Summary
Abstract
Infliximab (Remicade®) is a chimeric monoclonal antibody against tumour necrosis factor (TNF)-α that has shown efficacy in Crohn’s disease and rheumatoid arthritis with a disease-modifying activity and rapid onset of action. It is administered intravenously, generally in a schedule with initial infusions at 0, 2 and 6 weeks, followed by administration once every 8 weeks.
Infliximab is effective in the treatment of patients with moderately to severely active Crohn’s disease with an inadequate response to other treatment options or those with fistulising disease. In combination with methotrexate, infliximab reduced signs and symptoms and delayed disease progression in patients with active, methotrexate-refractory rheumatoid arthritis and in those with early disease. The drug was generally well tolerated. Recrudescence of tuberculosis infection and worsening of heart failure and demyelinating disease are among some of the concerns with anti-TNFα therapy, requiring cautious use of these agents in high-risk patients.
Current data suggest that infliximab may be cost effective, especially when long-term clinical outcomes and burden of the diseases are taken into account. More robust, prospective pharmacoeconomic studies are required to better ascertain the cost effectiveness of infliximab.
Direct head-to-head comparative trials of infliximab with other biological agents are not yet available and would be helpful in determining with greater certainty the place of infliximab in the management of these diseases. Nonetheless, infliximab, like other biological agents, is a valuable treatment option in patients with moderately to severely active Crohn’s disease (including fistulising disease) or rheumatoid arthritis (including early disease).
Pharmacological Properties
Infliximab acts by binding to both the soluble and the transmembrane forms of TNFα and antagonising the proinflammatory actions of TNFα. It also downregulates other proinflammatory cytokines in patients with Crohn’s disease or rheumatoid arthritis. In addition, in in vitro studies, infliximab decreased the migration of inflammatory cells to the site of inflammation by reducing levels of chemokines and endothelial adhesion molecules. Importantly, infliximab does not produce a generalised suppression of cellular immune function.
The pharmacokinetics of infliximab are linear within the therapeutic dosage range. The steady-state volume of distribution, clearance, mean residence time and elimination half-life are all dose independent in patients with rheumatoid arthritis or Crohn’s disease. Infliximab has a long elimination half-life (median 8–12 days) and is still detectable in plasma up to 12 weeks after the last dose. However, repeated treatment does not lead to any clinically relevant systemic accumulation. Concomitant administration of methotrexate in patients with rheumatoid arthritis may sustain serum infliximab concentrations for longer. The pharmacokinetic profile in paediatric patients with Crohn’s disease was similar to that in adult patients.
Therapeutic Efficacy
The usual treatment regimen for infliximab in clinical trials involved intravenous administration as a single infusion, followed by subsequent doses 2 and 6 weeks later and then every 8 weeks (or every 4 weeks in one trial) thereafter. Although a broad range of infliximab doses (1–20 mg/kg) was used in these trials, no dose-response relationship was evident and, generally, no significant added benefit was reported when using doses higher than 5 mg/kg in Crohn’s disease and 3 mg/kg in rheumatoid arthritis patients, which are the currently recommended dosages. However, some patients may benefit from increasing the dose up to 10 mg/kg; increasing the frequency of administration to once every 4 weeks may be useful in some rheumatoid arthritis patients. Infliximab treatment induced a rapid onset of clinical response both in Crohn’s disease (within 2 weeks) and rheumatoid arthritis (48 hours) patients.
In randomised, double-blind trials, single infusions of infliximab (dose range 5–20 mg/kg) were effective in the induction of response in patients with moderately to severely active Crohn’s disease. In addition, repeated treatment with infliximab 5 or 10 mg/kg during longer-term therapy (54 weeks) sustained the clinical remission achieved with single infusions. Clinical response was accompanied by endoscopic evidence of mucosal healing. Infliximab treatment was also associated with significant improvements in the health-related quality of life (HR-QOL) of patients. Similarly, in patients with fistulising disease, repeated treatment (54 weeks) with infliximab 5 mg/kg was effective in maintaining remission in patients who had responded to an initial infusion. Sustained closure and healing of fistulas was seen in these patients.
Limited data available from double-blind and noncomparative trials in paediatric patients with Crohn’s disease with or without fistulas indicate that single infusions of infliximab were effective in reducing disease activity and that repeated treatment may be effective in producing a clinical response or inducing and maintaining remission.
