyclic sialic acid analogues with potent anti-influenza activity. J Am Chem Soc 1997; 119: 681–90
Hayden FG, Rollins BS. In vitro activity of the neuraminidase inhibitor GS4071 against influenza viruses [abstract no. 159]. Antiviral Res 1997; 34: A86
Mendel DB, Tai CY, Escarpe PA, et al. Oral administration of a prodrug of the influenza virus neuraminidase inhibitor GS4071 protects mice and ferrets against influenza infection. Antimicrob Agents Chemother 1998; 42(3): 640–6
Li W, Escarpe PA, Eisenberg EJ, et al. Identification of GS4104 as an orally bioavailable prodrug of the influenza virus neuraminidase inhibitor GS4071. Antimicrob Agents Chemother 1998; 42(3): 647–53
Cundy K, Eisenberg G, Bidgood A, et al. The novel influenza neuraminidase inhibitor prodrug GS4104 is highly bioavailable in animals [abstract H-135]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 237
Eisenberg G, Bidgood A, Lynch G, et al. Penetration of GS4071, a novel influenza neuraminidase inhibitor, into rat bronchoalveolar lining fluid following oral administration of the prodrug GS4104. Antimicrob Agents Chemother 1997; 41: 1949–52
Investigational drug brochure: Ro64-0796. 2-1997. Gilead-Sciencs and F Hoffman-La Roche Ltd: 1–26 (Data on file)
Fenton RJ, Morley PJ, Owens IJ, et al. Activities of zanamivir (GG167) and GS4104 in a series of influenza A virus animal models [abstract no. 173]. Antiviral Res 1998; 37: A88
Wood ND, Aitken M, Sharp S, et al. Tolerability and pharmacokinetics of the influenza neuraminidase inhibitor Ro-64-0802 (GS4071) following oral administration of the prodrug Ro-64-0796 (GS4104) to healthy male volunteers [abstract no. A-123]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 25
Hayden FG, Lobo M, Treanor JJ, et al. Efficacy and tolerability of oral GS4104 for early treatment of experimental influenza in humans [abstract no. LB-26]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 14
Barnett J, Dempsey M, Rothbarth PH, et al. Susceptibility monitoring of influenza virus clinical isolates to the neuraminidase inhibitor zanamivir (GG167) during phase II clinical efficacy trials [abstract no. H-93]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1997 Sep 28–Oct 1; Toronto (ON), 230
McKimm-Breschkin JL, Blick TJ, Sahasrabudhe A. Influenza virus variants with decreased sensitivity to 4-amino- and 4-guanidino-Neu5Ac2en. In: Brown LE, Hampson AW, Webster RG, editors. Options for the control of influenza III. Amsterdam: Elsevier Science; 1996: 726–34
McKimm-Breschkin JL, Blick TJ, Sahasrabudhe A, et al. Generation and characterization of variants of NWS/G70C influenza virus after in. vitro passage in 4-amino-Neu5Ac2en and 4-guanidino-Neu5Ac2en. Antimicrob Agents Chemother 1996; 40(1): 40–6
Penn CR, Barnett J, Bethell R, et al. Selection of influenza virus with reduced sensitivity in vitro to the neuraminidase inhibitor GG167 (4-guanidino-Neu5Ac2en): changes in haemagglutinin may compensate for loss of neuraminidase activity. In: Brown LE, Hampson AW, Webster RG, editors. Options for the control of influenza III. Amsterdam, Elsevier Science B.V.; 1996:735–40
Blick TJ, Tiong T, Sahasrabudhe A, et al. Generation and characterization of an influenza virus neuraminidase variant with decreased sensitivity to the neuraminidase-specific inhibitor 4-guanidino-Neu5Ac2en. Virology 1995; 214(2): 475–84
Staschke KA, Colacino JM, Baxter AJ, et al. Molecular basis for the resistance of influenza viruses to 4-guanidino-Neu5Ac2en. Virology 1995; 214(2): 642–6
Gubareva LV, Bethell R, Hart GJ, et al. Characterization of mutants of influenza A virus selected with the neuraminidase inhibitor 4-guanidino-Neu5Ac2en. J Virol 1996; 70(3): 1818–27
Gubareva LV, Robinson MJ, Bethell RC, et al. Catalytic and framework mutations in the neuraminidase active site of influenza viruses that are resistant to 4-guanidino-Neu5Ac2en. J Virol 1997; 71(5): 3385–90
Gubareva LV, Bethell R, Penn CR, et al. In vitro characterization of 4-guanidino-Neu5Ac2en-resistant mutants of influenza A virus. In: Brown LE, Hampson AW, Webster RG, editors. Options for the control of influenza III. Amsterdam, Elsevier Science; 1996: 753–60
McKimm-Breschkin JL, Sahasrabudhe A, Blick TJ, et al. Mutations in a conserved residue in the influenza virus neuraminidase active site decreases sensitivity to Neu5Ac2en-derived inhibitors. J Virol 1998; 72(3): 2456–62
Sahasrabudhe A, Blick TJ, McKimm-Breschkin JL. Influenza virus variants resistant to GG167 with mutations in the hemagglutinin. In: Brown LE, Hampson AW, Webster RG, editors. Options for the control of influenza III. Amsterdam: Elsevier Science; 1996: 748–52
Gubareva LV, Matrosovich MN, Brenner MK, et al. Evidence for zanamivir resistance in an immunocompromised child infected with influenza B virus. J Infect Dis. In press
von Itzstein M, Dyason JC, Oliver SW, et al. A study of the active site of influenza virus sialidase: an approach to the rational design of novel anti-influenza drugs. J Med Chem 1996; 19; 39(2): 388–91
White CL, Janakiraman MN, Laver WG, et al. A sialic acid-derived phosphonate analog inhibits different strains of influenza virus neuraminidase with different efficiencies. J Mol Biol 1995; 245(5): 623–34
Singh S, Jedrzejas MJ, Air GM, et al. Structure-based inhibitors of influenza virus sialidase: a benzoic acid lead with novel interaction. J Med Chem 1995; 38(17): 3217–25
Murakami M, Ikeda K, Achiwa K. Chemoenzymatic synthesis of neuraminic acid analogs structurally varied at C-5 and C-9 as potential inhibitors of the sialidase from influenza virus. Carbohydr Res 1996; 280(1): 101–10
Sollis S, Smith PW, Howes PD. Novel inhibitors of influenza sialidase related to GG167._Synthesis of 4-amino and guanidino-4H-pyran-2-carboxylic acid-6-propylamides; selective inhibitors of influenza virus sialidase. Bioorg Med Chem 1996; 6: 1805–8
Barrere B, Driguez PA, Maudrin J, et al. A novel synthetic reversible inhibitor of sialidase efficiently blocks secondary but not primary influenza virus infection of MDCK cells in culture. Arch Virol 1997; 142(7): 1365–80
Nagai T, Moriguchi R, Suzuki Y, et al. Mode of action of the anti-influenza virus activity of plant flavonoid, 5,7,4′-trihydroxy-8-methoxyflavone, from the roots of Scutellaria baicalensis. Antiviral Res 1995; 26(1): 11–25
Nagai T, Miyaichi Y, Tomimori T, et al. In vivo anti-influenza virus activity of plant flavonoids possessing inhibitory activity for influenza virus sialidase. Antiviral Res 1992; 19(3): 207–17
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Calfee, D.P., Hayden, F.G. New Approaches to Influenza Chemotherapy. Drugs 56, 537–553 (1998). https://doi.org/10.2165/00003495-199856040-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199856040-00003