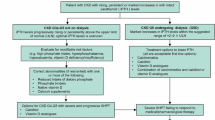
Abstract
Patients with chronic kidney disease (CKD) are generally affected by secondary hyperparathyroidism (SHPT). High phosphate, low calcium and vitamin D deficiency represent the classical ‘triad’ involved into the pathogenesis of SHPT in renal insufficiency, in which downregulation of the parathyroid vitamin D receptor and calcium-sensing receptor represents a critical step. Recently, new studies indicate that fibroblast growth factor 23 may play a central role in the regulation of phosphate-vitamin D metabolism in patients with CKD.
These new insights into the pathogenesis of SHPT will possibly improve the treatment of this condition in patients with CKD. The ‘modern’ treatment of SHPT in CKD patients consists of free-calcium and aluminium phosphate binders, vitamin D receptor activators and calcimimetics. However, calcium- and aluminium-based phosphate binders and calcitriol are therapeutic tools that are not without complications, including increasing the risk of cardiovascular calcification in patients with CKD. This review summarizes the current understanding and evidence supporting strategies for SHPT treatment in CKD patients, with particular focus on the elderly, although specific guidelines for control of this disorder in this age group are lacking.
Similar content being viewed by others
References
Jadoul M, Albert JM, Akiba T, et al. Incidence and risk factors for hip or other bone fractures among hemodialysis patients in the Dialysis Outcomes and Practice Pattern Study. Kidney Int 2006; 70: 1358–66
Ganesh SK, Stack AG, Levin NW, et al. Association of elevated serum PO4, CaPO4 product, and parathyroid hormone levels with cardiac mortality risk in chronic hemodialysis patients. J Am Soc Nephrol 2001; 12: 2131–8
Block GA, Klassen PS, Lazarus JM, et al. Mineral metabolism, mortality, and morbidity in maintenance hemodialysis patients. J Am Soc Nephrol 2004; 15: 2208–18
Young EW, Albert JM, Satayathum S, et al. Predictors and consequences of altered mineral metabolism: the Dialysis Outcomes and Practice Pattern Study. Kidney Int 2005; 67: 1179–87
Cozzolino M, Galassi A, Gallieni M, et al. Pathogenesis and treatment of secondary hyperparathyroidism in dialysis patients: the role of paricalcitol. Curr Vasc Pharmacol 2008 Apr; 6(2): 148–53
Emmett M. What does serum fibroblast growth factor 23 do in hemodialysis patients? Kidney Int 2008 Jan; 73(1): 3–5
Gutierrez OM, Mannstadt M, Isakova T, et al. Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med 2008; 359: 584–92
Heaney RP. Thinking straight about calcium. N Engl J Med 1993; 328: 503–5
National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis 2003; 42Suppl. 3: 63–200
Moe S, Drüeke T, Cunningham J, et al. Kidney Disease: Improving Global Outcomes (KDIGO) definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 2006 Jun; 69(11): 1945–53
Cozzolino M, Galassi A, Pasho S, et al. Preventive measures and new pharmacological approaches of calcium and phosphate disorders. Contrib Nephrol 2008; 161: 234–9
Bro S, Olgaard K. Effects of excess PTH on nonclassical target organs. Am J Kidney Dis 1997; 30: 606–20
Elder G. Pathophysiology and recent advances in the management of renal osteodystrophy. J Bone Miner Res 2002; 17: 2094–105
London GM. Cardiovascular calcifications in uremic patients: clinical impact on cardiovascular function. J Am Soc Nephrol 2003; 14: S305–9
Cozzolino M, Brancaccio D, Gallieni M, et al. Pathogenesis of vascular calcification in chronic kidney disease. Kidney Int 2005; 68(2): 429–36
Cozzolino M, Mazzaferro S, Pugliese F, et al. Vascular calcification and uremia: what do we know? Am J Nephrol 2008; 28: 339–46
