Summary
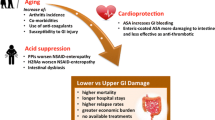
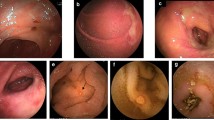
There is no doubt that nonsteroidal anti-inflammatory drugs (NSAIDs) cause gastrointestinal injury. The most serious consequences are gastric and duodenal ulcers which can cause bleeding and perforation, and which may lead to the premature death of 3000 to 4000 patients in the UK annually. The immediate actions of NSAIDs operate at a subcellular level; in particular altering of mitochondrial function which causes depletion of ATP and renders the cell vulnerable to oxidant stress. Secondary consequences follow, such as the inhibition of prostaglandin synthesis which delays cellular repair. While adaptation can be shown in volunteers despite continued NSAID ingestion, studies in patients suggest mucosal damage develops continuously and cumulatively even with low doses of aspirin.
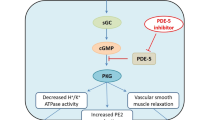
Histamine H2-receptor antagonists and proton pump inhibitors heal NSAID-related ulcers, though healing rates with H2-antagonists are slower in patients who continue NSAID treatment. They have little role in preventing damage. In addition to acid suppression, prostaglandin analogues cause bicarbonate secretion and enhance mucosal blood flow. They have a specific role in both prevention and treatment of NSAID-related damage. The use of misoprostol offers a rational approach to reduce the high prevalence of unwanted gastroduodenal damage from NSAIDs. On a purely financial basis more information is needed before routine coprescribing can be recommended. However, for any patient on NSAIDs with a previous ulcer or for patients aged over 60 years (where the risks and seriousness of complications are markedly increased), the use of misoprostol should be considered. Further developments in prostaglandin analogues may reduce their adverse effects and perhaps thereby improve their efficacy at symptom control.
Similar content being viewed by others
References
Abramson S, Korchak H, Ludewig R, Edelson H, Haines K, et al. Modes of action of aspirin like drugs. Proceedings of the National Academy of Sciences of the United States of America 82: 7227–7231, 1985
Abramson S, Weissman G. The mechanism of action of nonsteroidal anti-inflammatory drugs. Arthritis and Rheumatism 32: 1–9, 1989
Agrawal N, Roth S, Graham DY, White RH, Germani B, et al. Misoprostol compared with sucralfate in the prevention of nonsteroidal anti-inflammatory drug induced gastric ulceration. Annals of Internal Medicine 115: 195–200, 1991
Altmann GG. Changes in the mucosa of the small intestine following methotrexate administration or abdominal X-irradiation. American Journal of Anatomy 140: 263–280, 1974
Andreone P, Baraldini M, Cursaro C, Bortoluzzi F, Micaletti E, et al. Prostanoids in peptic ulcer: no evidence of their lack. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 393, Medicine Group (UK) Ltd, Abingdon, 1990
Armstrong CP, Blower AL. Nonsteroidal anti-inflammatory drugs and life threatening complications of peptic ulceration. Gut 28: 527–532, 1987
Banergee RK. Nonsteroidal anti-inflammatory drugs inhibit gastric peroxidase activity. Biochimica et Biophysica Acta 1034: 275–280, 1990
Bardhan KD, Griffin WM, Fenn GC, Shield MJ, Bjarnason IT. Prevention and reversal of NSAID damage by misoprostol. Abstract. Gastroenterology 100: 30, 1991
Beardon PHG, Brown SV, McDevitt DG. Gastrointestinal events in patients prescribed NSAIDs: a controlled study using record linkage in Tayside. Quarterly Journal of Medicine 71: 497–505, 1989