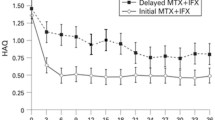
In a randomised, double-blind (54-week, followed by 54-week unblinded extension) trial in patients with active, methotrexate-refractory rheumatoid arthritis, infliximab (3 or 10 mg/kg) plus methotrexate was superior to methotrexate plus placebo in producing clinical response and attenuating radiographic progression of the disease. There was also a significant improvement in functional status and HR-QOL. Infliximab 3 or 6 mg/kg for 54 weeks, in combination with methotrexate, was also significantly more effective than methotrexate plus placebo in 1004 patients with early (≤3 year), methotrexate-naive rheumatoid arthritis with respect to improvements in signs and symptoms, disease progression, physical function and HR-QOL. The results of two smaller, double-blind, placebo-controlled studies using more sensitive imaging techniques (e.g. magnetic resonance imaging) have supported the notion that treatment with infliximab retarded disease progression in patients with early rheumatoid arthritis.
Tolerability
Infliximab was generally well tolerated for up to 102 weeks in clinical trials in patients with Crohn’s disease or rheumatoid arthritis, with the incidence of serious adverse events being generally similar in infliximab and placebo recipients. Infusion-related reactions are the most frequent adverse reactions to infliximab therapy. Although there appears to be no need for regular laboratory monitoring of infliximab therapy, patient monitoring, cautious use and/or discontinuation are recommended for other potentially serious adverse events possibly associated with TNFα antagonists as a class. These include tuberculosis and other serious infections, malignancy (particularly lymphoma), heart failure and CNS demyelination disorders.
Pharmacoeconomic Issues
Infliximab, like other biological agents, is a costly drug and generally more expensive than other available therapies for Crohn’ s disease. However, preliminary data have shown that it may be cost saving as a single-infusion induction regimen and cost effective as maintenance treatment of moderately to severely active Crohn’s disease. Moreover, pharmacoeconomic analyses have demonstrated that the incremental cost of infliximab maintenance therapy might be substantially offset by a reduction in other costs, such as those related to hospitalisation or surgery. In modelled cost-utility analyses, infliximab plus methotrexate was generally associated with acceptable incremental cost-effectiveness ratios relative to methotrexate monotherapy in patients with refractory rheumatoid arthritis. The cost effectiveness of infliximab in early disease has not been evaluated, but is expected to be improved when reductions with early treatment in the costs associated with the long-term disease outcomes are considered. Nevertheless, more robust pharmacoeconomic studies are needed before any conclusion may be drawn regarding the relative cost effectiveness of infliximab.








Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Head K, Jurenka JS. Inflammatory bowel disease. Part II: Crohn’s disease-pathophysiology and conventional and alternative treatment options. Altern Med Rev 2004; 9(4): 360–401
Lee DM, Weinblatt ME. Rheumatoid arthritis. Lancet 2001 Sep 15; 358: 903–11
Klinkhoff A. Biological agents for rheumatoid arthritis: targeting both physical function and structural damage. Drugs 2004; 64(12): 1267–83
Lim WC, Hanauer SB. Emerging biologic therapies in inflammatory bowel disease. Rev Gastroenterol Disord 2004 Spring; 4(2): 66–85
Onrust SV, Lamb HM. Infliximab: a review of its use in Crohn’s disease and rheumatoid arthritis. BioDrugs 1998 Nov; 10(5): 397–422
Keating GM, Perry CM. Infliximab: an updated review of its use in Crohn’s disease and rheumatoid arthritis. BioDrugs 2002; 16(2): 111–48
Markham A, Lamb HM. Infliximab: a review of its use in the management of rheumatoid arthritis. Drugs 2000 Jun; 59(6): 1341–59
Robinson DM, Keating GM. Infliximab: in ankylosing spondylitis. Drugs 2005; 65(9): 1283–91