Lips P, Wiersinga A, van Ginkel FC, et al. The effect of vitamin D supplementation on vitamin D status and parathyroid function in elderly subjects. J Clin Endocrinol Metab 1988; 67: 644–50
Lips P. Vitamin D deficiency and secondary hyperparathyroidism in the elderly: consequences for bone loss and fractures and therapeutic implications. Endocrine Rev 2001; 22(4): 477–501
Bruce DG, St John A, Nicklason F, et al. Secondary hyperparathyroidism in patients from Western Australia with hip fracture: relationship to type of hip fracture, renal function, and vitamin D deficiency. J Am Geriatr Soc 1999; 47: 354–9
Woitge HW, Scheidt-Nave C, Kissling C, et al. Seasonal variation of biochemical indexes of bone turnover: results of a population-based study. J Clin Endocrinol Metab 1998; 83: 68–75
Demiaux B, Arlot ME, Chapuy MC, et al. Serum osteocalcin is increased in patients with osteomalacia: correlations with biochemical and histomorphometric findings. J Clin Endocrinol Metab 1992; 74: 1146–51
Sahota O, Masud T, San P, et al. Vitamin D insufficiency increases bone turnover markers and enhances bone loss at the hip in patients with established vertebral osteoporosis. Clin Endocrinol (Oxf) 1999; 51: 217–21
Khosla S, Atkinson EJ, Melton III LJ, et al. Effects of age and estrogen status on serum parathyroid hormone levels and biochemical markers of bone turnover in women: a population-based study. J Clin Endocrinol Metab 1997; 82: 1522–7
Ooms ME, Lips P, Roos JC, et al. Vitamin D status and sex hormone binding globulin: determinants of bone turnover and bone mineral density in elderly women. J Bone Miner Res 1995; 10: 1177–84
Cozzolino M, Brancaccio D, Gallieni M, et al. Pathogenesis of parathyroid hyperplasia in renal failure. J Nephrol 2005; 18(1): 5–8
Szabo A, Merke J, Beier E, et al. 1,25(OH)2vitamin D3 inhibits parathyroid cell proliferation in experimental uremia. Kidney Int 1989; 35: 1049–56
Healy KD, Vanhooke JL, Prahl JM, et al. Parathyroid hormone decreases renal vitamin D receptor expression in vivo. Proc Natl Acad Sci U S A 2005; 102: 4724–8
Brown AJ, Zhong M, Finch J, et al. The roles of calcium and 1,25-dihydroxyvitamin D3 in the regulation of vitamin D receptor expression by rat parathyroid glands. Endocrinology 1995; 136: 1419–25
Brown AJ, Zhong M, Finch J, et al. Rat calcium-sensing receptor is regulated by vitamin D but not by calcium. Am J Physiol 1996; 270: F454–60
Slatopolsky E, Weerts C, Thielan J, et al. Marked suppression of secondary hyperparathyroidism by intravenous administration of 1,25-dihydroxy-cholecalciferol in uremic patients. J Clin Invest 1984; 74(6): 2136–43
Sjoden G, Lindgren JU, DeLuca HF. Antirachitic activity of 1 alpha-hydroxyergocholecalciferol and 1 alpha-hydroxycholecalciferol in rats. J Nutr 1984; 114: 2043–204
Slatopolsky E, Finch J, Ritter C, et al. A new analog of calcitriol, 19-nor-1,25(OH)2D2, suppresses parathyroid hormone secretion in uremic rats in the absence of hypercalcemia. Am J Kidney Dis 1995; 26: 852–60
Brown AJ, Finch J, Grieff M, et al. The mechanism for the disparate actions of calcitriol and 22-oxacalcitriol in the intestine. Endocrinology 1993; 133: 1158–64
Gonzalez EA, Sachdeva A, Oliver DA, et al. Vitamin D insufficiency and deficiency in chronic kidney disease: a single center observational study. Am J Nephrol 2004; 24: 503–10
Levin A, Bakris GL, Molitch M, et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int 2007; 71(1): 31–8
Rix M, Andreassen H, Eskildsen P, et al. Bone mineral density and biochemical markers of bone turnover in patients with pre-dialysis chronic renal failure. Kidney Int 1999; 56: 1084–93