Berkowitz JM, Rogenes PR, Sharp JT, Warner CW. Ranitidine protects against gastroduodenal mucosal damage associated with chronic aspirin therapy. Archives of Internal Medicine 147: 2137–2139, 1987
Bernerson B, Johnsen R, Straume B, Bushol PG, Jenssen TG, et al. Towards a true prevalence of peptic ulcer: the Sorreisa gastrointestinal disorder study. Gut 31: 989–992, 1990
Bianchi Porro G, Pace F, Caruso I. Why are NSAIDs important in peptic ulceration? Alimentary Pharmacology and Therapeutics 1(suppl. 1): 540–547, 1987
Bianchi Porro G, Santalucia F, Petrillo M. Omeprazole against sucralfate in the treatment of NSAID induced gastric and duodenal lesions. Abstract. Gut 31: 1175, 1990
Bijlsma JWJ. Treatment of NSAID induced gastrointestinal lesions with cimetidine: an international multi-centre collaborative study. Alimentary Pharmacology and Therapeutics 2(suppl. 2): 85–96, 1988
Bjarnason I. Non-steroidal antiinflammatory drug induced small intestinal inflammation in man. In Pounder R (Ed.) Recent advances in gastroenterology, Vol. 7, pp. 23–46, Churchill Livingstone, Edinburgh, 1988
Bjarnason I, Turner Stokes L, Levi AJ, Gumpel MJ, Hayllar JS. The role of bacteria in the pathogenesis of NSAID induced enteropathy. Abstract. Gut 31: 593, 1990
Bolten W, Brodenfeldt R. Efficacy of misoprostol in the therapy of NSAID-induced symptoms and lesions of the upper GI tract under continued NSAID medication. Abstract no. 104. Clinical and Experimental Rheumatology 8(suppl. 4): 59, 1990
Bonnevic O. Survival in peptic ulcer. Gastroenterology 75: 1055–1060, 1978
Bradley JD, Brandt KD, Katz BP, Kalasinski LA, Regan SI. Comparison of an anti-inflammatory dose of ibuprofen, an analgesic dose of ibuprofen and acetaminophen in the treatment of patients with osteoarthritis of the knee. New England Journal of Medicine 325: 87–91, 1991
Brune K, Rainsford KD, Schweitzer A. Biodistribution of mild analgaesics. British Journal of Clinical Pharmacology (Suppl. 10): 279s–284s, 1980
Carson JL, Strom BL, Morse ML. The relative gastrointestinal toxicity of the nonsteroidal anti-inflammatory drugs. Archives of Internal Medicine 147: 1054–1059, 1987a
Carson JL, Strom BL, Soper KA, West SL, Morse ML. The association of NSAIDs with upper gastrointestinal tract bleeding. Archives of Internal Medicine 147: 85–88, 1987b
Caruso I, Bianchi Porro G. Gastroscopic evaluation of anti-inflammatory agents. British Medical Journal 280: 75–78, 1980
Cockel R. NSAIDs: should every pack carry a government health warning? Gut 28: 515–518, 1987
Collins AJ, DuToit JA. Upper gastrointestinal findings and faecal occult blood in patients with rheumatic diseases taking nonsteroidal anti-inflammatory agents. British Journal of Rheumatology 26: 295–298, 1987
Dajani EZ. Perspective on the gastric antisecretory effects of misoprostol in man. Prostaglandins 33 (suppl.): 68–77, 1987
Daneshmend TK, Stein AG, Bhaskar NK, Hawkey CJ. Abolition by omeprazole of aspirin induced mucosal injury in man. Gut 31: 514–517, 1990
Davies J, Collins AJ, St J Dixon A. The influence of cimetidine on peptic ulcer in patients with arthritis taking anti-inflammatories. British Journal of Rheumatology 25: 54–58, 1986
de Rossi A, Brodenfeldt R, Bolten W. Prevention of NSAID induced GI symptoms and lesion by a low daily dose of misoprostol. Abstract no. 105. Clinical and Experimental Rheumatology 8(suppl. 4): 58, 1990
Dorta G, Saraga E, Nicolet M, Schneeg JF, Vouillamoz D, et al. Influence of omeprazole on healing and prevention of gastroduodenal mucosal lesions during administration of NSAIDs. Abstract no. 293. Gastroenterology 100: 55, 1991
