Abstract
Increasingly, computer simulation models are used for economic and policy evaluation in cancer prevention and control. A model’s predictions of key outcomes, such as screening effectiveness, depend on the values of unobservable natural history parameters. Calibration is the process of determining the values of unobservable parameters by constraining model output to replicate observed data. Because there are many approaches for model calibration and little consensus on best practices, we surveyed the literature to catalogue the use and reporting of these methods in cancer simulation models.
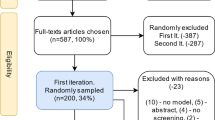
We conducted a MEDLINE search (1980 through 2006) for articles on cancer-screening models and supplemented search results with articles from our personal reference databases. For each article, two authors independently abstracted pre-determined items using a standard form. Data items included cancer site, model type, methods used for determination of unobservable parameter values and description of any calibration protocol. All authors reached consensus on items of disagreement. Reviews and non-cancer models were excluded. Articles describing analytical models, which estimate parameters with statistical approaches (e.g. maximum likelihood) were catalogued separately.Models that included unobservable parameters were analysed and classified by whether calibration methods were reported and if so, the methods used.
The review process yielded 154 articles that met our inclusion criteria and, of these, we concluded that 131 may have used calibration methods to determine model parameters. Although the term ‘calibration’ was not always used, descriptions of calibration or ‘model fitting’ were found in 50% (n = 66) of the articles, with an additional 16% (n = 21) providing a reference to methods. Calibration target data were identified in nearly all of these articles. Other methodological details, such as the goodness-of-fit metric, were discussed in 54% (n = 47 of 87) of the articles reporting calibration methods, while few details were provided on the algorithms used to search the parameter space.
Our review shows that the use of cancer simulation modelling is increasing, although thorough descriptions of calibration procedures are rare in the published literature for these models. Calibration is a key component of model development and is central to the validity and credibility of subsequent analyses and inferences drawn from model predictions. To aid peer-review and facilitate discussion of modelling methods, we propose a standardized Calibration Reporting Checklist for model documentation.




Similar content being viewed by others
References
Ramsey SD, McIntosh M, Etzioni RD, et al. Simulation modeling of outcomes and cost-effectiveness. Hematol Oncol Clin North Am 2000 Aug; 14 (4): 925–38
Knudsen AB, McMahon PM, Gazelle GS. Use of modeling to evaluate the cost-effectiveness of cancer screening programs. J Clin Oncol 2007; 25: 203–8
Feuer EJ, Etzioni RD, Cronin KA, et al. The use of modeling to understand the impact of screening on US mortality: examples from mammography and PSA testing. Stat Methods Med Res 2004 Dec; 13: 421–42
Goldie SJ. Chapter 15: public health policy and cost-effectiveness analysis. J Natl Cancer Inst Monogr 2003; 31: 102–10
Goldie SJ, Goldhaber-Fiebert JD, Garnett G. Chapter 18: public health policy for cervical cancer prevention. The role of decision science, economic evaluation, and mathematical modeling. Vaccine 2006 Aug 31; 24 (S3): 155–63
Weinstein MC. Recent developments in decision-analytic modelling for economic evaluation. Pharmacoeconomics 2006; 24 (11): 1043–53
Goldie SJ, Grima D, Kohli M, et al. A comprehensive natural history model of HPV infection and cervical cancer to estimate the clinical impact of a prophylactic HPV-16/18 vaccine. Int J Cancer 2003; 106: 896–904
Yeh JM, Kuntz KM, Ezzati M, et al. Development of an empirically calibrated model of gastric cancer in two high-risk countries. Cancer Epidemiol Biomarkers Prev 2008 May 1; 17 (5): 1179–87
Mandelblatt JS, Lawrence WF, Womack SM, et al. Benefits and costs of using HPV testing to screen for cervical cancer. JAMA 2002 May 8; 287 (18): 2372–81