Rix M, Eskildsen P, Olgaard K. Effect of 18 months of treatment with alfacalcidol on bone in patients with mild to moderate chronic renal failure. Nephrol Dial Transplant 2004 Apr; 19(4): 870–6
Slatopolsky E, Cozzolino M, Finch JL. Differential effects of 19-nor-1,25-(OH)2D2 and 1alpha-hydroxyvitamin D2 on calcium and phosphorus in normal and uremic rats. Kidney Int 2002; 62(4): 1277–84
Takahashi F, Finch JL, Denda M, et al. A new analog of 1,25-(OH)2D3, 19-nor-1,25-(OH)2D2, suppresses serum PTH and parathyroid gland growth in uremic rats without elevation of intestinal vitamin D receptor content. Am J Kidney Dis 1997; 30: 105–12
Mizobuchi M, Finch JL, Martin DR, et al. Differential effects of vitamin D receptor activators on vascular calcification in uremic rats. Kidney Int 2007; 72(6): 709–15
Cardus A, Panizo S, Parisi E, et al. Differential effects of vitamin D analogs on vascular calcification. J Bone Miner Res 2007 Jun; 22(6): 860–6
Martin K, Gonzalez EA, Gellens M, et al. 19-Nor-1-alpha-25-dihydroxyvitamin D2 (paricalcitol) safely and effectively reduces the levels of intact parathyroid hormone in patients on hemodialysis. J Am Soc Nephrol 1998; 9(8): 1427–32
Sprague SM, Llach F, Amdahl M, et al. Paricalcitol versus calcitriol in the treatment of secondary hyperparathyroidism. Kidney Int 2003; 63(4): 1483–90
Teng M, Wolf M, Lowrie E, et al. Survival of patients undergoing hemodialysis with paricalcitol or calcitriol. N Engl J Med 2003; 349: 446–56
Dobrez DG, Mathes A, Amdahl M, et al. Paricalcitol-treated patients experience improved hospitalization outcomes compared with calcitriol-treated patients in real-world clinical settings. Nephrol Dial Transplant 2004; 19(5): 1174–81
Palmer SC, McGregor DO, Macaskill P, et al. Metaanalysis: vitamin D compounds in chronic kidney disease. Ann Intern Med 2007 Dec 18; 147(12): 840–53
Hergesell O, Ritz E. Phosphate binders in uraemia: pharmacodynamics, pharmacoeconomics, pharmacoethics. Nephrol Dial Transplant 2002; 17: 14–7
Gallieni M, Cozzolino M, Carpani P, et al. Sevelamer reduces calcium load and maintains a low calcium-phosphorus ion product in dialysis patients. J Nephrol 2001; 14: 176–83
Posner BM, Borman CL, Morgan JL, et al. The validity of a telephone administered 24-hour recall methodology. Am J Clin Nutr 1982; 36: 546–53
Lorenzo V, Martín M, Rufino M, et al. Protein intake, control of serum phosphorus, and relatively low levels of parathyroid hormone in elderly hemodialysis patients. Am J Kidney Dis 2001; 37: 1260–6
Lorenzo Sellares V, Torres Ramírez A. Management of hyper-phosphataemia in dialysis patients: role of phosphate binders in the elderly. Drugs Aging 2004; 21: 153–65
Joy MS, Finn WF, LAM-302 Study Group. Randomized, double-blind, placebo-controlled, dose-titration, phase III study assessing the efficacy and tolerability of lanthanum carbonate: a new phosphate binder for the treatment of hyperphosphatemia. Am J Kidney Dis 2003; 42: 96–107
Mehrotra R, Martin KJ, Fishbane S, et al. Higher strength lanthanum carbonate provides serum phosphorus control with a low tablet burden and is preferred by patients and physicians: a multicenter study. Clin J Am Soc Nephrol 2008; 3: 1437–45
Ben-Dov IZ, Pappo O, Sklair-Levy M, et al. Lanthanum carbonate decreases PTH gene expression with no hepatotoxicity in uraemic rats. Nephrol Dial Transplant 2007 Feb; 22(2): 362–8
Spasovski GB, Sikole A, Gelev S, et al. Evolution of bone and plasma concentration of lanthanum in dialysis patients before, during 1 year of treatment with lanthanum carbonate and after 2 years of follow-up. Nephrol Dial Transplant. 2006 Aug; 21(8): 2217–24