Dreyling KW, Hoppe U, Kleine A, Kozuschek W, Peskar BM. Leukotriene formation in the gastrointestinal tract and the effects of anti-inflammatory drugs. In Rainsford & Velo (Eds) Side effects of anti-inflammatory drugs, Part 2: studies in major organ systems, pp. 101–111, MTP Press, Lancaster, 1985
Duggan JM. Aspirin in chronic gastric ulcer: an Australian experience. Gut 17: 378–384, 1976
Editorial. Misoprostol, ulcer prophylaxis at what cost? Lancet 2: 1293–1294, 1988
Ehsanullah RSB, Page MC, Tildesley G, Wood JR. Prevention of gastroduodenal damage induced by nonsteroidal anti-inflammatory drugs: controlled trial of ranitidine. British Medical Journal 297: 1017–1021, 1988
Elliott SL, Yeomans ND, Buchanan RRC, Duckham MV, Boyden KN, et al. Long term effects of misoprostol on gastropathy induced by NSAIDs. Abstract no. 102. Clinical and Experimental Rheumatology 8(suppl. 4): 58, 1990
Euler AR, Safdi M, Rao J, et al. A report of three multicenter trials evaluating arbaprostil in arthritic patients with ASA/NSAID gastric mucosal damage. Gastroenterology 98: 1549–1557, 1990
Faulkner G, Prichard P, Somerville K, Langman MJS. Aspirin and bleeding peptic ulcers in the elderly. British Medical Journal 297: 1311–1313, 1988
Freeman TC, Playford RJ, Quinn C, Beardshall K, Poulter L, et al. Pancreatic secretory trypsin inhibitor in the gastrointestinal mucosa and gastric juice. Gut 31: 1318–1323, 1990
Fry J. Peptic ulcer: a profile. British Medical Journal 2: 809–812, 1964
Fries JF, Miller SR, Williams CA, Hubert HB, Block DA. Towards an epidemiology of gastropathy associated with NSAID use. Gastroenterology 96: 647–655, 1989
Gabriel SE, Bombardier C. NSAID induced ulcers: an emerging epidemic? Journal of Rheumatology 17: 1–4, 1990
Geis GS, Stead H. Prevention of diclofenac induced gastroduodenal mucosal damage by Cytotec. Abstract no. 101. Clinical and Experimental Rheumatology 8(suppl. 4): 58, 1990
Goodlad RA, Madgwick AJ, Moffatt MR, Levin S, Allan JL, et al. Prostaglandins and the gastric epithelium: the effects of misoprostol on gastric epithelial cell proliferation in the dog. Gut 30: 316–321, 1989
Graham DY, Campylobacter pylori and peptic ulcer disease. Gastroenterology 96 (suppl.): 615–625, 1989
Graham DY. The relationship between NSAID use and peptic ulcer disease. Gastroenterology Clinics of North America 19: 171–182, 1990
Graham DY, Agrawal NM, Roth S. Prevention of nonsteroidal anti-inflammatory drug induced gastric ulceration with misoprostol. Lancet 2: 1277–1280, 1988a
Graham DY, Klein PD, Opekun AR, Boutton TW. Effect of age on the frequency of active HP infection diagnosed by the 13C urea breath test in normal subjects and in patients with peptic ulcer disease. Journal of Infectious Diseases 157: 777–780, 1988b
Graham DY, Lacey Smith J, Spjut HJ, Torres E. Gastric adaptation: studies in humans during continuous aspirin administration. Gastroenterology 95: 327–333, 1988c
Graham DY, Lidsky MD, Cox AM, Evans DJ, Evans DG, et al. Long term nonsteroidal anti-inflammatory drug use and helicobacter pylori infection. Gastroenterology 100: 1653–1657, 1991a
Graham DY, Smith JL. Aspirin and the stomach. Annals of Internal Medicine 104: 390–398, 1986
Graham DY, Smith JL, Holmes GI, Davies RO. Nonsteroidal antiinflammatory effect of sulindac sulfoxide and sulfide on the gastric mucosa. Clinical Pharmacology and Therapeutics 38: 65–70, 1985
Graham DY, Stromatt SC, Jascewski R, White RH, Triadafilopoulos G. Prevention of duodenal ulcer in arthritics who are chronic NSAID users: a multicenter trial of the role of misoprostol. Abstract no. 239. Gastroenterology 100: 75, 1991b
