Abstract
Background: Treatment guidelines recommend combination therapy to control blood pressure (BP) in the majority of hypertensive patients. This longterm open-label study assessed a treatment algorithm based on olmesartan medoxomil (hereafter olmesartan), amlodipine and hydrochlorothiazide (HCTZ).
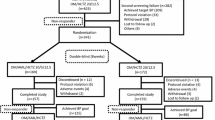
Methods: Patients with moderate-to-severe hypertension who were inadequately controlled with amlodipine 5mg/day monotherapy and who subsequently completed 16 weeks of double-blind combination treatment with olmesartan and amlodipine entered a 28-week open-label phase in which all patients initially received olmesartan/amlodipine 40/5 mg/day. After 4, 10 and 19 weeks, patients with inadequately controlled hypertension (seated trough diastolic [DBP] and systolic [SBP] BP ≥90 mmHg and ≥140 mmHg, respectively) had their doses increased in a step-wise manner to: (i) olmesartan/amlodipine 40/10 mg; (ii) olmesartan/amlodipine/HCTZ 40/10/12.5 mg; and (iii) olmesartan/amlodipine/HCTZ 40/10/25 mg.
Results: In total, 692 patients entered the open-label phase (691 on olmesartan/amlodipine 40/5 mg). The majority of patients remained on olmesartan/amlodipine 40/5 mg without dose elevation, and, of these, 74.3% achieved goal BP at study completion or early termination. Additional patients achieved goal BP with each successive uptitration of therapy: in patients who finished the study on olmesartan/amlodipine 40/10 mg and olmesartan/amlodipine/HCTZ 40/10/12.5 mg, the respective proportions who reached goal BP were 59.0% and 47.1%. Overall, 66.9% of patients achieved the European guideline recommended goal BP of SBP <140 mmHg and DBP <90 mmHg for patients without diabetes mellitus, and SBP <130 mmHg and DBP <80 mmHg for patients with diabetes. Treatment was generally well tolerated, with no unexpected safety concerns.
Conclusions: A treatment algorithm based on olmesartan/amlodipine (± HCTZ) provides a high degree of BP control in patients with moderateto-severe hypertension. The open-label study design suggests similar results are obtainable in clinical practice.





Similar content being viewed by others
References
Kearney PM, Whelton M, Reynolds K, et al. Worldwide prevalence of hypertension: a systematic review. J Hypertens 2004 Jan; 22(1): 11–9
Ong KL, Cheung BM, Man YB, et al. Prevalence, awareness, treatment, and control of hypertension among United States adults 1999–2004. Hypertension 2007 Jan; 49(1): 69–75
Lewington S, Clarke R, Qizilbash N, et al. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002 Dec 14; 360(9349): 1903–13
Staessen JA, Li Y, Thijs L, et al. Blood pressure reduction and cardiovascular prevention: an update including the 2003–2004 secondary prevention trials. Hypertens Res 2005 May; 28(5): 385–407
Chobanian AV, Bakris GL, Black HR, et al. The seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA 2003 May 21; 289(19): 2560–72
Mancia G, De Backer G, Dominiczak A, et al. 2007 Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 2007 Jun; 25(6): 1105–87
Wang YR, Alexander GC, Stafford RS. Outpatient hypertension treatment, treatment intensification, and control in Western Europe and the United States. Arch Intern Med 2007 Jan 22; 167(2): 141–7
Volpe M, Tocci G, Trimarco B, et al. Blood pressure control in Italy: results of recent surveys on hypertension. J Hypertens 2007 Jul; 25(7): 1491–8
Ruilope LM, Agabiti-Rosei EA, Bakris GL, et al. Angiotensin receptor blockers: therapeutic targets and cardiovascular protection. Blood Press 2005; 14(4): 196–209
National Institute for Health and Clinical Excellence. Hypertension: management of hypertension in adults in primary care [online]. Available from URL: http://www.nice.org.uk/ CG034 [Accessed 2009 Mar 30]
Jamerson K, Weber MA, Bakris GL, et al. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359(23): 2417–28
Barrios V, Laeis P, Ammentorp B, et al. Olmesartan medoxomil (OLM) plus amlodipine (AML) provides BP reduction and improves goal rate attainment in patients (pts) with moderate-to-severe hypertension. J Hypertension 2008; 26Suppl. 1: S472–3