Centocor Inc. Product information (US): Remicade for IV injection (infliximab 100mg vial) [online]. Available from URL: http://www.remicade.com [Accessed 2005 May 10]
Bell SJ, Kamm MA. Review article: the clinical role of anti-TNEalpha antibody treatment in Crohn’s disease. Aliment Pharmacol Ther 2000 May; 14(5): 501–14
Knight DM, Trinh H, Le J, et al. Construction and initial characterization of a mouse-human chimeric anti-TNF antibody. Mol Immunol 1993; 30(16): 1443–53
van den Brande JMH, Hommes DW, Peppelenbosch MP. Infliximab induced T lymphocyte apoptosis in Crohn’s disease. J Rheumatol Suppl 2005 Mar; 74: 26–30
Agnholt J, Dahlerup JF, Kaltoft K. The effect of etanercept and infliximab on the production of tumour necrosis factor alpha, interferon-gamma and GM-CSF in in vivo activated intestinal T lymphocyte cultures. Cytokine 2003 Aug 7; 23(3): 76–85
Scallon BJ, Moore MA, Trinh H, et al. Chimeric anti-TNFα monoclonal antibody cA2 binds recombinant transmembrane TNFα and activates immune effector functions. Cytokine 1995; 7(3): 251–9
Mitoma H, Horiuchi T, Hatta N, et al. Infliximab induces potent anti-inflammatory responses by outside-to-inside signals through transmembrane TNF-alpha. Gastroenterology 2005 Feb; 128(2): 376–92
Ohshima S, Saeki Y, Mima T, et al. Long-term follow-up of the changes in circulating cytokines, soluble cytokine receptors, and white blood cell subset counts in patients with rheumatoid arthritis (RA) after monoclonal anti-TNF alpha antibody therapy. J Clin Immunol 1999 Sep; 19(5): 305–13
Cornillie F, Shealy D, D’Haens G, et al. Infliximab induces potent anti-inflammatory and local immunomodulatory activity but no systemic immune suppression in patients with Crohn’s disease. Aliment Pharmacol Ther 2001 Apr; 15(4): 463–73
Cope AP, Londei M, Chu NR, et al. Chronic exposure to tumor necrosis factor (TNF) in vitro impairs the activation of T cells through the T cell receptor/CD3 complex; reversal in vivo by anti-TNF antibodies in patients with rheumatoid arthritis. J Clin Invest 1994 Aug; 94: 749–60
Guidi L, Costanzo M, Ciarniello M, et al. Increased levels of NF-κB inhibitors (IκBα and IκBγ) in the intestinal mucosa of Crohn’s disease patients during infliximab treatment. Int J Immunopathol Pharmacol 2005; 18(1): 155–64
Centocor B.V. Product information (EU): Remicade 100mg powder for concentrate for solution for infusion (infliximab 100mg vial) [online]. Available from URL: http:// www.emea.eu.int/ [Accessed 2005 May 10]
Kavanaugh A, St Clair EW, McCune WJ, et al. Chimeric anti-tumor necrosis factor-alpha monoclonal antibody treatment of patients with rheumatoid arthritis receiving methotrexate therapy. J Rheumatol 2000 Apr; 27(4): 841–50
Boyle A, Tawadros R, Zhu Y, et al. Comparative pharmacokinetics of single and multiple-dose infliximab in Crohn’s disease patients. Gastroenterology 2002 Apr; 122 Suppl. 1: A614–A 615. Plus poster presented at Digestive Disease Week and the 103rd Annual Meeting of the American Gastroenterological Association; 2002 May 19–23; San Francisco (CA)
St Clair EW, Wagner CL, Fasanmade AA, et al. The relationship of serum infliximab concentrations to clinical improvement in rheumatoid arthritis: results from ATTRACT, a multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum 2002 Jun; 46: 1451–9
Rutgeerts P, D’Haens G, Targan S, et al. Efficacy and safety of retreatment with anti-tumor necrosis factor antibody (infliximab) to maintain remission in Crohn’s disease. Gastroenterology 1999 Oct; 117(4): 761–9
Maini RN, Breedveld FC, Kalden JR, et al. Sustained improvement over two years in physical function, structural damage, and signs and symptoms among patients with rheumatoid arthritis treated with infliximab and methotrexate. Arthritis Rheum 2004 Apr; 50(4): 1051–65
Tanabe Seiyaku Co. Ltd. Product information (Japan): Remicade for IV infusion 100 (infliximab 100mg vial). 2003 Jul
Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet 2002 May 4; 359: 1541–9
