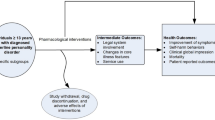
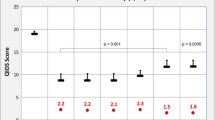
Abstract
Real-world, effectiveness studies add an important new dimension to the evaluation of the benefits of individual antipsychotics. Efficacy studies have already shown the unique effectiveness of clozapine, and suggested improved outcomes for olanzapine compared with some atypical antipsychotics and a reduced tendency to produce acute and chronic movement disorders for atypical compared with typical drugs. Recent effectiveness studies largely confirm these prior observations. The CATIE (Clinical Antipsychotic Trials of Intervention Effectiveness), CUtLASS (Cost Utility of the Latest Anti-psychotic Drugs in Schizophrenia Study) and SOHO (Schizophrenia Outpatient Health Outcomes) programmes confirmed the superiority of clozapine over other antipsychotics; CATIE and SOHO also confirmed olanzapine as probably the second most effective antipsychotic. Effectiveness studies have confirmed the high incidence of adverse metabolic effects with clozapine, olanzapine and (with less certainty) quetiapine but the ZODIAC (Ziprasi-done Observational Study of Cardiac Outcomes) study found no excess cardiovascular events or deaths for olanzapine compared with ziprasidone. Prior observations on reduced frequency of movement disorders for second-generation versus first-generation antipsychotics were also largely (but not uniformly) supported.
Overall, recent real-world studies have done much to confirm prior ob-servations from efficacy-based randomized, controlled trials.








Similar content being viewed by others
References
Stroup TS, McEvoy JP, Swartz MS, et al. The National Institute of Mental Health Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) project: schizophrenia trial design and protocol development. Schizophr Bull 2003; 29(1): 15–31
Strom BL, Faich GA, Reynolds RF, et al. The Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC): design and baseline subject characteristics. J Clin Psychiatry 2008; 69(1): 114–21
Stroup TS, Lieberman JA, McEvoy JP, et al. Effectiveness of olanzapine, quetiapine, risperidone, and ziprasidone in patients with chronic schizophrenia following discontinuation of a previous atypical antipsychotic. Am J Psychiatry 2006; 163(4): 611–22
Lieberman JA, McEvoy JP, Swartz MS, et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 2005; 353: 1209–23
McEvoy JP, Lieberman JA, Stroup TS, et al. Effectiveness of clozapine versus olanzapine, quetiapine, and risperidone in patients with chronic schizophrenia who did not respond to prior atypical antipsychotic treatment. Am J Psychiatry 2006; 163(4): 600–10
Stroup TS, Lieberman JA, McEvoy JP, et al. Results of phase 3 of the CATIE schizophrenia trial. Schizophr Res 2009; 107(1): 1–12
Miller DD, Caroff SN, Davis SM, et al. Extrapyramidal side-effects of antipsychotics in a randomised trial. Br J Psychiatry 2008; 193(4): 279–88
Meyer JM, Davis VG, Goff DC, et al. Change in metabolic syndrome parameters with antipsychotic treatment in the CATIE Schizophrenia Trial: prospective data from phase 1. Schizophr Res 2008; 101(1–3): 273–86
Essock SM, Covell NH, Davis SM, et al. Effectiveness of switching antipsychotic medications. Am J Psychiatry 2006; 163(12): 2090–5
Rosenheck RA, Davis S, Covell N, et al. Does switching to a new antipsychotic improve outcomes? Data from the CATIE Trial. Schizophr Res 2009; 107(1): 22–9
Rosenheck RA, Leslie DL, Sindelar J, et al. Cost-effectiveness of second-generation antipsychotics and perphenazine in a randomized trial of treatment for chronic schizophrenia. Am J Psychiatry 2006; 163(12): 2080–9
Jones PB, Barnes TR, Davies L, et al. Randomized controlled trial of the effect on quality of life of second- versus first-generation antipsychotic drugs in schizophrenia: Cost Utility of the Latest Antipsychotic Drugs in Schizophrenia Study (CUtLASS 1). Arch Gen Psychiatry 2006; 63(10): 1079–87
Davies LM, Lewis S, Jones PB, et al. Cost-effectiveness of first- v second-generation antipsychotic drugs: results from a randomised controlled trial in schizophrenia responding poorly to previous therapy. Br J Psychiatry 2007; 191: 14–22
Davies LM, Barnes TR, Jones PB, et al. A randomized controlled trial of the cost-utility of second-generation antipsychotics in people with psychosis and eligible for clozapine. Value Health 2008; 11(4): 549–62
Lewis SW, Barnes TR, Davies L, et al. Randomized controlled trial of effect of prescription of clozapine versus other second-generation antipsychotic drugs in resistant schizophrenia. Schizophr Bull 2006; 32(4): 715–23