Grier CE, Palmer RH, Wallin BA, Wetherington JD, Fox MY. Increased risk of gastrointestinal ulceration in elderly NSAID patients and its reduction by cimetidine. Abstract. Gastroenterology 100: 8, 1991
Griffin MR, Piper JM, Dougherty JR, Snowden M, Ray WA. Nonsteroidal anti-inflammatory drug use and increased risk for peptic ulcer disease in elderly persons. Annals of Internal Medicine 114: 257–263, 1991
Griffin MR, Ray WA, Schaffner W. NSAID use and death from peptic ulcer in elderly persons. Annals of Internal Medicine 109: 359–364, 1988
Gumbiner B. Structure, biochemistry and assembly of epithelial tight junctions. American Journal of Physiology 253: 749–758, 1987
Hawkey CJ. Synthesis of prostaglandin E2, thromboxane B2 and prostaglandin catabolism in gastritis and gastric ulcer. Gut 27: 1484–1492, 1986
Hawkey CJ, Kemp RT, Walt RP, Bhaskar NH, Davies J, et al. Evidence that adaptive cytoprotection in rat is not mediated by prostaglandins. Gastroenterology 94: 948–954, 1988
Hayllar JS, Somasundaram S, Sarathchandra P, Levi AJ, Bjarnason I. Early cellular events in the pathogenesis of NSAID enteropathy in the rat. Abstract. Gastroenterology 100: 216, 1991
Henriksson AEK, Uribe A, Johansson C. The effect of a PG1 analogue on the gastroduodenal mucosa in patients with rheumatoid arthritis. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 392, Medicine Group (UK) Ltd, Abingdon, 1990
Hillman AL, Bloom BS. Economic effects of prophylactic use of misoprostol to prevent gastric ulcer in patients taking NSAIDs. Archives of Internal Medicine 149: 2061–2065, 1989
Hirose H, Takenchi K, Okabe S. Effect of indomethacin on gastric mucosal blood flow around acetic acid induced ulcers in the rat. Gastroenterology 100: 1259–1265, 1991
Hoftiezer JW, Silvoso GR, Burks M, Ivey KJ. Comparison of the effects of regular and enteric coated aspirin on the gastroduodenal mucosa. Lancet 2: 609–612, 1980
Holvoet J, Terriere L, Van Hee W, Verbist L, Fierens E, et al. The relation of upper gastrointestinal bleeding to NSAID and aspirin: a case control study. Gut 32: 730–734, 1991
Hudson N, Everitt S, Edwards T, Filipowicz B, Hawkey CJ. Elevation of gastric mucosal LTB4 levels in patients on longstanding NSAID treatment. Abstract. Gastroenterology 100: 279, 1991
Iglehart IW, Edlow DW, Mills L, Morrison SA, Hochberg MC. The presence of campylobacter pylori in NSAID associated gastritis. Journal of Rheumatology 16: 599–603, 1989
Jachuck SJ. Too much to swallow. British Medical Journal 303: 360, 1991
Jick H, Feld AD, Perera DR. Certain NSAIDs and hospitalisation for oesophageal bleeding in the elderly. Pharmacotherapy 5: 280–284, 1985
Johnsen R, Bernersen B, Straume B, Forde OH, Bostad L, et al. Prevalence of endoscopic and histologic findings in subjects with and without dyspepsia. British Medical Journal 302: 749–752, 1991
Jones DB, Howden CW, Burget DW, Kerr GD, Hunt RH. Acid suppression in duodenal ulcer: a meta-analysis to define optimal dosing with anti-secretory drugs. Gut 28: 1120–1127, 1987
Kauffman G. Aspirin-induced gastric mucosal injury: lessons learned from animal models. Gastroenterology 96: 606–614, 1989
Kimmey MB, Silverstein FE. The role of H2 receptor blockers in the prevention of gastric injury from NSAIDs. American Journal of Medicine 84(suppl. 2A): 49–52, 1988
Knill-Jones R, Drummond M, Kohli H, Davies L. Economic evaluation of gastric ulcer prophylaxis in patients with arthritis receiving nonsteroidal anti-inflammatory drugs. Postgraduate Medical Journal 66: 639–649, 1990
