Abstract
Objective: To assess the efficacy of lamivudine treatment on hepatitis B e antigen (HBeAg) and/or hepatitis B surface antigen (HBsAg) seroconversion, on other virological and serological markers of response including hepatitis B virus (HBV) DNA and serum aminotransferases, and the safety of lamivudine treatment in hepatitis B patients.
Patients: This phase III open-label study evaluated the virological and biochemical response to lamivudine in 70 Portuguese patients with HBeAg positive chronic hepatitis B. Patients were treated with lamivudine 100mg once daily for 12 months.
Methods: Antiviral activity was assessed by measuring alanine aminotransferase (ALT)/aspartate aminotransferase (AST) levels at all protocol visits, and hepatitis B serology and HBV DNA were performed at baseline and at month 12 visits. Evaluation of safety and tolerance was based on clinical adverse events and laboratory analyses.
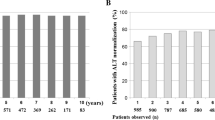
Results: The primary endpoint was virological response at month 12, defined as loss of detectable HBeAg from serum with a reduction of HBV DNA to undetectable levels, and this was observed in 19/69 (27.5%) of patients. Almost half of the patients were HBV DNA negative by this time. Mean ALT values decreased steadily during treatment and by 12 months 61% of patients had values within the normal range. HBeAg seroconversion (HBeAg negative, HBeAb positive) was achieved in 27.9% of patients by 12 months, although all patients remained HBsAg positive.
Conclusion: Lamivudine was well tolerated and the incidence of adverse events was similar to those reported in previous studies. Lamivudine treatment resulted in virological and biochemical improvements in HBeAg positive chronic hepatitis B patients, with HBeAg seroconversion in one-third of patients.




Similar content being viewed by others
References
Lee WM. Hepatitis B viras infection. N Engl J Med 1997; 337: 1733–45
De Jongh FE, Jamssea HL, De Man RA, et al. Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology 1992; 103: 1630–5
Hadziyannis SJ, Lieberman HM, Karvountzis GG, et al. Analysis of liver disease, nuclear HBcAg, viral replication, and hepatitis B virus DNA in liver and serum of HBeAg vs anti-HBe positive carriers of hepatitis B virus. Hepatology 1983; 3: 656–62
Alberti A, Pontisso P, Fattovich G, et al. Changes in serum hepatitis B viras (HBV) DNA positively in chronic HBV infection: Results of a long-term follow-up study of 138 patients. J Infect Dis 1986; 154: 562–9
Chu CM, Liaw YF. Intrahepatic distribution of hepatitis B surface and core antigens in chronic hepatitis B virus infection. Gastroenterology 1987; 92: 220–5
Liaw YF, Tai DI, Chu CM, et al. The development of cirrhosis in patients with chronic type B hepatitis: a prospective study. Hepatology 1988; 8: 493–6
Fattovich G, Brollo L, Giustina G, et al. Natural history and prognostic factors for chronic hepatitis type B. Gut 1991; 32: 294–8
Weissberg JI, Andres LL, Smith CI, et al. Survival in chronic hepatitis B: an analysis of 379 patients. Ann Intern Med 1984; 101: 613–6
Beasley RP. Hepatitis B virus: the major etiology of hepatocellular carcinoma. Cancer 1988; 61: 1942–56
Lai CL, Chien RN, Leung NWY, et al. A one year trial of lamivudine for chronic hepatitis B. N Engl J Med 1998; 339: 61–8
Dienstag J, Schiff ER, Wright TL, et al. Lamivudine as initial treatment for chronic hepatitis B in the United States. N Engl J Med 1999; 341: 1256–63
Schalm SW, Heathcote J, Cianciara J, et al. Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomised trial. Gut 2000; 46: 562–8
Goodman Z, Dhillon AP, Wu PC, et al. Lamivudine treatment reduces progression to cirrhosis in patients with chronic hepatitis B [abstract]. J Hepatol 1999; 30 Suppl. 1: 59
Hoofnagle JH, Alter HJ. Chronic viral hepatitis. In: Vyas GN, Dienstag JL, Hoofnagle GH, editors. Viral hepatitis and liver disease. New York: Grune & Stratton, 1984: 91–113
Liaw YF, Chu CM, Su IJ, et al. Clinical and histological events preceding hepatitis B e antigen seroconversion in chronic type B hepatitis. Gastroenterology 1983; 84: 216–9
Fattovich G, Rugge M, Brollo L, et al. Clinical, virologic and histologic outcome following seroconversion from HBeAg to anti-HBe in chronic hepatitis type B. Hepatology 1986; 6: 167–72
Hoofnagle JH, Dusheiko GM, Seeff LB, et al. Seroconversion from hepatitis B e antigen to antibody in chronic type B hepatitis. Ann Intern Med 1981; 94: 744–8