Urban N, Drescher C, Etzioni R, et al. Use of a stochastic simulation model to identify an efficient protocol for ovarian cancer screening. Control Clin Trials 1997 Jun; 18 (3): 251–70
Myers ER, McCrory DC, Nanda K, et al. Mathematical model for the natural history of human papillomavirus infection and cervical carcinogenesis. Am J Epidemiol 2000 June 15, 2000; 151 (12): 1158–71
Law AM, Kelton WD. Simulation modeling and analysis. 3rd ed. Boston (MA): McGraw-Hill, 2000
Clarke LD, Plevritis SK, Boer R, et al. A comparative review of CISNET breast models used to analyze U.S. breast cancer incidence and mortality trends. J Natl Cancer Inst Monogr 2006; 36: 96–105
Banks J, editor. Handbook of simulation: principles, methodology, advances, applications, and practice. New York: John Wiley & Sons. Inc., 1998
Bruning JL, Kintz BL. Computational handbook of statistics. 4th ed. Boston (MA): Allyn & Bacon, 1997
Nelder JA, Mead R. A simplex method for function minimization. Comput J 1965; 7 (4): 308–13
Kirkpatrick S, Gelatt CD, Vecchi MP. Optimization by simulated annealing. Science 1983; 220 (4598): 671–80
Press WH, Teukolsky SA, Vetterling WT, et al. Numerical recipes in C++. 2nd ed. New York (NY): Cambridge University Press, 2002
Wong DF, Leong HW, Liu CL. Simulated annealing for VLSI design. Boston (MA): Kluwer Academic Publishers, 1988
Holland JH. Adaptation in natural and artificial systems. Ann Arbor (MI): University of Michigan Press, 1975
Goldberg DE. Genetic algorithms in search, optimization and machine learning. Boston (MA): Kluwer Academic Publishers, 1989
Manikas TW, Cain JT. Genetic algorithms vs. simulated annealing: a comparison of approaches for solving the circuit partitioning problem. Pittsburgh (PA): University of Pittsburgh, 1996
Ingber L, Rosen B. Genetic algorithms and very fast simulated annealing: a comparison. Math Comput Model 1992; 16 (11): 87–100
Glover F. Tabu search: part I. ORSA J Computing 1989; 1 (3): 190–206
Glover F. Tabu search: part II. ORSA J Computing 1990; 2 (1): 4–32
Kong CY, McMahon PM, Gazelle GS. Calibration of disease simulation models using an engineering approach. Value Health 2009; 12 (4): 521–9
Baker RD. Use of a mathematical model to evaluate breast cancer screening policy. Health Care Manage Sci 1998 Oct; 1 (2): 103–13
Beckett JR, Kotre CJ, Michaelson JS. Analysis of benefit: risk ratio and mortality reduction for the UK Breast Screening Programme. Br J Radiol 2003 May; 76 (905): 309–20
Beemsterboer PM, Warmerdam PG, Boer R, et al. Radiation risk of mammography related to benefit in screening programmes: a favourable balance? J Med Screen 1998; 5 (2): 81–7
Berry DA, Cronin KA, Plevritis SK, et al. Effect of screening and adjuvant treatment on mortality from breast cancer. N Engl J Med 2005 Oct 27; 353 (17): 1784–92
Berry DA, Inoue L, Shen Y, et al. Modeling the impact of treatment and screening on U.S. breast cancer mortality: a Bayesian approach. J Natl Cancer Inst Monogr 2006; 36: 30–6
Blanchard K, Colbert JA, Puri D, et al. Mammographic screening: patterns of use and estimated impact on breast carcinoma survival. Cancer 2004 Aug 1; 101 (3): 495–507
Boer R, de Koning H, Threlfall A, et al. Cost effectiveness of shortening screening interval or extending age range of NHS breast screening programme: computer simulation study. BMJ 1998 Aug 8; 317 (7155): 376–9
Boer R, de Koning H, van Oortmarssen G, et al. Stage distribution at first and repeat examinations in breast cancer screening. J Med Screen 1999; 6 (3): 132–8
Carter R, Glasziou P, van Oortmarssen G, et al. Cost-effectiveness of mammographic screening in Australia. Aust J Public Health 1993 Mar; 17 (1): 42–50
Carter KJ, Castro F, Kessler E, et al. A computer model for the study of breast cancer. Comput Biol Med 2003 Jul; 33: 345–60
Carter KJ, Castro F, Kessler E, et al. Simulation of breast cancer screening: quality assessment of two protocols. J Healthc Qual 2004 Nov-Dec; 26 (6): 31–8
Castro F, Carter KJ, Kessler E, et al. The relation of breast cancer staging to screening protocol compliance: a computer simulation study. Comput Biol Med 2005 Feb; 35 (2): 91–101
Chia YL, Salzman P, Plevritis SK, et al. Simulation-based parameter estimation for complex models: a breast cancer natural history modelling illustration. Stat Methods Med Res 2004 Dec; 13 (6): 507–24