Heinrichs DW, Hanlon TE, Carpenter WT, et al. The Quality of Life Scale: an instrument for rating the schizophrenic deficit syndrome. Schizophr Bull 1984; 10(3): 388–98
Kind P. The EuroQoL instrument: an indicator of health related quality of life and pharmacoeconomics on clinical trials. In: Spilker B, editor. Quality of life and pharmacoeconomics in clinical trials. New York: Lippincott-Raven, 1996: 191–202
Endicott J, Spitzer RL, Fleiss JL, et al. The global assessment scale: a procedure for measuring overall severity of psychiatric disturbance. Arch Gen Psychiatry 1976; 33(6): 766–71
Haro JM, Edgell ET, Frewer P, et al. The European Schizophrenia Outpatient Health Outcomes Study: baseline findings across country and treatment. Acta Psychiatr Scand Suppl 2003; (416): 7–15
Guy W. The Clinical Global Impressions Scale. In: Guy W, editor. ECDEU assessment manual for psychopharma-cology [rev. ed.]. Rockville (MD): National Institute of Mental Health, 1976: 157–69
Haro JM, Edgell ET, Novick D, et al. Effectiveness of antipsychotic treatment for schizophrenia: 6-month results of the Pan-European Schizophrenia Outpatient Health Outcomes (SOHO) study. Acta Psychiatr Scand 2005; 111(3): 220–31
Dossenbach M, Rango-Davila C, Silva IH, et al. Response and relapse in patients with schizophrenia treated with olanzapine, risperidone, quetiapine, or haloperidol: 12-month follow-up of the Intercontinental Schizophrenia Outpatient Health Outcomes (IC-SOHO) study. J Clin Psychiatry 2005; 66(8): 1021–30
Haro JM, Kamath SA, Ochoa S, et al. The Clinical Global Impression-Schizophrenia scale: a simple instrument to measure the diversity of symptoms present in schizophrenia. Acta Psychiatr Scand Suppl 2003; (416): 16–23
Haro JM, Novick D, Belger M, et al. Antipsychotic type and correlates of antipsychotic treatment discontinuation in the outpatient treatment of schizophrenia. Eur Psychiatry 2006 Jan; 21(1): 41–7
Novick D, Haro JM, Suarez D, et al. Symptomatic remission in previously untreated patients with schizophrenia: 2-year results from the SOHO study. Psychopharmacology (Berl) 2007; 191(4): 1015–22
Haro JM, Novick D, Suarez D, et al. Antipsychotic treatment discontinuation in previously untreated patients with schizophrenia: 36-month results from the SOHO study. J Psychiatr Res 2009; 43(3): 265–73
Haro JM, Novick D, Suarez D, et al. Remission and relapse in the outpatient care of schizophrenia: three-year results from the Schizophrenia Outpatient Health Outcomes study. J Clin Psychopharmacol 2006; 26(6): 571–8
Alonso J, Croudace T, Brown J, et al. Health-related quality of life (HRQL) and continuous antipsychotic treatment: 3-year results from the Schizophrenia Health Outcomes (SOHO) study. Value Health 2009; 12(4): 536–43
Tenback DE, van Harten PN, Slooff CJ, et al. Effects of antipsychotic treatment on tardive dyskinesia: a 6-month evaluation of patients from the European Schizophrenia Outpatient Health Outcomes (SOHO) Study. J Clin Psychiatry 2005; 66(9): 1130–3
Tenback DE, van Harten PN, Slooff CJ, et al. Evidence that early extrapyramidal symptoms predict later tardive dyskinesia: a prospective analysis of 10,000 patients in the European Schizophrenia Outpatient Health Outcomes (SOHO) Study. Am J Psychiatry 2006; 163(8): 1438–40
Novick D, Haro JM, Perrin E, et al. Tolerability of outpatient antipsychotic treatment: 36-month results from the European Schizophrenia Outpatient Health Outcomes (SOHO) study. Eur Neuropsychopharmacol 2009; 19(8): 542–50
Bitter I, Basson BR, Dossenbach MR. Antipsychotic treatment and sexual functioning in first-time neuroleptic-treated schizophrenic patients. Int Clin Psychopharmacol 2005; 20(1): 19–21
Dossenbach M, Dyachkova Y, Pirildar S, et al. Effects of atypical and typical antipsychotic treatments on sexual function in patients with schizophrenia: 12-month results from the Intercontinental Schizophrenia Outpatient Health Outcomes (IC-SOHO) study. Eur Psychiatry 2006; 21(4): 251–8
Strom BL, Eng SM, Faich G, et al. Comparative mortality associated with ziprasidone and olanzapine in real-world use among 18,154 patients with schizophrenia: the Ziprasidone Observational Study of Cardiac Outcomes (ZODIAC). Am J Psychiatry 2011; 168(2): 193–201
Kane J, Honigfeld G, Singer J, et al. Clozapine for the treatment-resistant schizophrenic: a double-blind comparison with chlorpromazine. Arch Gen Psychiatry 1988; 45: 789–96
Leucht S, Komossa Rummel-Kluge C, et al. A metaanalysis of head-to-head comparisons of second-generation antipsychotics in the treatment of schizophrenia. Am J Psychiatry 2009; 166: 152–63
Allison D, Mentore J, Heo M, et al. Antipsychotic-induced weight gain: a comprehensive research synthesis. Am J Psychiatry 1999; 156: 1686–96