Konturek SJ, Brzozowski T, Amon I. Nocloprost, stable PGE2 analogue with unique local gastroprotective and ulcer healing activity. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 12, Medicine Group (UK) Ltd, Abingdon, 1990
Kurata JH, Abbey DE. The effect of chronic aspirin use on duodenal and gastric ulcer hospitalisations. Journal of Clinical Gastroenterology 12: 260–266, 1990
Lacy ER. Prostaglandins and histological changes in the gastric mucosa. Digestive Diseases and Sciences 30 (suppl.): 83–94, 1985
Laine L, Marinsorensen M, Weinstein WM. Helicobacter pylori prevalence and mucosal injury in gastric ulcer: relation to chronic NSAID ingestion. Abstract. Gastroenterology 100: 278, 1991
Lambert R, Delmas D, Elhevenaux C, Capron MH. Prevention of NSAID induced gastric and duodenal lesions. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 606, Medicine Group (UK) Ltd, Abingdon, 1990
Lanas A, Sakar MC, Hirschowitz BI. Objective testing reveals a very strong association of acetyl salicylic acid with upper and lower gastrointestinal bleeding. Abstract. Gastroenterology 100: 106, 1991
Lancaster Smith M, Jaderberg ME, Jackson DA. Ranitidine in the treatment of NSAID associated gastric and duodenal ulcers. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 457, The Medicine Group (UK) Ltd, Abingdon, 1990
Langman MJS. Treating ulcers in patients receiving anti-arthritic drugs. Quarterly Journal of Medicine 73: 1089–1091, 1989
Lanza FL. A double blind study of the protective effect of misoprostol on lesions of the gastric and duodenal mucosa induced by oral administration of tolmetin in healthy subjects. Digestive Diseases and Science 31(suppl. 2): 131–136, 1986
Lanza FL, Graham DY, Davis RE, Rack MF. Endoscopic evaluation of cimetidine and sucralfate for prevention of naproxen-induced gastroduodenal injury: effect of scoring method. Digestive Diseases and Sciences 35: 1494–1499, 1990
Lanza FL, Kaye G, Nelson R. An endoscopic evaluation of the effects of nonsteroidal anti-inflammatory agents on the gastric mucosa. Gastrointestinal Endoscopy 21: 103–105, 1975
Lanza FL, Royer GL, Nelson RS, Chenn TT, Seckman CC, et al. The effects of ibuprofen, indomethacin, aspirin, naproxen and placebo on the gastric mucosa of normal volunteers. Digestive Diseases and Science 24: 823–828, 1979
Leonards JR, Levy G, Niemczura R. GI blood loss during prolonged aspirin administration. New England Journal of Medicine 289: 1020–1022, 1973
Levi S, Goodlad RA, Lee CY, Stamp G, Walport MJ, et al. Inhibitory effect of NSAID on mucosal cell proliferation associated with gastric ulcer healing. Lancet 2: 840–843, 1990
Levi S, Griffin WMG, Fenn GC, Shield MJ, Hodgson HJK. Prevention of non-steroidal anti-inflammatory drug induced gastric and duodenal damage by misoprostol: a randomised double-blind placebo-controlled study. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 451, Medicine Group (UK) Ltd, Abingdon, 1990
Ligumsky M, Golanska EM, Hansen DG, Kauffman GL. Aspirin can inhibit gastric mucosal cyclo-oxygenase without causing lesions in the rat. Gastroenterology 84: 756–761, 1983
MacDonald WC. Correlation of mucosal histology and aspirin intake in chronic gastric ulcer. Gastroenterology 65: 381–389, 1973
Marshall BJ, Goodwin CS, Warren RJ, Murray R, Blincow ED, et al. Prospective double blind trial of duodenal ulcer relapse after eradication of campylobacter pylori. Lancet 2: 1437–1442, 1988
Martin BK. Accumulation of drug anions in gastric mucosal cells. Nature 198: 896–897, 1963