Maini RN, Breedveld FC, Kalden JR, et al. Therapeutic efficacy of multiple intravenous infusions of anti-tumor necrosis factorα monoclonal antibody combined with low-dose weekly methotrexate in rheumatoid arthritis. Arthritis Rheum 1998; 41(9): 1552–63
Baldassano R, Braegger CP, Escher JC, et al. Infliximab (REMICADE) therapy in the treatment of pediatric Crohn’s disease. Am J Gastroenterol 2003 Apr; 98: 833–8
Lipsky PE, van der Heijde DM, St Clair EW, et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-Tumor Necrosis Factor Trial in Rheumatoid Arthritis with Concomitant Therapy Study Group. N Engl J Med 2000 Nov 30; 343(22): 1594–602
Targan SR, Hanauer SB, van Deventer SJH, et al. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor a for Crohn’s disease. N Engl J Med 1997; 337(15): 1029–35
D’haens G, Van Deventer S, Van Hogezand R, et al. Endoscopic and histological healing with infliximab anti-tumor necrosis factor antibodies in Crohn’s disease: a European multicenter trial. Gastroenterology 1999 May; 116(5): 1029–34
Present DH, Rutgeerts P, Targan S, et al. Infliximab for the treatment of fistulas in patients with Crohn’s disease. N Engl J Med 1999 May 6; 340(18): 1398–405
Sands BE, Anderson FH, Bernstein CN, et al. Infliximab maintenance therapy for fistulizing Crohn’s disease. N Engl J Med 2004 Feb 26; 350(9): 876–85
Kugathasan S, Werlin SL, Martinez A, et al. Prolonged duration of response to infliximab in early but not late pediatric Crohn’s disease. Am J Gastroenterol 2000 Nov; 95(11): 3189–94
Cezard JP, Nouaili N, Talbotec C, et al. A prospective study of the efficacy and tolerance of a chimeric antibody to tumor necrosis factors (remicade) in severe pediatric crohn disease. J Pediatr Gastroenterol Nutr 2003 May; 36(5): 632–6
deRidder L, Escher JC, Bouquet J, et al. Infliximab therapy in 30 patients with refractory pediatric Crohn disease with and without fistulas in the Netherlands. J Pediatr Gastroenterol Nutr 2004 Jul; 39(1): 46–52
Borrelli O, Bascietto C, Viola F, et al. Infliximab heals intestinal inflammatory lesions and restores growth in children with Crohn’s disease. Dig Liver Dis 2004 May; 36(5): 342–7
Asakura H, Yao T, Matsui T, et al. Efficacy of treatment with chimeric monoclonal antibody (Infliximab) to tumor necrosis factor-alpha for Crohn’s disease in Japan: evaluation by rapid turnover proteins, and radiologie and endoscopie findings. J Gastroenterol Hepatol 2001 Jul; 16(7): 763–9
Feagan BG, Yan S, Bala M, et al. The effects of infliximab maintenance therapy on health-related quality of life. Am J Gastroenterol 2003 Oct; 98(10): 2232–8
Rutgeerts P, Feagan BG, Lichtenstein GR, et al. Comparison of scheduled and episodic treatment stretegies of infliximab in Crohn’s disease. Gastroenterology 2004; 126: 402–13
St Clair EW, van der Heijde DMFM, Smolen JS, et al. Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: a randomised controlled trial. Arthritis Rheum 2004 Nov; 50(11): 3432–43
Quinn MA, Conaghan PG, O’Connor PJ, et al. Very early treatment with infliximab in addition to methotrexate in early, poor-prognosis rheumatoid arthritis reduces magnetic resonance imaging evidence of synovitis and damage, with sustained benefit after infliximab withdrawal: results from a twelve-month randomized, double-blind, placebo-controlled trial. Arthritis Rheum 2005 Jan; 52(1): 27–35
Taylor PC, Steuer A, Gruber J, et al. Comparison of ultrasonographic assessment of synovitis and joint vascularity with radiographic evaluation in a randomized, placebo-controlled study of infliximab therapy in early rheumatoid arthritis. Arthritis Rheum 2004 Apr; 50(4): 1107–16
Yocum D, Rahman M, Han C. Infliximab induces disease remission in patients with rheumatoid arthritis: results from the start clinical trial [abstract no. SAT0043]. Ann Rheum Dis 2005; 64 Suppl. III: 422. Plus poster presented at the European League Against Rheumatism Annual Congress; 2005 Jun 8–11; Vienna