Cong XJ, Shen Y, Miller AB. Estimation of age-specific sensitivity and sojourn time in breast cancer screening studies. Stat Med 2005 Oct 30; 24 (20): 3123–38
Connor RJ, Boer R, Prorok PC, et al. Investigation of design and bias issues in case-control studies of cancer screening using microsimulation. Am J Epidemiol 2000 May 15; 151 (10): 991–8
de Koning HJ, van Ineveld BM, van Oortmarssen GJ, et al. Breast cancer screening and cost-effectiveness: policy alternatives, quality of life considerations and the possible impact of uncertain factors. Int J Cancer 1991 Oct 21; 49: 531–7
de Koning HJ, Boer R, Warmerdam PG, et al. Quantitative interpretation of age-specific mortality reductions from the Swedish breast cancer-screening trials. J Natl Cancer Inst 1995 Aug 16; 87 (16): 1217–23
Eddy DM. Screening for breast cancer. Ann Intern Med 1989; 111 (5): 389–99
Feldstein M, Zelen M. Inferring the natural time history of breast cancer: implications for tumor growth rate and early detection. Breast Cancer Res Treat 1984; 4: 3–10
Fett MJ. Computer modelling of the Swedish two county trial of mammographic screening and trade offs between participation and screening interval. J Med Screen 2001; 8 (1): 39–45
Fracheboud J, Groenewoud JH, Boer R, et al. Seventy-five years is an appropriate upper age limit for population-based mammography screening. Int J Cancer 2006 Apr 15; 118 (8): 2020–5
Fryback DG, Stout NK, Rosenberg MA, et al. The Wisconsin breast cancer epidemiology simulation model. J Natl Cancer Inst Monogr 2006; 36: 37–47
Hanin LG, Miller A, Zorin AV, et al. The University of Rochester model of breast cancer detection and survival. J Natl Cancer Inst Monogr 2006; 36: 66–78
Hsieh HJ, Chen TH, Chang SH. Assessing chronic disease progression using non-homogeneous exponential regression Markov models: an illustration using a selective breast cancer screening in Taiwan. Stat Med 2002 Nov 30; 21 (22): 3369–82
Hunter DJ, Drake SM, Shortt SE, et al. Simulation modeling of change to breast cancer detection age eligibility recommendations in Ontario, 2002–2021. Cancer Detect Prev 2004; 28 (6): 453–60
Jacobi CE, Jonker MA, Nagelkerke NJ, et al. Prevalence of family histories of breast cancer in the general population and the incidence of related seeking of health care [letter]. J Med Genet 2003 Jul; 40 (7): e83
Jacobi CE, Nagelkerke NJ, van Houwelingen JH, et al. Breast cancer screening, outside the population-screening program, of women from breast cancer families without proven BRCA1/BRCA2 mutations: a simulation study. Cancer Epidemiol Biomarkers Prev 2006 Mar; 15 (3): 429–36
Jansen JT, Zoetelief J. MBS: a model for risk benefit analysis of breast cancer screening. Br J Radiol 1995 Feb; 68 (806): 141–9
Jansen JT, Zoetelief J. Assessment of lifetime gained as a result of mammographic breast cancer screening using a computer model. Br J Radiol 1997 Jun; 70 (834): 619–28
Jansen JT, Zoetelief J. Optimisation of mammographic breast cancer screening using a computer simulation model. Eur J Radiol 1997 Feb; 24 (2): 137–44
Knox EG. Evaluation of a proposed breast cancer screening regimen. BMJ 1988 Sep 10; 297 (6649): 650–4
Koscielny S, Tubiana M, Valleron AJ. A simulation model of the natural history of human breast cancer. Br J Cancer 1985 Oct; 52 (4): 515–24
Lee S, Zelen M. A stochastic model for predicting the mortality of breast cancer. J Natl Cancer Inst Monogr 2006; 36: 79–86
Mandelblatt JS, Schechter CB, Yabroff KR, et al. Benefits and costs of interventions to improve breast cancer outcomes in African American women. J Clin Oncol 2004 Jul 1; 22 (13): 2554–66
Mandelblatt J, Schechter CB, Lawrence W, et al. The SPECTRUM population model of the impact of screening and treatment on U.S. breast cancer trends from 1975 to 2000: principles and practice of the model methods. J Natl Cancer Inst Monogr 2006; 36: 47–55
Manton KG, Stallard E. Demographics (1950–1987) of breast cancer in birth cohorts of older women. J Gerontol 1992 Nov; 47 Spec No: 32–42
Michaelson JS, Halpern E, Kopans DB. Breast cancer: computer simulation method for estimating optimal intervals for screening. Radiology 1999 Aug; 212 (2): 551–60
Michaelson JS, Satija S, Moore R, et al. Estimates of breast cancer growth rate and sojourn time from screening database information. J Wom Imag 2003 Feb; 5 (1): 11–9