Leucht S, Corves C, Arbter D, et al. Second-generation versus first-generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet 2009; 373(9657): 31–41
Beasley C, Dellva M, Tamura R. Randomised double-blind comparison of the incidence of tardive dyskinesia in patients with schizophrenia during long-term treatment with olanzapine or haloperidol. Br J Psychiatry 1999; 174: 23–30
McCue RE, Waheed R, Urcuyo L, et al. Comparative effectiveness of second-generation antipsychotics and haloperidol in acute schizophrenia. Br J Psychiatry 2006; 189:433–40
Kane JM, Osuntokun O, Kryzhanovskaya LA, et al. A 28-week, randomized, double-blind study of olanzapine versus aripiprazole in the treatment of schizophrenia. J Clin Psychiatry 2009; 70(4): 572–81
Nasrallah HA. The roles of efficacy, safety, and tolerability in antipsychotic effectiveness: practical implications of the CATIE schizophrenia trial. J Clin Psychiatry 2007; 68 Suppl. 1:5–11
Newcomer JW. Metabolic considerations in the use of antipsychotic medications: a review of recent evidence. J Clin Psychiatry 2007; 68 Suppl. 1: 20–7
Hogan TP, Awad AG, Eastwood R. A self-report scale predictive of drug compliance in schizophrenics: reliability and discriminative validity. Psychol Med 1983; 13(1): 177–83
Naber D, Lambert M. The CATIE and CUtLASS studies in schizophrenia: results and implications for clinicians. CNS Drugs 2009; 23(8): 649–59
Casey DE. Implications of the CATIE trial on treatment: extrapyramidal symptoms. CNS Spectr 2006; 11(7 Suppl. 7): 25–31
Correll CU, Leucht S, Kane JM. Lower risk for tardive dyskinesia associated with second-generation antipsychotics: a systematic review of 1-year studies. Am J Psychiatry 2004; 161(3): 414–25
Oosthuizen PP, Emsley RA, Maritz JS, et al. Incidence of tardive dyskinesia in first-episode psychosis patients treated with low-dose haloperidol. J Clin Psychiatry 2003; 64(9): 1075–80
Kraemer HC, Glick ID, Klein DF. Clinical trials design lessons from the CATIE study. Am J Psychiatry 2009; 166(11): 1222–8
Kane JM. Commentary on the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE). J Clin Psychiatry 2006; 67(5): 831–2
Lieberman JA. Comparative effectiveness of antipsychotic drugs: a commentary on Cost Utility Of The Latest Antipsychotic Drugs In Schizophrenia Study (CUtLASS 1) and Clinical Antipsychotic Trials Of Intervention Effectiveness (CATIE). Arch Gen Psychiatry 2006; 63(10): 1069–72
Sparshatt A, Taylor D, Patel MX, et al. Relationship between daily dose, plasma concentrations, dopamine receptor occupancy, and clinical response to quetiapine: a review. J Clin Psychiatry 2011; 72(8): 1108–23
Sparshatt A, Jones S, Taylor D. Quetiapine: dose-response relationship in schizophrenia. CNS Drugs 2008; 22(1): 49–68
Citrome L, Stauffer VL, Chen L, et al. Olanzapine plasma concentrations after treatment with 10, 20, and 40mg/d in patients with schizophrenia: an analysis of correlations with efficacy, weight gain, and prolactin concentration. J Clin Psychopharmacol 2009; 29(3): 278–83
Soares BG, Fenton M, Chue P. Sulpiride for schizophrenia. Cochrane Database Syst Rev 2000; (2): CD001162
Wahlbeck K, Tuunainen A, Ahokas A, et al. Dropout rates in randomised antipsychotic drug trials. Psychopharma-cology (Berl) 2001; 155(3): 230–3
Carpenter WT, Buchanan RW. Lessons to take home from CATIE. Psychiatr Serv 2008; 59(5): 523–5
Foussias G, Remington G. Antipsychotics and schizophrenia: from efficacy and effectiveness to clinical decision-making. Can J Psychiatry 2010; 55(3): 117–25
Agid O, Arenovich T, Sajeev G, et al. An algorithm-based approach to first-episode schizophrenia: response rates over 3 prospective antipsychotic trials with a retrospective data analysis. J Clin Psychiatry 2011; 72(11): 1439–44
Swartz MS, Stroup TS, McEvoy JP, et al. What CATIE found: results from the schizophrenia trial. Psychiatr Serv 2008; 59(5): 500–6
Tenback DE, van Harten PN, Slooff CJ, et al. Tardive dyskinesia in schizophrenia is associated with prolactin-related sexual disturbances. Neuropsychopharmacology 2006; 31(8): 1832–7
Acknowledgements
Azizah Attard has no conflicts of interest that are directly relevant to the content of this review. David Taylor has received research funding and/or honoraria from Novartis, Eli Lilly, AstraZeneca, Janssen, Sanofi-Aventis and Lundbeck.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Attard, A., Taylor, D.M. Comparative Effectiveness of Atypical Antipsychotics in Schizophrenia. CNS Drugs 26, 491–508 (2012). https://doi.org/10.2165/11632020-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11632020-000000000-00000