Westhovens R, Rahman MU, Han C. Infliximab treatment results in a rapid improvement in all aspects of quality of life assessed by the sf-36 in patients with rheumatoid arthritis: results from the START trial [abstract no. FRI0450]. Ann Rheum Dis 2005; 64 Suppl. III: 392. Plus poster presented at the European League Against Rheumatism Annual Congress; 2005 Jun 8–11; Vienna
Westhovens R, Wolfe F, Rahman MU. Infliximab dose escalation in patients with rheumatoid arthritis: results from the START trial [abstract no. SAT0045]. Ann Rheum Dis 2005; 64 Suppl. III: 423. Plus poster presented at the European League Against Rheumatism Annual Congress; 2005 Jun 8–11; Vienna
Shergy WJ, Isern RA, Cooley DA, et al. Open label study to assess infliximab safety and timing of onset of clinical benefit among patients with rheumatoid arthritis. J Rheumatol 2002 Apr; 29: 667–77
Durez P, Van den Bosch F, Corluy L, et al. A dose adjustment in patients with rheumatoid arthritis not optimally responding to a standard dose of infliximab of 3 mg/kg every 8 weeks can be effective: a Belgian prospective study. Rheumatology (Oxford) 2005; 44(4): 465–8
Maini R, St Clair EW, Breedveld F, et al. Infliximab (chimeric anti-tumour necrosis factor alpha monoclonal antibody) versus placebo in rheumatoid arthritis patients receiving concomitant methotrexate: a randomised phase III trial. ATTRACT Study Group. Lancet 1999 Dec 4; 354(9194): 1932–9
Breedveld FC, Han C, Bala M, et al. Association between baseline radiographic damage and improvement in physical function after treatment of patients with rheumatoid arthritis. Ann Rheum Dis 2005 Jan; 64(1): 52–5
Breedveld FC, Emery P, Keystone E, et al. Infliximab in active early rheumatoid arthritis. Ann Rheum Dis 2004 Feb 1; 63(2): 149–55
Khanna D, McMahon M, Furst DE. Safety of tumour necrosis factor alpha antagonists. Drug Saf 2004; 27(5): 307–24
Cheifetz A, Smedley M, Martin S, et al. The incidence and management of infusion reactions to infliximab: a large center experience. Am J Gastroenterol 2003 Jun; 98(6): 1315–24
Schaible TF. Long term safety of infliximab. Can J Gastroenterol 2000 Sep; 14 Suppl. C: 29C–32C
Baert F, Noman M, Vermeire S, et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn’s disease. N Engl J Med 2003 Feb 13; 348: 601–8
Farrell RJ, Alsahli M, Jeen YT, et al. Intravenous hydrocortisone premedication reduces antibodies to infliximab in Crohn’s disease: a randomized controlled trial. Gastroenterology 2003 Apr; 124: 917–24
Hanauer SB, Rutgeerts PJ, D’Haens G, et al. Delayed hypersensitivity to infliximab (Remicaide®) re-infusion after a 2–4 year interval without treatment [abstract no. G3174]. Gastroenterology 1999 Apr; 116 (4 Pt 2): 731
Colombel JF, Loftus EVJr, Tremaine WJ, et al. The safety profile of infliximab in patients with Crohn’s disease: the Mayo Clinic experience in 500 patients. Gastroenterology 2004 Jan; 126(1): 19–31
Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor α-neutralizing agent. N Engl J Med 2001 Oct 21; 345(15): 1098–104
Scheinfeld N. A comprehensive review and evaluation of the side effects of the tumor necrosis factor alpha blockers etanercept, infliximab and adalimumab. J Dermatolog Treat 2004 Sep; 15(5): 280–94
Geborek P, Bladstrom A, Turesson C, et al. TNF blockers do not increase overall tumour risk in patients with rheumatoid arthritis, but may be associated with increased risk of lymphomas. Ann Rheum Dis 2005; 64(5): 699–703
Brown SL, Greene MH, Gershon SK, et al. Tumor necrosis factor antagonist therapy and lymphoma development: twenty-six cases reported to the Food and Drug Administration. Arthritis Rheum 2002 Dec; 46(12): 3151–8
Kwon HJ, Cote TR, Cuffe MS, et al. Case reports of heart failure after therapy with a tumor necrosis factor antagonist. Ann Intern Med 2003 May 20; 138(10): 807–11
Chung ES, Packer M, Lo KH, et al. Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-alpha, in patients with moderate-to-severe heart failure: results of the anti-TNF Therapy Against Congestive Heart Failure (ATTACH) trial. Circulation 2003; 107(25): 3133–40
Centocor Inc. Important drug warning [Dear Healthcare Professional letter; online]. Available from URL: http:// www.fda.gov/medwatch [Accessed 2005 May 16]