Michaelson JS, Satija S, Moore R, et al. Estimates of the sizes at which breast cancers become detectable on mammographic and clinical grounds. J Wom Imag 2003 Feb; 5 (1): 3–10
Myles JP, Nixon RM, Duffy SW, et al. Bayesian evaluation of breast cancer screening using data from two studies. Stat Med 2003 May 30; 22 (10): 1661–74
Okubo I, Glick H, Frumkin H, et al. Cost-effectiveness analysis of mass screening for breast cancer in Japan. Cancer 1991 Apr 15; 67 (8): 2021–9
Paci E, Boer R, Zappa M, et al. A model-based prediction of the impact on reduction in mortality by a breast cancer screening programme in the city of Florence, Italy. Eur J Cancer 1995; 31A (3): 348–53
Plevritis SK. A mathematical algorithm that computes breast cancer sizes and doubling times detected by screening. Math Biosci 2001 Jun; 171 (2): 155–78
Plevritis SK, Kurian AW, Sigal BM, et al. Cost-effectiveness of screening BRCA1/2 mutation carriers with breast magnetic resonance imaging. JAMA 2006 May 24; 295 (20): 2374–84
Plevritis SK, Sigal BM, Salzman P, et al. A stochastic simulation model of U.S. breast cancer mortality trends from 1975 to 2000. J Natl Cancer Inst Monogr 2006; 36: 86–95
Plevritis SK, Salzman P, Sigal BM, et al. A natural history model of stage progression applied to breast cancer. Stat Med 2006; 26 (3): 581–95
Shen Y, Huang X. Nonparametric estimation of asymptomatic duration from a randomized prospective cancer screening trial. Biometrics 2005 Dec; 61 (4): 992–9
Shen Y, Zelen M. Robust modeling in screening studies: estimation of sensitivity and preclinical sojourn time distribution. Biostatistics 2005 Oct; 6 (4): 604–14
Stout NK, Rosenberg MA, Trentham-Dietz A, et al. Retrospective cost-effectiveness analysis of screening mammography. J Natl Cancer Inst 2006 Jun 7; 98 (11): 774–82
Szeto KL, Devlin NJ. The cost-effectiveness of mammography screening: evidence from a microsimulation model for New Zealand. Health Policy 1996 Nov; 38 (2): 101–15
Tan SYGL, van Oortmarssen GJ, de Koning HJ, et al. The MISCAN-Fadia continuous tumor growth model for breast cancer. J Natl Cancer Inst Monogr 2006; 36: 56–65
Tubiana M, Koscielny S. The natural history of breast cancer: implications for a screening strategy. Int J Radiat Oncol Biol Phys 1990 Nov; 19 (5): 1117–20
van der Maas PJ, de Koning HJ, van Ineveld BM, et al. The cost-effectiveness of breast cancer screening. Int J Cancer 1989 Jun 15; 43 (6): 1055–60
van Oortmarssen GJ, Habbema JD, Lubbe JT, et al. A model-based analysis of the HIP project for breast cancer screening. Int J Cancer 1990 Aug 15; 46 (2): 207–13
van Oortmarssen GJ, Habbema JD, van der Maas PJ, et al. A model for breast cancer screening. Cancer 1990 Oct 1; 66 (7): 1601–12
Vervoort MM, Draisma G, Fracheboud J, et al. Trends in the usage of adjuvant systemic therapy for breast cancer in the Netherlands and its effect on mortality. Br J Cancer 2004 Jul 19; 91 (2): 242–7
Berkhof J, de Bruijne MC, Zielinski GD, et al. Natural history and screening model for high-risk human papillomavirus infection, neoplasia and cervical cancer in the Netherlands. Int J Cancer 2005 Jun 10; 115 (2): 268–75
Berkhof J, de Bruijne MC, Zielinski GD, et al. Evaluation of cervical screening strategies with adjunct high-risk human papillomavirus testing for women with borderline or mild dyskaryosis. Int J Cancer 2006 Apr 1; 118 (7): 1759–68
Eddy DM. The frequency of cervical cancer screening: comparison of a mathematical model with empirical data. Cancer 1987 Sep 1; 60 (5): 1117–22
Eddy DM. Screening for cervical cancer. Ann Intern Med 1990 Aug 1; 113 (3): 214–26
Goldie SJ, Weinstein MC, Kuntz KM, et al. The costs, clinical benefits, and cost-effectiveness of screening for cervical cancer in HIV-infected women. Ann Intern Med 1999 Jan 19; 130 (2): 97–107
Goldie SJ, Kuhn L, Denny L, et al. Policy analysis of cervical cancer screening strategies in low-resource settings: clinical benefits and cost-effectiveness. JAMA 2001 Jun 27; 285 (24): 3107–15
Goldie SJ, Kim JJ, Wright TCJ. Cost-effectiveness of human papillomavirus DNA testing for cervical cancer screening in women aged 30 years or more. Obstet Gynecol 2004 Apr; 103 (4): 619–31
Goldie SJ, Kohli M, Grima D, et al. Projected clinical benefits and cost-effectiveness of a human papillomavirus 16/18 vaccine. J Natl Cancer Inst 2004 Apr 21; 96 (8): 604–15