Hutchison AJ, Barnett ME, Krause R, et al. Long-term efficacy and safety profile of lanthanum carbonate: results for up to 6 years of treatment. Nephron Clin Pract 2008; 110: c15–23
Chertow GM, Burke SK, Raggi P; Treat to Goal Working Group. Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients. Kidney Int 2002; 62: 245–52
Block GA, Raggi P, Bellasi A, et al. Mortality effect of coronary calcification and phosphate binder choice in incident hemodialysis patients. Kidney Int 2007; 71: 438–41
Suki WN, Zabaneh R, Cangiano JL, et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int 2007; 72: 1130–7
Ketteler M, Rix M, Fan S, et al. Efficacy and tolerability of sevelamer carbonate in hyperphosphatemic patients who have chronic kidney disease and are not on dialysis. Clin J Am Soc Nephrol 2008; 3: 1125–30
Delmez J, Block G, Robertson J, et al. A randomized, double-blind, crossover design study of sevelamer hydrochloride and sevelamer carbonate in patients on hemodialysis. Clin Nephrol 2007; 68: 386–91
Gallieni M, Cozzolino M, Brancaccio D. Transient decrease of serum bicarbonate levels with sevelamer hydrochloride as the phosphate binder. Kidney Int 2000; 57: 1776–7
Qunibi W, Moustafa M, Muenz LR, et al. A 1-year randomized trial of calcium acetate versus sevelamer on progression of coronary artery calcification in hemodialysis patients with comparable lipid control: the Calcium Acetate Renagel Evaluation-2 (CARE-2) study. Am J Kidney Dis 2008; 51: 952–65
Brown EM. Calcium receptor and regulation of parathyroid hormone secretion. Rev Endocr Metab Disord 2000; 1: 307–15
Block GA, Martin KJ, de Francisco ALM. Cinacalcet for secondary hyperparathyroidism in patients receiving hemodialysis. N Engl J Med 2004; 350: 1516–25
Valle C, Rodriguez M, Santamaría R, et al. Cinacalcet reduces the set point of the PTH-calcium curve. J Am Soc Nephrol 2008; 19: 2430–6
Messa P, Macério F, Yaqoob M, et al. The OPTIMA study: assessing a new cinacalcet (Sensipar/Mimpara) treatment algorithm for secondary hyperparathyroidism. Clin J Am Soc Nephrol 2008; 3: 36–45
Tominaga Y, Uchida K, Haba T, et al. More than 1000 cases of total parathyroidectomy with forearm autograft for renal hyperparathyroidism. Am J Kidney Dis 2001; 38Suppl. 1: S166–71
Kakuta T, Suzuki Y, Tadaki F, et al. Long-term prognosis of parathyroid function for chronic dialysis patients after minimally invasive radioguided parathyroidectomy (MIRP). Nephrol Dial Transplant 2003; 18Suppl. 3: 71–5
Kitaoka M, Fukagawa M, Ogata E, et al. Reduction of functioning parathyroid cell mass by ethanol injection in chronic dialysis patients. Kidney Int 1994; 46: 1110–7
Shiizaki K, Hatamura I, Negi S, et al. Percutaneous maxacalcitol injection therapy regresses hyperplasia of parathyroid and induces apoptosis in uremia. Kidney Int 2003; 64: 992–1003
Koiwa F, Kakuta T, Tanaka R, et al. Efficacy of percutaneous ethanol injection therapy (PEIT) is related to the number of parathyroid glands in haemodialysis patients with secondary hyperparathyroidism. Nephrol Dial Transplant 2007; 22: 522–8
Acknowledgements
No sources of funding were used to assist in the preparation of this review. Mario Cozzolino has received honoraria from Shire, Abbott, Amgen, Genzyme and Roche. Maurizio Gallieni has received honoraria from Genzyme and Amgen. Diego Brancaccio has received honoraria from Abbott, Shire and Amgen. The other authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cozzolino, M., Gallieni, M., Pasho, S. et al. Management of Secondary Hyperparathyroidism in the Elderly Patient with Chronic Kidney Disease. Drugs Aging 26, 457–468 (2009). https://doi.org/10.2165/00002512-200926060-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-200926060-00002