Mohan N, Edwards ET, Cupps TR, et al. Demyelination occurring during anti-tumor necrosis factor alpha therapy for inflammatory arthritides. Arthritis Rheum 2001 Dec; 44(12): 2862–9
Katz JA, Antoni C, Keenan GF, et al. Outcome of pregnancy in women receiving infliximab for the treatment of Crohn’s disease and rheumatoid arthritis. Am J Gastroenterol 2004 Dec; 99(12): 2385–92
Bodger K. Economic implications of biological therapies for Crohn’s Disease: review of infliximab. Pharmacoeconomics 2005; 23(9): 875–88
Lyseng-Williamson KA, Foster RH. Infliximab: a pharmacoeconomic review of its use in rheumatoid arthritis. Pharmacoeconomics 2004; 22(2): 107–32
Wong JB, Loftus Jr EV, Sandborn WJ, et al. Estimating the cost-effectiveness of infliximab for Crohn’s disease [abstract no. G0451]. Gastroenterology 1999 Apr; 116 (4 Pt 2): 104–5
Wong JB, Loftus EV, Sandborn WJ, et al. A cost-effectiveness analysis of maintenance infliximab for chronic active Crohn’s disease in Canada [abstract no. G398]. Gut 2004; 53 (Suppl. 6): 232
Colombel JF, Rutgeerts P, Yan S, et al. Infliximab maintenance treatment results in lower hospitalization rate in Crohn’s disease patients [abstract no. W1344]. Gastroenterology 2002 Apr; 122 Suppl. 1: A613
Wong JB, Ballina J, Fernandez-Sueiro JL, et al. Cost-effectiveness of infliximab for rheumatoid arthritis in Spain [abstract no. SAT0464]. Ann Rheum Dis 2004 Jun 12; 63 Suppl. 1: 520–1
Wong JB, Breedveld FC, Smolen JS, et al. Cost-effectiveness of 102-weeks of infliximab for rheumatoid arthritis [abstract no. 1551]. Arthritis Rheum 2001 Sep; 44 (9 Suppl.): S311
Wong JB, Breedveld FC, Smolen JS, et al. Estimating the cost-effectiveness of lifelong infliximab for rheumatoid arthritis [abstract no. SAT0300]. The European League Against Rheumatism Annual Congress; 2002 Jun 12–15; Stockholm
Wong JB, Branco JC, Cobrado N, et al. Cost-effectiveness of lifelong infliximab for rheumatoid arthritis in Portugal. Ann Rheum Dis 2003 Jul; 62 Suppl. 1: 360
Singh G, Mithal A, Tandon N. Costs of infliximab therapy for rheumatoid arthritis in a large state Medicaid program [abstract no. 368]. Arthritis Rheum 2004 Sep; 50 (9 Suppl.): 188
Maymo J, Perez C, Poveda JL, et al. Cost-analysis of infliximab and etanercept in the treatment of rheumatoid arthritis patients in Spain, based on a pharmacoeconomic model, and budgetary impact analysis of the results. Arthritis Rheum 2004 Sep; 50 (9 Suppl.): 43
Jaisson-Hot I, Flourie B, Descos L, et al. Management for severe Crohn’s disease: a lifetime cost-utility analysis. Int J Technol Assess Health Care 2004 Summer; 20(3): 274–9
Arseneau KO, Cohn SM, Cominelli F, et al. Cost-utility of initial medical management for Crohn’s disease perianal fistulae. Gastroenterology 2001 Jun; 120(7): 1640–56
Lichtenstein GR, Yan S, Bala M, et al. Infliximab maintenance treatment reduces hospitalizations, surgeries, and procedures in fistulizing Crohn’s disease. Gastroenterology 2005 Apr; 128(4): 862–9
Lichtenstein GR, Yan S, Bala M, et al. Remission in patients with Crohn’s disease is associated with improvement in employment and quality of life and a decrease in hospitalizations and surgeries. Am J Gastroenterol 2004 Jan; 99(1): 91–6
Wong JB, Singh G, Kavanaugh A. Estimating the cost-effectiveness of 54 weeks of infliximab for rheumatoid arthritis. Am J Med 2002 Oct 1; 113: 400–8
Kobelt G, Jonsson L, Young A, et al. The cost-effectiveness of infliximab (Remicade®) in the treatment of rheumatoid arthritis in Sweden and the United Kingdom based on the ATTRACT study. Rheumatology (Oxford) 2003 Feb; 42: 326–35
Ollendorf DA, Peterson AN, Doyle J, et al. Impact of leflunomide versus biologic agents on the costs of care for rheumatoid arthritis in a managed care population. Am J Manag Care 2002 May; 8 Suppl.: 203–13
Nuijten MJC, Engelfriet P, Duijn K, et al. A cost-cost study comparing etanercept with infliximab in rheumatoid arthritis. Pharmacoeconomics 2001; 19(10): 1051–64