Goldie SJ, Gaffikin L, Goldhaber-Fiebert JD, et al. Cost-effectiveness of cervical-cancer screening in five developing countries. N Engl J Med 2005 Nov 17; 353 (20): 2158–68
Gustafsson L, Adami HO. Natural history of cervical neoplasia: consistent results obtained by an identification technique. Br J Cancer 1989 Jul; 60 (1): 132–41
Gustafsson L, Adami HO. Cytologic screening for cancer of the uterine cervix in Sweden evaluated by identification and simulation. Br J Cancer 1990 Jun; 61 (6): 903–8
Gustafsson L, Adami HO. Optimization of cervical cancer screening. Cancer Causes Control 1992 Mar; 3 (2): 125–36
Gyrd-Hansen D, Holund B, Andersen P. A cost-effectiveness analysis of cervical cancer screening: health policy implications. Health Policy 1995 Oct; 34 (1): 35–51
Habbema JD, van Oortmarssen GJ, Lubbe JT, et al. Model building on the basis of Dutch cervical cancer screening data. Maturitas 1985 May; 7 (1): 11–20
Habbema JDF, Lubbe JTN, van Oortmarssen GJ, et al. A simulation approach to cost-effectiveness and cost-benefit calculations of screening for the early detection of disease. Eur J Oper Res 1987 May; 29 (2): 159–66
Helfand M, O’Connor GT, Zimmer-Gembeck M, et al. Effect of the Clinical Laboratory Improvement Amendments of 1988 (CLIA ‘88) on the incidence of invasive cervical cancer. Med Care 1992 Dec; 30 (12): 1067–82
Kim JJ, Wright TCJ, Goldie SJ. Cost-effectiveness of alternative triage strategies for atypical squamous cells of undetermined significance. JAMA 2002 May 8; 287 (18): 2382–90
Kim JJ, Leung GM, Woo PP, et al. Cost-effectiveness of organized versus opportunistic cervical cytology screening in Hong Kong. J Public Health (Oxf) 2004 Jun; 26 (2): 130–7
Knox EG. A simulation system for screening procedures. In: McLachlan G, editor. Future and present indicatives, problems and progress in medical care. 9th rev. Oxford: Nuffield Provincial Hospitals Trust, Oxford University Press, 1973: 17–55
Koong SL, Yen AM, Chen TH. Efficacy and cost-effectiveness of nationwide cervical cancer screening in Taiwan. J Med Screen 2006; 13 Suppl. 1: S44–7
Koopmanschap MA, Lubbe KT, van Oortmarssen GJ, et al. Economic aspects of cervical cancer screening. Soc Sci Med 1990; 30 (10): 1081–7
Kulasingam SL, Myers ER, Lawson HW, et al. Cost-effectiveness of extending cervical cancer screening intervals among women with prior normal pap tests. Obstet Gynecol 2006 Feb; 107 (2 Pt 1): 321–8
Mandelblatt JS, Lawrence WF, Gaffikin L, et al. Costs and benefits of different strategies to screen for cervical cancer in less-developed countries. J Natl Cancer Inst 2002 Oct 2; 94 (19): 1469–83
Mandelblatt J, Lawrence W, Yi B, et al. The balance of harms, benefits, and costs of screening for cervical cancer in older women. Arch Intern Med 2004 Feb 9; 164: 245–7
Matsunaga G, Tsuji I, Sato S, et al. Cost-effective analysis of mass screening for cervical cancer in Japan. J Epidemiol 1997 Sep; 7 (3): 135–41
Office of Technology Assessment. The costs and effectiveness of screening for cervical cancer in elderly women: background paper, OTA-BP-H-65. Washington, DC: U.S. Congress, 1990
Parkin DM. A computer simulation model for the practical planning of cervical cancer screening programmes. Br J Cancer 1985 Apr; 51 (4): 551–68
Parkin DM, Moss SM. An evaluation of screening policies for cervical cancer in England and Wales using a computer simulation model. J Epidemiol Community Health 1986 Jun; 40 (2): 143–53
Radensky PW, Mango LJ. Interactive neural-network-assisted screening: an economic assessment. Acta Cytol 1998 Jan-Feb; 42 (1): 246–52
Sato S, Matunaga G, Tsuji I, et al. Determining the cost-effectiveness of mass screening for cervical cancer using common analytic models. Acta Cytol 1999 Nov-Dec; 43 (6): 1006–14
Sawaya GF, McConnell KJ, Kulasingam SL, et al. Risk of cervical cancer associated with extending the interval between cervical-cancer screenings. N Engl J Med 2003 Oct 16; 349 (16): 1501–9
Schechter CB. Cost-effectiveness of rescreening conventionally prepared cervical smears by PAPNET testing. Acta Cytol 1996 Nov-Dec; 40 (6): 1272–82
Sherlaw-Johnson C, Gallivan S, Jenkins D. Withdrawing low risk women from cervical screening programmes: mathematical modelling study. BMJ 1999 Feb 6; 318 (7180): 356–60
Sherlaw-Johnson C, Philips Z. An evaluation of liquid-based cytology and human papillomavirus testing within the UK Cervical Cancer Screening Programme. Br J Cancer 2004 Jul 5; 91 (1): 84–91