Davis GL, Hoofnagle JH, Waggoner JG. Spontaneous reactivation of chronic hepatitis B virus infection. Gastroenterology 1984; 86: 230–5
Perrillo RP, Brunt EM. Hepatic histologic and immunohistochemical changes in chronic hepatitis B after prolonged clearance of hepatitis B e antigen and hepatitis B surface antigen. Ann Intern Med 1991; 115: 113–5
Korenman J, Baker B, Waggoner J, et al. Long-Term remission of chronic hepatitis B after alpha-interferon therapy. Ann Intern Med 1991; 114: 629–34
Wong DKH, Cheung AM, O’Rourke K, et al. Effect of alpha-interferon treatment in patients with hepatitis B e antigen-positive chronic hepatitis B. Ann Intern Med 1993; 119: 312–23
Wong JB, Koff RS, Tinè Fabio, et al. Cost-effectiveness of interferon-α2b treatment for hepatitis B e antigen-positive chronic hepatitis B. Ann Intern Med 1995; 122: 664–75
Perrillo RP, Schiff ER, Davis GL, et al. A randomised, controlled trial of interferon alfa-2b alone and after prednisone withdrawal for the treatment of chronic hepatitis B. N Engl J Med 1990; 323: 295–301
Kassianides C, Di Bisceglie A, Hoofnagle J, et al. Alpha interferon therapy in patients with decompensated chronic type B hepatitis In: Zuckerman AJ, editor. Viral hepatitis and liver diseases. New York: Alan R. Liss, 1988: 840–843
Hoofnagle HH, Di Bisceglie AM, Waggoner JG, et al. Interferon alfa for patients with clinically apparent cirrhosis due to chronic hepatitis B. Gastroenterology 1993; 104: 1116–21
Nevens F, Goubau P, Van Eyken P, et al. Treatment of decompensated viral hepatitis B-induced cirrhosis with low doses of interferon alpha. Liver 1993; 13: 15–9
Schiff E, Karayalcin S, Grimm I, et al. A placebo controlled study of lamivudine and interferon alpha-2b in patients with chronic hepatitis B who previously failed interferon therapy [abstract]. Hepatology 1998; 28: 388A
Liaw YF, Leung NWY, Chang TT, et al. Effects of extended lamivudine therapy in Asian patients with chronic hepatitis B. Gastroenterology 2000; 119: 172–80
Rivkina A, Rybalov S. Chronic hepatitis B: current and future treatment options. Pharmacotherapy 2002; 22(6): 721–37
Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, et al. Adefovir dipivoxil for the treatment of hepatitis B e antigen negative chronic hepatitis B. N Eng J Med. 2003 Feb 27; 348(9): 800–7
Marcellin P, Chang TT, Lim SG, et al. Adefovir dipivoxil for the treatment of hepatitis B e antigen positive chronic hepatitis B. N Eng J Med. 2003 Feb 27; 348(9): 808–16
Di Bisceglie, Goodman ZD, Ishak KG, et al. Long-term clinical and histopathological follow-up of chronic post-transfusion hepatitis. Hepatology 1991; 14: 969–74
Niederau C, Heintges T, Lange S, et al. Long-term follow-up of HBeAg-positive patients treated with interferon alfa for chronic hepatitis B. N Engl J Med 1996; 3334: 1422–7
Leung NWY, Lai CL, Chang TT, et al. Extended lamivudine treatment in patients with chronic hepatitis B enhances hepatitis B e antigen seroconversion rates: results after three years of therapy. Hepatology 2001; 33: 1527–32
Chang TT, Lai CL, Liaw YF, et al. Incremental increases in HBeAg seroconversion and continued ALT normalization in Asian chronic HBV (CHB) patients treated with lamivudine for four years [abstract]. Antivir Ther 2000; 5 Suppl 1): 44
Acknowledgements
Funding for the study (protocol no. NUCPT01) was provided by Glaxo Wellcome Portugal.
We gratefully acknowledge technical assistance from Dr Graça Salcedo at the Laboratório de Endocrinologia e Patologia Clínica, Porto, Portugal and the other members of the Portuguese Lamivudine Study group: J. Areias, F. Calinas, A. Porto, A. Carvalho, D. Freitas, G. Macedo, R. Noronha, J. Cotter, A. Meliço-Silvestre, R. Peixe, J. Pratas, D. Barrote, R. Teixeira, F. Augusto, I. Carrilho, F. Campante, J. Velosa, L. Carvalho, M.A. Duarte, H. Guerreiro, C. Pires, A. Silva, I. Cotrim, F. Guedes, L. Tomé, M. Marcelino, C. Gonçalves, E. Ferreira, L. Matos, P. Peixe, J. Esteves, T. Valente, C. Simões, C. Marinho, L. Jasmins, M.J. Vieira, R. Marinho, P. Matos, J. Estevens, J. Carrasquinho, P. Parada, C. Teixeira.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Areias, J., Calinas, F., Porto, A. et al. One Year of Lamivudine Therapy for Portuguese Patients with Chronic Hepatitis B. Clin. Drug Investig. 23, 339–346 (2003). https://doi.org/10.2165/00044011-200323050-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200323050-00004