McCarthy DM. NSAID induced ulcers: management by traditional therapies. Gastroenterology 96: 662–674, 1989
McDougall P, Markham AS, Cameron I, Sweetman AJ. The mechanism of inhibition of mitochondrial oxidative phosphorylation by the nonsteroidal anti-inflammatory agent diflusinal. Biochemical Pharmacology 32: 2595–2598, 1983
McIntosh JH, Fung CS, Berry G, Piper DW. Smoking, NSAIDs and acetaminophen in gastric ulcer. American Journal of Epidemiology 128: 761–770, 1988
Menguy R, Desbaillets L, Masters YF. Mechanism of stress ulcer: influence of hypovolaemic shock on energy metabolism in the gastric mucosa. Gastroenterology 66: 46–55, 1974
Metzger WJ, McAdam L, Bluestone R. Acute gastric mucosal injury during continuous or interrupted aspirin ingestion in humans. American Journal of Digestive Diseases 21: 963–968, 1976
Meyer RA, Mcginley D, Posalaky Z. Effects of aspirin on tight junction structure of canine gastric mucosa. Gastroenterology 91: 351–359, 1986
Mizokami Y, Veno M, Morita S, Ito Y, Shiozawa K, et al. NSAID gastropathy in rheumatoid arthritis patients. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 978, Medicine Group (UK) Ltd, Abingdon, 1990
Moncada S, Vane JRS. The mode of action of aspirin like drugs. Advances in Internal Medicine 24: 1–22, 1979
Naschitz JE, Yeshurun D, Odeh M, Bassan H, Rosner I, et al. Overt GI bleeding in the course of chronic low dose aspirin administration for secondary prevention of arterial occlusive disease. American Journal of Gastroenterology 85: 408–411, 1990
Oddsson E, Gudjonsson H, Thjodleifsson B. Protective effect of omeprazole or ranitidine against naproxen induced damage to human gastroduodenal mucosa. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 22, Medicine Group (UK) Ltd, Abingdon, 1990
O’Laughlin JC, Hofteizer JW, Ivey KJ. Effect of aspirin on the human stomach in normals, endoscopic comparison of damage produced at 1 hour, 24 hours and 2 weeks after administration. Scandinavian Journal of Gastroenterology 16(suppl. 67): 211–214, 1981
O’Laughlin JC, Silvoso GK Ivey KJ. Resistance to medical therapy of gastric ulcers in rheumatic disease patients taking aspirin. Digestive Diseases and Sciences 27: 976–980, 1982
Playford RJ, Batten J, Freeman T, Fenn GC, Calam J. Misoprostol stimulates the gastric secretion of pancreatic secretory trypsin inhibitor: a newly recognised protective mechanism. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 448, Medicine Group (UK) Ltd, Abingdon, 1990
Pugh S, Williams SE, Levin MR, Ishaque M, Barton TP, et al. Duodenal and antral mucosal PGE2 synthesis in a study of normal subjects and all stages of duodenal ulcer disease treated by H2-receptor antagonists. Gut 30: 161–165, 1989
Quinn CM, Price AB, Bjarnason I. Endoscopic and microscopic findings in symptomatic and asymptomatic patients on long term non-steroidal anti-inflammatory drugs. Abstract. Gastroenterology 98: 109, 1990
Rachmilewitz D. The role of H2-receptor antagonists in the prevention of NSAID-induced gastro-intestinal damage. Alimentary Pharmacology and Therapeutics 2(suppl. 2): 65–73, 1988
Rainsford KD. Prevention of indomethacin induced GI ulcerastion in rats by glucose citrate formulations: the role of ATP in mucosal defences. British Journal of Rheumatology 26: 81, 1987
Raskin J, White R, Jaszewski R. Double blind controlled study of the efficacy and safety of misoprostol and ranitidine in the prevention of NSAID induced gastric ulcer and upper gastrointestinal symptoms. Abstract. Digestion 41(suppl. 1): 50–51, 1991