Shun-Zhang Y, Miller AB, Sherman GJ. Optimising the age, number of tests, and test interval for cervical screening in Canada. J Epidemiol Community Health 1982 Mar; 36 (1): 1–10
Sreenivas V, Prabhakar AK, Ravi R, et al. A simulation approach for estimating the loss of woman years due to cervical cancer and probability of developing cervical cancer. Neoplasma 1989; 36 (5): 623–7
van Ballegooijen M, van den Akker-van Marle E, Patnick J, et al. Overview of important cervical cancer screening process values in European Union (EU) countries, and tentative predictions of the corresponding effectiveness and cost-effectiveness. Eur J Cancer 2000 Nov; 36 (17): 2177–88
van den Akker-van Marle ME, van Ballegooijen M, van Oortmarssen GJ, et al. Cost-effectiveness of cervical cancer screening: comparison of screening policies. J Natl Cancer Inst 2002 Feb 6; 94 (3): 193–204
Clemen RT, Lacke CJ. Analysis of colorectal cancer screening regimens. Health Care Manage Sci 2001 Dec; 4 (4): 257–67
Frazier AL, Colditz GA, Fuchs CS, et al. Cost-effectiveness of screening for colorectal cancer in the general population. JAMA 2000 Oct 18; 284 (15): 1954–61
Gyrd-Hansen D, Søgaard J, Kronborg O. Analysis of screening data: colorectal cancer. Int J Epidemiol 1997 Dec; 26 (6): 1172–81
Gyrd-Hansen D, Søgaard J, Kronborg O. Colorectal cancer screening: efficiency and effectiveness. Health Econ 1998 Feb; 7 (1): 9–20
Haug U, Brenner H. A simulation model for colorectal cancer screening: potential of stool tests with various performance characteristics compared with screening colonoscopy. Cancer Epidemiol Biomarkers Prev 2005 Feb; 14 (2): 422–8
Khandker RK, Dulski JD, Kilpatrick JB, et al. A decision model and cost-effectiveness analysis of colorectal cancer screening and surveillance guidelines for average-risk adults. Int J Technol Assess Health Care 2000 Summer; 16 (3): 799–810
Ladabaum U, Chopra CL, Huang G, et al. Aspirin as an adjunct to screening for prevention of sporadic colorectal cancer: a cost-effectiveness analysis. Ann Intern Med 2001 Nov 6; 135 (9): 769–81
Ladabaum U, Scheiman JM, Fendrick AM. Potential effect of cyclooxygenase-2-specific inhibitors on the prevention of colorectal cancer: a cost-effectiveness analysis. Am J Med 2003 May; 114 (7): 546–54
Lejeune C, Arveux P, Dancourt V, et al. A simulation model for evaluating the medical and economic outcomes of screening strategies for colorectal cancer. Eur J Cancer Prev 2003 Feb; 12 (1): 77–84
Lejeune C, Arveux P, Dancourt V, et al. Cost-effectiveness analysis of fecal occult blood screening for colorectal cancer. Int J Technol Assess Health Care 2004 Fall; 20 (4): 434–9
Loeve F, Boer R, van Oortmarssen GJ, et al. The MISCAN-COLON simulation model for the evaluation of colorectal cancer screening. Comput Biomed Res 1999 Feb; 32 (1): 13–33
Loeve F, Brown ML, Boer R, et al. Endoscopic colorectal cancer screening: a cost-saving analysis. J Natl Cancer Inst 2000 Apr 5; 92 (7): 557–63
Loeve F, Boer R, van Oortmarssen GJ, et al. Impact of systematic false-negative test results on the performance of faecal occult blood screening. Eur J Cancer 2001 May; 37 (7): 912–7
Neilson AR, Whynes DK. Cost-effectiveness of screening for colorectal cancer: a simulation model. IMA J Math Appl Med Biol 1995 Sep-Dec; 12 (3–4): 355–67
Ness RM, Holmes AM, Klein R, et al. Cost-utility of one-time colonoscopic screening for colorectal cancer at various ages. Am J Gastroenterol 2000 Jul; 95 (7): 1800–11
Song K, Fendrick AM, Ladabaum U. Fecal DNA testing compared with conventional colorectal cancer screening methods: a decision analysis. Gastroenterology 2004 May; 126 (5): 1270–9
Vijan S, Hwang EW, Hofer TP, et al. Which colon cancer screening test? A comparison of costs, effectiveness, and compliance. Am J Med 2001 Dec 1; 111 (8): 593–601
Wagner JL, Herdman RC, Wadhwa S. Cost effectiveness of colorectal cancer screening in the elderly. Ann Intern Med 1991 Nov 15; 115 (10): 807–17
Wagner JL, Tunis S, Brown M, et al. Cost-effectiveness of colorectal cancer screening in average-risk adults. In: Young G, Rosen P, Levin B, editors. Prevention and early detection of colorectal cancer. Philadelphia (PA): Saunders, 1996: 321–56