Chrysant SG, Melino M, Karki S, et al. The combination of olmesartan medoxomil and amlodipine besylate in controlling high blood pressure: COACH, a randomized, double-blind, placebo-controlled, 8-week factorial efficacy and safety study. Clin Ther 2008 Apr; 30(4): 587–604
Volpe M, Brommer P, Haag U, et al. Efficacy and tolerability of olmesartan medoxomil combined with amlodipine in patients with moderate to severe hypertension after amlodipine monotherapy. Clin Drug Invest 2009; 29(1): 11–25
Wolf-Maier K, Cooper RS, Kramer H, et al. Hypertension treatment and control in five European countries, Canada, and the United States. Hypertension 2004 Jan; 43(1): 10–7
Collins R, MacMahon S. Blood pressure, antihypertensive drug treatment and the risks of stroke and of coronary heart disease. Br Med Bull 1994 Apr; 50(2): 272–98
Mancia G, Grassi G. Systolic and diastolic blood pressure control in antihypertensive drug trials. J Hypertens 2002 Aug; 20(8): 1461–4
Burnier M, Brunner HR. Angiotensin II receptor antagonists in hypertension. Kidney Int Suppl 1998 Dec; 68: S107–111
ONTARGET Investigators, Yusuf S, Teo KK, et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med 2008 Apr 10; 358(15): 1547–59
Messerli FH. Vasodilatory edema: a common side effect of antihypertensive therapy. Curr Cardiol Rep 2002 Nov; 4(6): 479–82
Sica DA. Calcium channel blocker-related peripheral edema: can it be resolved? J Clin Hypertens (Greenwich) 2003 Jul–Aug;5(4): 291–24, 7
Atkins CE, Rausch WP, Gardner SY, et al. The effect of amlodipine and the combination of amlodipine and enalapril on the renin-angiotensin-aldosterone system in the dog. J Vet Pharmacol Ther 2007 Oct; 30(5): 394–400
Cappuccio FP, Markandu ND, Sagnella GA, et al. Effects of amlodipine on urinary sodium excretion, reninangiotensin-aldosterone system, atrial natriuretic peptide and blood pressure in essential hypertension. J Hum Hypertens 1991 Apr; 5(2): 115–9
Fabia MJ, Abdilla N, Oltra R, et al. Antihypertensive activity of angiotensin II AT1 receptor antagonists: a systematic review of studies with 24 h ambulatory blood pressure monitoring. J Hypertens 2007 Jul; 25(7): 1327–36
Poldermans D, Glazes R, Kargiannis S, et al. Tolerability and blood pressure-lowering efficacy of the combination of amlodipine plus valsartan compared with lisinopril plus hydrochlorothiazide in adult patients with stage 2 hypertension. Clin Ther 2007 Feb; 29(2): 279–89
Philipp T, Smith TR, Glazer R, et al. Two multicenter, 8-week, randomized, double-blind, placebo-controlled, parallel-group studies evaluating the efficacy and tolerability of amlodipine and valsartan in combination and as monotherapy in adult patients with mild to moderate essential hypertension. Clin Ther 2007 Apr; 29(4): 563–80
Kohlmann Jr O, Oigman W, Mion Jr D, et al. The “LOTHAR” study: evaluation of efficacy and tolerability of the fixed combination of amlodipine and losartan in the treatment of essential hypertension [in Portuguese]. Arq Bras Cardiol 2006 Jan; 86(1): 39–51
Acknowledgements
This study was sponsored by Daiichi Sankyo Europe GmbH, Munich, Germany.
The authors would like to thank Dr Peter Brommer of Gemeinschaftspraxis, Tann/Rhön, Germany for assistance in the conduct of this study; Dr Winfried Koch of HaaPACS GmbH, Schriesheim, Germany for assistance with the analysis of data presented in this report; and Jackie Campbell from Wolters Kluwer Health Medical Communications, Chester, UK who provided medical writing support on behalf of Daiichi-Sankyo Europe.
Massimo Volpe has served as a speaker for Daichii Sankyo, Guidotti and Malesci; has served on an International Advisory Board for Daichii Sankyo; and has received a research grant from Pfizer. Cristina Miele has no conflicts of interest to declare. Uwe Haag is a consultant to Daiichi Sankyo.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volpe, M., Miele, C. & Haag, U. Efficacy and Safety of a Stepped-Care Regimen Using Olmesartan Medoxomil, Amlodipine and Hydrochlorothiazide in Patients with Moderate-to-Severe Hypertension. Clin. Drug Investig. 29, 381–391 (2009). https://doi.org/10.2165/00044011-200929060-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200929060-00002