Barbieri M, Wong JB, Drummond M. The cost effectiveness of infliximab for severe treatment-resistant rheumatoid arthritis in the UK. Pharmacoeconomics 2005; 23(6): 607–18
van der Heijde D, Dijkmans B, Geusens P, et al. Efficacy and safety of infliximab in patients with ankylosing spondylitis: results of a randomized, placebo-controlled trial (ASSERT). Arthritis Rheum 2005; 52(2): 582–91
Antoni CE, Kavanaugh A, Kirkham B, et al. Sustained benefits of infliximab therapy for dermatologic and articular manifestations of psoriatic arthritis: results from the infliximab multinational psoriatic arthritis controlled trial (IMPACT). Arthritis Rheum 2005; 52(4): 1227–36
Antoni C, Krueger GG, d Vlam K, et al. Infliximab improves signs and symptoms of psoriatic arthritis: results of the IMPACT 2 trial. Ann Rheum Dis 2005; 64(8): 1150–7
Rutgeerts P, Feagan BG, Olson A, et al. A randomized placebo-controlled trial of infliximab therapy for active ulcerative colitis: ACT I trial [abstract no. 689]. Gastroenterology 2005; 128 (4 Suppl. 2): 35–6
Sandborn WJ, Rachmilewitz D, Hanauer SB, et al. Infliximab induction and maintenance therapy for ulcerative colitis: the ACT 2 trial [abstract no. 688]. Gastroenterology 2005; 128 (4 Suppl. 2): 36
Loftus EVJr. Clinical epidemiology of inflammatory bowel disease: incidence, prevalence, and environmental influences. Gastroenterology 2004; 126(6): 1504–17
Rutgeerts PJ. Challenges in Crohn’s disease. Rev Gastroenterol Disord 2004; 4 Suppl. 3: S1–2
Yao T, Matsui T, Hiwatashi N. Crohn’s disease in Japan: diagnostic criteria and epidemiology. Dis Colon Rectum 2000; 43 (10 Suppl.): S85–93
Hanauer SB, Sandborn W, Practice Parameters Committee of the American College of Gastroenterology. Management of Crohn’s disease in adults. Am J Gastroenterol 2001 Mar; 96: 635–43
Panaccione R, Sandborn WJ. Medical therapy of Crohn disease. Curr Opin Gastroenterol 2004 Jul; 20(4): 351–9
Carter MJ, Lobo AJ, Travis SPL, on behalf of the IBD Section of the British Society of Gastroenterology. Guidelines for the management of inflammatory bowel disease in adults. Gut 2004; 53 Suppl. 5: V1–V16
Rutgeerts PJ. Reveiew article: the limitations of corticosteroid therapy in Crohn’s disease. Aliment Pharmacol Ther 2001 Oct; 15(10): 1515–25
American Gastroenterological Association Clinical Practice Committee. American Gastroenterological Association medical position statement: perianal Crohn’s disease. Gastroenterology 2003; 125(5): 1503–7
West RL, van der Woude CJ, Hansen BE, et al. Clinical and endosonographic effect of ciprofloxacin on the treatment of perianal fistulae in Crohn’s disease with infliximab: a double-blind placebo-controlled study. Aliment Pharmacol Ther 2004 Dec; 20(11–12): 1329–36
Hanauer SB, Cohen RD, Becker RV 3rd, et al. Advances in the management of Crohn’s disease: economic and clinical potential of infliximab. Clin Ther 1998; 20(5): 1009–28
Carswell CI, Perry CM, Ibbotson T. Management of rheumatoid arthritis: defining the role of leflunomide. Dis Manage Health Outcomes 2003; 11(1): 745–65
American College of Rheumatology Subcommittee on Rheumatoid Arthritis Guidelines. Guidelines for the management of rheumatoid arthritis: 2002 update. Arthritis Rheum 2002 Feb; 46: 328–46
Gabriel SE, Coyle D, Moreland LW. Clinical and economic review of disease-modifying antirheumatic drugs. Pharmacoeconomics 2001; 19(7): 715–28
Furst DE, Breedveld FC, Kalden JR, et al. Updated consensus statement on biological agents, specifically tumour necrosis factor alpha (TNFalpha) blocking agents and interleukin-1 receptor antagonist (IL-1ra), for the treatment of rheumatic diseases, 2004. Ann Rheum Dis 2004 Nov; 63 Suppl. 2: ii2–ii12
Ledingham J, Deighton C, on behalf of the British Society for Rheumatology Standards Guidelines and Audit Working Group (SGAWG). Update on the British Society for Rheumatology guidelines for prescribing TNFα blockers in adults with rheumatoid arthritis (update of previous guidelines of April 2001). Rheumatology (Oxford) 2005 Feb; 44(2): 157–63