Whynes DK, Neilson AR, Walker AR, et al. Faecal occult blood screening for colorectal cancer: is it cost-effective? Health Econ 1998 Feb; 7 (1): 21–9
Wong JM, Yen MF, Lai MS, et al. Progression rates of colorectal cancer by Dukes’ stage in a high-risk group: analysis of selective colorectal cancer screening. Cancer J 2004 May-Jun; 10 (3): 160–9
Yang KC, Liao CS, Chiu YH, et al. Colorectal cancer screening with faecal occult blood test within a multiple disease screening programme: an experience from Keelung, Taiwan. J Med Screen 2006; 13 Suppl. 1: S8–13
Garside R, Pitt M, Somerville M, et al. Surveillance of Barrett’s oesophagus: exploring the uncertainty through systematic review, expert workshop and economic modelling. Health Technol Assess 2006 Mar; 10 (8): 1–142
Davies R, Crabbe D, Roderick P, et al. A simulation to evaluate screening for Helicobacter pylori infection in the prevention of peptic ulcers and gastric cancers. Health Care Manage Sci 2002 Nov; 5 (4): 249–58
Fendrick AM, Chernew ME, Hirth RA, et al. Clinical and economic effects of population-based Helicobacter pylori screening to prevent gastric cancer. Arch Intern Med 1999 Jan 25; 159 (2): 142–8
Roderick P, Davies R, Raftery J, et al. The cost-effectiveness of screening for Helicobacter pylori to reduce mortality and morbidity from gastric cancer and peptic ulcer disease: a discrete-event simulation model. Health Technol Assess 2003; 7 (6): 1–86
Roderick P, Davies R, Raftery J, et al. Cost-effectiveness of population screening for Helicobacter pylori in preventing gastric cancer and peptic ulcer disease, using simulation. J Med Screen 2003; 10 (3): 148–56
Das P, Ng AK, Earle CC, et al. Computed tomography screening for lung cancer in Hodgkin’s lymphoma survivors: decision analysis and cost-effectiveness analysis. Ann Oncol 2006 May; 17 (5): 785–93
Flehinger BJ, Kimmel M. The natural history of lung cancer in a periodically screened population. Biometrics 1987 Mar; 43 (1): 127–44
Flehinger BJ, Kimmel M, Melamed MR. Natural history of adenocarcinoma-large cell carcinoma of the lung: conclusions from screening programs in New York and Baltimore. J Natl Cancer Inst 1988 May 4; 80 (5): 337–44
Flehinger BJ, Kimmel M, Polyak T, et al. Screening for lung cancer: the Mayo Lung Project revisited. Cancer 1993 Sep 1; 72 (5): 1573–80
Gorlova OY, Kimmel M, Henschke C. Modeling of long-term screening for lung carcinoma. Cancer 2001 Sep 15; 92 (6): 1531–40
Mahadevia PJ, Fleisher LA, Frick KD, et al. Lung cancer screening with helical computed tomography in older adult smokers: a decision and cost-effectiveness analysis. JAMA 2003 Jan 15; 289 (3): 313–22
Marshall D, Simpson KN, Earle CC, et al. Potential cost-effectiveness of one-time screening for lung cancer (LC) in a high risk cohort. Lung Cancer 2001 Jun; 32 (3): 227–36
Yamaguchi N, Tamura Y, Sobue T, et al. Evaluation of cancer prevention strategies by computerized simulation model: an approach to lung cancer. Cancer Causes Control 1991 May; 2 (3): 147–55
Yamaguchi N, Mizuno S, Akiba S, et al. A 50-year projection of lung cancer deaths among Japanese males and potential impact evaluation of anti-smoking measures and screening using a computerized simulation model. Jpn J Cancer Res 1992 Mar; 83 (3): 251–7
Yamaguchi N, Tamura Y, Sobue T, et al. Evaluation of cancer prevention strategies by computerized simulation model: methodological issues. Environ Health Perspect 1994 Nov; 102 Suppl. 8: 67–71
Girgis A, Clarke P, Burton RC, et al. Screening for melanoma by primary health care physicians: a cost-effectiveness analysis. J Med Screen 1996; 3 (1): 47–53
Downer MC, Jullien JA, Speight PM. An interim determination of health gain from oral cancer and precancer screening: 2. Developing a model of population screening. Community Dent Health 1997 Dec; 14 (4): 227–32
Myers ER, Havrilesky LJ, Kulasingam SL, et al. Genomic tests for ovarian cancer detection and management. Evid Rep Technol Assess (Full Rep) 2006 Oct; (145): 1–100
Skates SJ, Singer DE. Quantifying the potential benefit of CA 125 screening for ovarian cancer. J Clin Epidemiol 1991; 44 (4–5): 365–80
Skates SJ, Pauler DK, Jacobs IJ. Screening based on the risk of cancer calculation from Bayesian hierarchical change point and mixture models of longitudinal markers. J Am Stat Assoc 2001; 96 (454): 429–39
