Abstract
This paper investigates the contribution of pharmaceutical innovation to recent longevity growth in Germany and France. The effect of the vintage of prescription drugs (and other variables) on the life expectancy and ageadjusted mortality rates of residents of Germany is examined, using longitudinal, annual, state-level data during the period 2001v7. The estimates imply that about one-third of the 1.4–year increase in German life expectancy during the period 2001–7 was due to the replacement of older drugs by newer drugs. The effect of the vintage of chemotherapy treatments on age-adjusted cancer mortality rates of residents of France is also investigated, using longitudinal, annual, cancer-site-level data during the period 2002–6. The estimates imply that chemotherapy innovation accounted for at least one-sixth of the decline in French cancer mortality rates, and may have accounted for as much as half of the decline.










Similar content being viewed by others
Notes
Growth may also be driven by technological change arising from R&D investment by public organizations, e.g. the National Institutes of Health.
I hypothesize that innovations may be embodied in nondurable goods (e.g. drugs) and services as well as in durable equipment.
In 1997, “medical substances and devices firms had by far the highest combined R&D intensity at 11.8 percent, … well above the 4.2-percent average for all 500 top 1997 R&D spenders combined. The information and electronics sector ranked second in intensity at 7.0 percent.”[17] The pattern of 1997 R&D spending per employee is similar to that for R&D intensity, with medical substances and devices again the highest at $US29 095 per employee. Information and electronics is second at $US16 381. Combined, the top 500 R&D firms in 1997 spent $US10 457 per employee.
European Pharmaceutical Market Research Association (EphMRA) drug classification codes are shown in parentheses. The EphMRA classification is a modified version of the Anatomical Therapeutic Chemical (ATC) classification. See http://www.ephmra.org/classification/anatomical-classification.aspx.
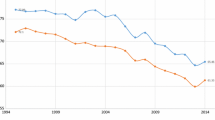
Population-weighted averages of my state-level life expectancy estimates were verified as being very consistent with published estimates for Germany as a whole.
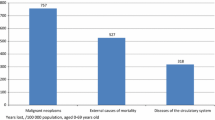
In the Federal Republic of Germany, health authorities must be informed about cases of certain notifiable diseases, which are listed in the Infection Protection Act.[21] Data on the incidence and prevalence of other diseases were not available.
Data on educational attainment by state and year were not available.
Lichtenberg found that diagnostic imaging innovation as well as pharmaceutical innovation increased life expectancy in the US during the period 1991–2004.[26]
The CEA Registry is produced by the Center for the Evaluation of Value and Risk in Health, part of the Institute for Clinical Research and Health Policy Studies at Tufts Medical Center in Boston, MA, USA.
As shown in table II, the average prescription is for a drug launched about 20 years earlier.
The Eurocare 3 and Eurocare 4 databases[32] provide data on survival rates of French cancer patients diagnosed during the following periods: 1983–5, 1986–8, 1989–91, 1992–4 and 1995–9.
Data on non-pharmaceutical innovation are not available for any period. According to the European Cancer Observatory, annual data on cancer incidence, by site, are only available during the period 1983–1997.[34]
If a patient was treated with n chemotherapy agents, that patient would be counted n times.
The age distribution of the French population in 2002 was used to obtain age-adjusted mortality rates.
GERS provides data on the quantity of each drug, by year, but not by cancer site.
The GERS vintage measures are based on the year each drug was first commercialized in France, rather than the world launch year, which is not available for all drugs. For the 11 drugs for which both dates were available, there is generally a close correspondence between the two dates. For 8 of the 11 drugs, the year of commercialization in France was 0–2 years after the world launch year.
References
Nordhaus WD. The health of nations: the contribution of improved health to living standards. In: Murphy KM, Topel RH, editors. Measuring the gains from medical research: an economic approach. Chicago (IL): University of Chicago Press, 2003: 9–40
Murphy KM, Topel RH. The value of health and longevity. J Polit Econ 2006 Aug; 114 (5): 871–904
Wikipedia. Human development index [online]. Available from URL: http://en.wikipedia.org/wiki/Human_Development_Index [Accessed 2011 Oct 29]
Solow RM. A contribution to the theory of economic growth. Quart J Econ 1956; 70 (1): 65–94
Solow RM. Technical change and the aggregate production function. Rev Econ Stat 1957; 39 (3): 312–20
Romer P. Endogenous technological change. J Polit Econ 1990 Oct; 98 (5 Pt 2): S71–102
Jones C. Introduction to economic growth. New York: Norton, 1998
Solow RM. Investment and technological progress. In: Arrow K, Karlin S, Suppes P, editors. Mathematical methods in social sciences 1959. Stanford (CA): Stanford University Press, 1960: 89–104
Grossman GM, Helpman E. Innovation and growth in the global economy. Cambridge (MA): MIT Press, 1993
Bresnahan TF, Gordon RJ. The economics of new goods. Chicago (IL): University of Chicago Press, 1996