Redfern JS, Blair AJ, Lee E, Feldman M. Gastrointestinal ulcer formation in rabbits immunised with prostaglandin E2. Gastroenterology 93: 744–752, 1987
Robert A. Cytoprotection by prostaglandins. Gastroenterology 77: 761–767, 1979
Robert A, Lancaster C, Davis JP, Field SO, Wickremasingh AJ, Thornburgh BA. Cytoprotection by prostaglandins occurs in spite of penetration of absolute ethanol into the gastric mucosa. Gastroenterology 88: 328–333, 1985
Robert A, Nezamis JE, Lancaster C, Davis JP, Field SO, et al. Mild irritants prevent gastric necrosis through adaptive cytoprotection mediated by prostaglandins. American Journal of Physiology 245: G113–G121, 1983
Robins PG. Ultrastructural observations on the pathogenesis of aspirin induced gastric erosions. British Journal of Experimental Pathology 61: 497–504, 1980
Robinson MC, Griffin JW, Bowers J, Kogan FJ, Kogut DG, et al. The effect of ranitidine on gastroduodenal damage induced by NSAIDs. Digestive Diseases and Sciences 34: 424–428, 1989
Roth S, Agrawal N, Mahowald M, Montega H, Robbins D, et al. Misoprostol heals gastroduodenal injury in patients with rheumatoid arthritis receiving aspirin. Archives of Internal Medicine 149: 775–779, 1989
Roth SH. NSAID and gastropathy: a rheumatologist’s review. Journal of Rheumatology 15: 912–919, 1988
Roth SH, Bennett RE. NSAID gastropathy recognition and response. Archives of Internal Medicine 147: 2093–2100, 1987
Roth SH, Bennett RE, Mitchell CS, Hartman RJ. Cimetidine therapy in NSAID gastropathy: double blind long term evaluation. Archives of Internal Medicine 147: 1798–1801, 1987
Saggioro A, Alvisi V, Blasi A. Misoprostol prevents NSAID induced gastroduodenal lesions in patients with osteoarthritis and rheumatoid arthritis. Italian Journal of Gastroenterology 23: 119–123, 1991
Samuelsson B. Leukotrienes: mediators of immediate hypersensitivity reactions and inflammation. Science 220: 568–575, 1983
Sanduja R, Loose-Mitchell D, Tsai AL, Wu KK. Inhibition of prostaglandin H synthase expression by aspirin and sodium salicylate: a novel pharmacological mechanism. International Conference on Prostaglandins and Related Compounds, Florence, May 28–June 1 1990. Abstract no. 7, 1990
Sato N, Kawano S, Fukuda M, Tsuji S, Kamada T. Misoprostol induced changes in gastric mucosal hemodynamics. American Journal of Medicine 83(suppl. 1A): 15–21, 1987
Schoen RT, Vender RJ. Mechanisms of nonsteroidal anti-inflammatory drug-induced gastric damage. American Journal of Medicine 86: 449–458, 1989
Shield MJ, Fenn GC, Kiff P, Stead H, Geis GS. Do differential rates of NSAID associated gastroduodenal damage exist — the results of a large cohort study in rheumatoid arthritis and osteoarthritis patients. Abstract no. 10. British Journal of Rheumatology 29(suppl. 1A): 5, 1990
Smallcross TM, Wyatt JI, Rathbone BJ, Heatley RV. Non-steroidal anti-inflammatory drugs, hiatus hernia and helicobacter pylori in patients with oesophageal ulceration. British Journal of Rheumatology 29: 288–290, 1990
Smith MJH, Dawkins PD. Salicylates and enzymes. Journal of Pharmacy and Pharmacology 23: 729–744, 1971
Soil AH, Kurata J, McGuigan JE. Ulcers, nonsteroidal anti-inflammatory drugs and related matters. Gastroenterology 96: 561–568, 1989
Soll AH. Nonsteroidal anti-inflammatory drugs and peptic ulcer disease. Annals of Internal Medicine 114: 307–319, 1991
Spenney JG, Bhown M. Effects of acetyl salicylic acid on gastric mucosa II. Gastroenterology 73: 995–999, 1977
Stalnikowicz R, Pollak D, Eliakim A. Cimetidine decreases indomethacin induced duodenal damage in patients with acute musculoskeletal disorders. Gut 29: 1578–1582, 1988