Hochberg MC, Tracy JK, Flores RH. The comparative efficacy of anakinra, etanercept, infliximab and leflunomide when added to methotrexate in patients with active rheumatoid arthritis [abstract no. FRI0034]. The European League Against Rheumatism Annual Congress; 2002 Jun 12–15; Stockholm
Nurmohamed MT, Dijkmans BAC. Efficacy, tolerability and cost effectiveness of disease-modifying antirheumatic drugs and biologic agents in rheumatoid arthritis. Drugs 2005; 65(5): 661–94
Van der Bijl AE, Goekoop-Ruiterman YP, Breedveld FC, et al. Many early rheumatoid arthritis patients with a good clinical response to infliximab can discontinue anti-TNFα therapy without relapse [abstract no. OP0010]. Ann Rheum Dis 2005; 64 Suppl. III: 59
Fleischmann RM, Cohen SB, Moreland LW, et al. Methotrexate dosage reduction in patients with rheumatoid arthritis beginning therapy with infliximab: the infliaixmab Rheumatoid Arthritis Methotrexate Tapering (iRAMT) trial. Curr Med Res Opin 2005; 21(8): 1181–90
van Vollenhoven RF, Harju A, Brannemark S, et al. Treatment with infliximab (Remicade) when etanercept (Enbrel) has failed or vice versa: data from the STURE registry showing that switching tumour necrosis factor-α blockers can make sense. Ann Rheum Dis 2003; 62: 1195–8
Amgen and Wyeth Pharmaceuticals. Product information (US): Enbrel (etanercpet) for subcutaneous injection (single-use prefilled syringe [50 mg/mL] or multiple-use vial [25mg]) [online]. Available from URL: http://www.wyeth.com/content/ShowLabeling.asp?id=101 [Accessed 2005 Jun 17]
Abbott Laboratories. Product information (US): Humira (adalimumab) for subcutaneous injection (single-use prefilled syringe [40 mg/mL]) [online]. Available from URL: http:// www.rxabbott.com/pdf/humira.pdf [Accessed 2005 Jun 17]
Michaud K, Wolfe F. Reduced mortality among ra patients treated with anti-tnf therapy and methotrexate [abstract no. OP0095]. Ann Rheum Dis 2005; 64 Suppl. III: 87
Lichtenstein GR, Cohen RD, Feagan BG, et al. Safety of infliximab in Crohn’s disease: data from the 5000-patient TREAT Registry [abstract no. 439]. Gastroenterology 2004 Apr; 126 (4 Suppl. 2): 54
Author information
Authors and Affiliations
Corresponding author
Additional information
Various sections of the manuscript reviewed by: H. Asakura, International Medical Information Center, Shinjukuku, Tokyo, Japan; F. Breedveld, Department of Rheumatology, Leiden University Hospital, Leiden, The Netherlands; T. Ikawa, Osaka Rehabilitation Hospital, Osaka, Japan; P.J. Rutgeerts, Gastroenterology Section, Faculty of Medicine, University of Leuven, Leuven, Belgium; S. Schreiber, Department of General Internal Medicine and Institute for Clinical Molecular Biology, Christian-Albrechts-University, Schittenhelmstrasse, Kiel, Germany; W.S. Selby, Department of Medicine, University of Sydney, Sydney, Australia; G. Valesini, Division of Rheumatology, La Sapienza University, Rome, Italy.
Data Selection
Sources: Medical literature published in any language since 1980 on ‘infliximab’, identified using MEDLINE and EMBASE, supplemented by AdisBase (a proprietary database of Adis International). Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug.
Search strategy: MEDLINE and EMBASE search terms were ‘infliximab’ and (‘Crohn disease’ or ‘rheumatoid arthritis’). AdisBase search terms were (‘infliximab’ or ‘anti-tnf-monoclonal-antibody’) and (‘Crohn’s disease’ or ‘rheumatoid arthritis’). Searches were last updated 19 September 2005.
Selection: Studies in patients with Crohn’s disease or rheumatoid arthritis who received intravenous infliximab. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. Relevant pharmacodynamic and pharmacokinetic data are also included.
Index terms: Infliximab, Crohn’s disease, rheumatoid arthritis, pharmacodynamics, pharmacokinetics, therapeutic use, pharmacoeconomics, tolerability.
Rights and permissions
About this article
Cite this article
Siddiqui, M.A.A., Scott, L.J. Infliximab. Drugs 65, 2179–2208 (2005). https://doi.org/10.2165/00003495-200565150-00014
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200565150-00014