Bils M. Measuring the growth from better and better goods [NBER working paper no. 10606]. 2004 Jul [online]. Available from URL: http://www.nber.org/papers/w10606 [Accessed 2011 Oct 29]
Hercowitz Z. The ‘embodiment’ controversy: a review essay. J Monetary Econ 1998; 41: 217–24
Wikipedia. Exogenous growth model [online]. Available from URL: http://en.wikipedia.org/wiki/Exogenous_growth_model [Accessed 2011 Oct 29]
Bahk BH, Gort M. Decomposing learning by doing in new plants. J Polit Econ 1993; 101 (4): 561–83
Hulten CR. Growth accounting when technical change is embodied in capital. Am Econ Rev 1992 Sep; 82 (4): 964–80
Sakellaris P, Wilson DJ. Quantifying embodied technological change. Rev Econ Dyn 2004; 7 (1): 1–26
National Science Foundation. US corporate R&D. Volume 1: top 500 firms in R&D by industry category [online]. Available from URL: http://www.nsf.gov/statistics/nsf00301/expendit.htm [Accessed 2011 Oct 29]
IMS. IMS national prescription analysis database [inGerman; online]. Available from URL: http://www.imshealth.de/sixcms/detail.php/375 [Accessed 2011 Oct 29]
US Food and Drug Administration. Drugs@FDA data files [online]. Available from URL: http://www.fda.gov/Drugs/InformationOnDrugs/ucm079750.htm [Accessed 2011 Oct 29]
Gesundheitsberichterstattung des Bundes. The information system of the Federal Health Monitoring [in German; online]. Available from URL: http://www.gbe-bund.de/ [Accessed 2011 Oct 29]
Robert Koch Institute [in German; online]. Available from URL: http://www.gbe-bund.de/oowa921-install/servlet/oowa/aw92/dboowasys921.xwdevkit/xwd_init?gbe.isgbetol/xs_start_neu/&p_aid=i&p_aid=94387221&nummer=179&p_sprache=E&p_indsp=-&p_aid=81869878 [Accessed 2011 Oct 29]
European Commission. Eurostat: statistics database [online]. Available from URL: http://epp.eurostat.ec.europa.eu/portal/page/portal/statistics/search_database [Accessed 2011 Oct 29]
Uchida E, Araki S, Murata K. Socioeconomic factors affecting the longevity of the Japanese population: a study for 1980 and 1985. J Biosoc Sci 1992; 24: 497–504
Hupfeld S. Non-monotonicity in the longevity-income relationship. J Pop Econ 2011; 24 (1): 191–211
Ruhm CJ. Macroeconomic conditions, health and mortality [NBER working paper no. 11007]. 2004 Dec [online]. Available from URL: http://www.nber.org/papers/w11007 [Accessed 2011 Oct 29]
Lichtenberg FR. The quality of medical care, behavioral risk factors, and longevity growth. Int J Health Care Finance Econ 2011; 11 (1): 1–34
OECD. OECD health data 2011 [online]. Available from URL: http://www.oecd.org/document/30/0,3746,en_2649_37407_12968734_1_1_1_37407,00.html [Accessed 2011 Oct 30]
Tufts Medical Center, Institute for Clinical Research and Health Policy Studies, The Center for Evaluation of Value and Risk in Health. Cost-Effectiveness Analysis Registry [online]. Available from URL: http://research.tuftsnemc.org/cear4/ [Accessed 2011 Oct 29]
Johnson JR, Williams G, Pazdur R. End points and United States Food and Drug Administration approval of oncology drugs. J Clin Oncol 2003; 21 (7): 1404–11
National Institute for Health and Clinical Excellence. The clinical effectiveness and cost-effectiveness of new drug treatments for rheumatoid arthritis: etanercept and infliximab. London: NICE, 2001 [online]. Available from URL: http://www.nice.org.uk/nicemedia/pdf/RAAssessmentReport.pdf [Accessed 2011 Oct 29]
Lichtenberg FR. Have newer cardiovascular drugs reduced hospitalization? Evidence from longitudinal country-level data on 20 OECD countries, 1995-2003. Health Econ 2009; 18 (5): 19–34
Instituto Superiore di Sanita. EUROCARE databases [online]. Available from URL: http://www.eurocare.it/Home/tabid/36/Default.aspx [Accessed 2011 Oct 29]
Welch HG, Schwartz LM, Woloshin S. Are increasing 5-year survival rates evidence of success against cancer? JAMA 2000; 283 (22): 2975–8
European Cancer Observatory. Table of cancer incidence and mortality rates [online]. Available from URL: http://eu-cancer.iarc.fr/16-table.html,en [Accessed 2011 Oct 29]
Lichtenberg FR. Has medical innovation reduced cancer mortality? [NBER working paper no. 15880]. 2010 Apr [online]. Available from URL: http://www.nber.org/papers/w15880 [Accessed 2011 Oct 29]
IMS [online]. Available from URL: http://ims-america.com/portal/site/imshealth/menuitem.a46c6d4df3db4b3d88f611019418c22a/?vgnextoid=1314e69040fb2210VgnVCM100000ed152ca2RCRD&cpsextcurrchannel=1 [Accessed 2011 Oct 30]
CépiDC, INSERM. Examination of data on causes of death from 1979 to 2009 [in French; online]. Available from URL: http://www.cepidc.vesinet.inserm.fr/inserm/html/index2.htm [Accessed 2011 Oct 29]
Eurostat. Causes of death: standardised death rate (per 100,000 inhabitants) (annual data) [online]. Available from URL: http://appsso.eurostat.ec.europa.eu/nui/show.do?dataset=hlth_cd_asdr&lang=en [Accessed 2011 Oct 31]
GERS [in French; online]. Available from URL: http://www.gie-gers.fr/index.php3 [Accessed 2011 Oct 29]
Acknowledgements
This research was supported by Merck Sharpe and Dohme Corporation and by Servier Laboratories. The sponsors placed no restrictions or limitations on the data, methods or conclusions, and had no right of review or control over the outcome of the research. The author has received honoraria and grants from the aforementioned pharmaceutical companies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lichtenberg, F.R. Contribution of Pharmaceutical Innovation to Longevity Growth in Germany and France, 2001–7. PharmacoEconomics 30, 197–211 (2012). https://doi.org/10.2165/11587150-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11587150-000000000-00000