Sun DC, Roth SH, Mitchell CS, England DW. Upper gastrointestinal disease in rheumatoid arthritis. American Journal of Digestive Disease 19: 405, 1974
Tarnawski A, Brzozowski T, Sarfeh J, Krause WJ, Ulich TR, et al. Prostaglandin protection of human isolated gastric glands against indomethacin and ethanol injury. Journal of Clinical Investigation 81: 1081–1089, 1988
Tsai BS, Keith RH, Collins PW, Gasiecki AF, Gaginella TS. Evidence that misoprostol free acid mediates the inhibitory activity of misoprostol on human neutrophil Superoxide production: involvement of a subtype prostaglandin E receptor. Abstract. Gastroenterology 100: 621, 1991
Tsai BS, Kessler LK, Schoenhard G, Collins PW, Bauer RF. Demonstration of specific E type prostaglandin receptors using enriched preparations of canine parietal cells and [3H] misoprostol free acid. American Journal of Medicine 83(suppl. 1A): 9–15, 1987
Tympner F. Gastroscopic findings after therapy with nonsteroidal antiinflammatory drugs. Journal of Rheumatology 40: 179–181, 1981
Vaananen PM, Meddings JB, Wallace JL. The role of reactive oxygen metabolites in experimental NSAID gastropathy. Abstract. Gastroenterology 100: 240, 1991
Vane JR, Botting RM. The mode of action of anti-inflammatory drugs. Postgraduate Medical Journal 66(suppl. 4): 2–17, 1990
Walen A, Bader JP, Classen M, Lamers CBHW, Piper DW, et al. Effect of omeprazole and ranitidine on ulcer healing and relapse rates in patients with benign gastric ulcer. New England Journal of Medicine 320: 69–75, 1989
Wallace JL. The role of leukotrienes and endothelin in gastric mucosal injury. Abstracts of the World Congresses of Gastroenterology, Sydney, pp. 21, Medicine Group (UK) Ltd, Abingdon, 1990
Wallace JL, Arfors KE, McKnight GW. A monoclonal antibody against the CD 18 leucocyte adhesion molecule prevents indomethacin induced gastric damage in the rabbit. Gastroenterology 100: 878–883, 1991a
Wallace JL, Keenan CM, Cucula M, Mugridge K, Parente L. The prevention of NSAID gastropathy by IL-1B: the role of neutrophils and acid secretion. Abstract. Gastroenterology 100: 279, 1991b
Wallace JL, Keenan CM, Grange DN. Gastric ulceration induced by NSAIDs is a neutrophil dependent process. American Journal of Physiology 259: 462–467, 1990
Wallin BA, Grier CE, Fox MJ, McCafferty JP, Wetherington JD, et al. Prevention of NSAID induced ulcers with cimetidine: results of a double blind placebo controlled trial. Abstract: Gastroenterology 98: 146, 1990
Walt R, Katschinski B, Logan R, Ashley J, Langman M. Rising frequency of ulcer perforation in elderly people in the UK. Lancet 1: 489–492, 1986
Weissman G. Aspirin. Scientific American 264: 58–64, 1991
Whitehouse MW. Uncoupling of oxidative phosphorylation by some aryl acetic drugs. Nature 201: 629–630, 1964
Wilson RG, Smith AN, Macintyre IMC. Complications of diverticular disease and NSAIDs: a prospective study. British Journal Of Surgery 77: 1103–1104, 1990
Yanagawa A. Prophylactic efficacy of ranitidine against gastroduodenal damage from NSAID. A randomised placebo controlled study. Abstracts of the World Congresses of Gastroenterology, Sydney, p. 996, The Medicine Group (UK) Ltd, Abingdon, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hayllar, J., Macpherson, A. & Bjarnason, I. Gastroprotection and Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). Drug-Safety 7, 86–105 (1992). https://doi.org/10.2165/00002018-199207020-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-199207020-00002