Cowen ME, Chartrand M, Weitzel WF. A Markov model of the natural history of prostate cancer. J Clin Epidemiol 1994 Jan; 47 (1): 3–21
Draisma G, Boer R, Otto SJ, et al. Lead times and overdetection due to prostate-specific antigen screening: estimates from the European Randomized Study of Screening for Prostate Cancer. J Natl Cancer Inst 2003 Jun 18; 95 (12): 868–78
Draisma G, De Koning HJ. MISCAN: estimating lead-time and over-detection by simulation. BJU Int 2003 Dec; 92 Suppl. 2: 106–11
Draisma G, Postma R, Schroder FH, et al. Gleason score, age and screening: modeling dedifferentiation in prostate cancer. Int J Cancer 2006 Nov 15; 119 (10): 2366–71
Etzioni R, Cha R, Cowen ME. Serial prostate specific antigen screening for prostate cancer: a computer model evaluates competing strategies. J Urol 1999 Sep; 162 (3 Pt 1): 741–8
Etzioni R, Legler JM, Feuer EJ, et al. Cancer surveillance series: interpreting trends in prostate cancer. Part III: quantifying the link between population prostate-specific antigen testing and recent declines in prostate cancer mortality. J Natl Cancer Inst 1999 Jun 16; 91 (12): 1033–9
Etzioni R, Penson DF, Legler JM, et al. Overdiagnosis due to prostate-specific antigen screening: lessons from U.S. prostate cancer incidence trends. J Natl Cancer Inst 2002 Jul 3; 94 (13): 981–90
Krahn MD, Mahoney JE, Eckman MH, et al. Screening for prostate cancer: a decision analytic view. JAMA 1994 Sep 14; 272 (10): 773–80
Parker C, Muston D, Melia J, et al. A model of the natural history of screen-detected prostate cancer, and the effect of radical treatment on overall survival. Br J Cancer 2006 May 22; 94 (10): 1361–8
Ross KS, Carter HB, Pearson JD, et al. Comparative efficiency of prostate-specific antigen screening strategies for prostate cancer detection. JAMA 2000 Sep 20; 284 (11): 1399–405
Ross KS, Guess HA, Carter HB. Estimation of treatment benefits when PSA screening for prostate cancer is discontinued at different ages. Urology 2005 Nov; 66 (5): 1038–42
Tsodikov A, Szabo A, Wegelin J. A population model of prostate cancer incidence. Stat Med 2006 Aug 30; 25 (16): 2846–66
Kimmel M, Flehinger BJ. Nonparametric estimation of the size-metastasis relationship in solid cancers. Biometrics 1991 Sep; 47 (3): 987–1004
Wang PE, Wang TT, Chiu YH, et al. Evolution of multiple disease screening in Keelung: a model for community involvement in health interventions? J Med Screen 2006; 13 Suppl. 1: S54–8
Garrison LP. The ISPOR good practice modeling principles: a sensible approach. Be transparent, be reasonable. Value Health 2003 Jan-Feb; 6 (1): 6–8
Kim JJ, Kuntz KM, Stout NK, et al. Multiparameter calibration of a natural history model of cervical cancer. Am J Epidemiol 2007 July 15, 2007; 166 (2): 137–50
Goldhaber-Fiebert JD, Stout NK, Ortendahl J, et al. Modeling human papillomavirus and cervical cancer in the United States for analyses of screening and vaccination. Popul Health Metr 2007; 5 (1): 11 [online]. Available from URL: http://www.pophealthmetrics.com/content/5/1/11 [Accessed 2009 Jul 1]
Weinstein MC, O’Brien B, Hornberger J, et al. Principles of good practice for decision analytic modeling in health-care evaluation. Report of the ISPOR task force on good research practices: modeling studies. Value Health 2003 Jan-Feb; 6 (1): 9–17
Acknowledgements
The authors gratefully acknowledge the support of Drs Eric Feuer and Karen Kuntz and members of the NCI Cancer Intervention and Surveillance Modeling Network. This work was supported in part by grants from the National Cancer Institute: F32 CA125984 (Natasha K. Stout), R25 CA92203 (Amy B. Knudsen), K99 126147 (Pamela M. McMahon, Chung Yin Kong) and R01 97337 (G. Scott Gazelle, Pamela M. McMahon, Chung Yin Kong). The funding agreements ensured the authors’ independence in designing the study, collecting, analysing and interpreting the data and writing and publishing the report. An earlier version of this work was presented at the 2007 Society for Medical Decision Making Annual Meeting.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Stout, N.K., Knudsen, A.B., Kong, C.Y. et al. Calibration Methods Used in Cancer Simulation Models and Suggested Reporting Guidelines. Pharmacoeconomics 27, 533–545 (2009). https://doi.org/10.2165/11314830-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11314830-000000000-00000