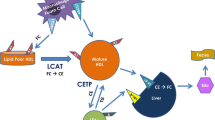
Abstract
Low high-density lipoprotein (HDL) cholesterol is an important risk factor for coronary heart disease (CHD). In vitro, HDL exerts several potentially anti-atherogenic activities. HDLs mediate the reverse cholesterol transport (RCT) from peripheral cells to the liver, inhibit oxidation of low-density lipoprotein (LDL), adhesion of monocytes to the endothelium, apoptosis of vascular endothelial and smooth muscle cells and platelet activation, and stimulate the endothelial secretion of vasoactive substances as well as smooth muscle cell proliferation. Hence, raising HDL-cholesterol levels has become an interesting target for anti-atherosclerotic drug therapy. Levels of HDL cholesterol and the composition of HDL subclasses in plasma are regulated by apolipoproteins, lipolytic enzymes, lipid transfer proteins, receptors and cellular transporters. The interplay of these factors leads to RCT and determines the composition and, thereby, the anti-atherogenic properties of HDL. Several inborn errors of metabolism, as well as genetic animal models, are characterised by both elevated HDL cholesterol and increased rather than decreased cardiovascular risk. These findings suggest that the mechanism of HDL modification rather than simply increasing HDL cholesterol determine the efficacy of anti-atherosclerotic drug therapy.
In several controlled and prospective intervention studies, patients with low HDL cholesterol and additional risk factors benefited from treatment with fibric acid derivatives (fibrates) or HMG-CoA reductase inhibitors (statins). However, only in some trials was prevention of coronary events in patients with low HDL cholesterol and hypertriglyceridaemia related to an increase in HDL cholesterol. We discuss the clinical and metabolic effects of fibrates, statins, nicotinic acid and sex steroids, and present novel therapeutic strategies that show promise in modifying HDL metabolism.
In conclusion, HDL-cholesterol levels increase only moderately after treatment with currently available drugs and do not necessarily correlate with the functionality of HDL. Therefore, the anti-atherosclerotic therapy of high-risk cardiovascular patients should currently be focused on the correction of other risk factors present besides low HDL cholesterol. However, modification of HDL metabolism and improvement of RCT remain an attractive target for the development of new regimens of anti-atherogenic drug therapy.





Similar content being viewed by others
References
Lusis AJ. Atherosclerosis. Nature 2000; 407: 233–41
Glass CK, Witztum JL. Atherosclerosis: the road ahead. Cell 2001; 104: 503–16
Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 1994; 344: 1383-9
Shepherd J, Cobbe SM, Ford I, et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia: West of Scotland Coronary Prevention Study Group. N Engl J Med 1995; 333: 1301–7
Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels: Cholesterol and Recurrent Events Trial investigators. N Engl J Med 1996; 335: 1001–9
Prevention of cardiovascular events and death with pravastatin in patients with coronary heart disease and a broad range of initial cholesterol levels. The Long-Term Intervention with Pravastatin in Ischaemic Disease (LIPID) Study Group. N Engl J Med 1998; 339: 1349–57
Downs JR, Clearfield M, Weis S, et al. Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study. JAMA 1998; 279: 1615–22
MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20 536 high-risk individuals: a randomised placebo-controlled trial. Lancet 2002; 360: 7-22
Gordon DJ, Rifkind BM. High-density lipoprotein: the clinical implications of recent studies. N Engl J Med 1989; 321: 1311–6
Genest JJ, McNamara JR, Salem DN, et al. Prevalence of risk factors in men with premature coronary artery disease. Am J Cardiol 1991; 67: 1185–9
Bobak M, Hense HW, Kark J, et al. An ecological study of determinants of coronary heart disease rates: a comparison of Czech, Bavarian and Israeli men. Int J Epidemiol 1999; 28: 437–44
Hergenc G, Schulte H, Assmann G, et al. Associations of obesity markers, insulin, and sex hormones with HDL-cholesterol levels in Turkish and German individuals. Atherosclerosis 1999; 145: 147–56
Manninen V, Tenkanen L, Koskinen P, et al. Joint effects of serum triglyceride and LDL cholesterol and HDL cholesterol concentrations on coronary heart disease risk in the Helsinki Heart Study: implications for treatment. Circulation 1992; 85: 37–45
Rubins HB, Robins SJ, Collins D, et al. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol: Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med 1999; 341: 410–8
Gotto Jr AM, Whitney E, Stein EA, et al. Relation between baseline and on-treatment lipid parameters and first acute major coronary events in the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS). Circulation 2000; 101: 477–84
Executive summary of the third report of the National Cholesterol Education Program (NCEP): expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001; 285: 2486-97
International Task Force for Prevention of Coronary Heart Disease. Coronary heart disease: reducing the risk: the scientific background to primary and secondary prevention of coronary heart disease: a world wide view. Nutr Metab Cardiovasc Dis 1998; 8: 205–71
Fruchart JC, Brewer Jr HB, Leitersdorf E. Consensus for the use of fibrates in the treatment of dyslipoproteinemia and coronary heart disease: Fibrate Consensus Group. Am J Cardiol 1998; 81: 912–7
Barter PJ, Baker PW, Rye KA. Effect of high-density lipoproteins on the expression of adhesion molecules in endothelial cells. Curr Opin Lipidol 2002; 13: 285–8
Nofer JR, Fobker M, Hobbel G, et al. Activation of phosphatidylinositol-specific phospholipase C by HDL-associated lysosphingolipid: involvement in mitogenesis but not in cholesterol efflux. Biochemistry 2000; 39: 15199–207
Nofer JR, Levkau B, Wolinska I, et al. Suppression of endothelial cell apoptosis by high density lipoproteins (HDL) and HDL-associated lysosphingolipids. J Biol Chem 2001; 276: 34480–5
von Eckardstein A, Nofer JR, Assmann G. High density lipoproteins and arteriosclerosis: role of cholesterol efflux and reverse cholesterol transport. Arterioscler Thromb Vasc Biol 2001; 21: 13–27
Segrest JP, Jackson RL, Morrisett JD, et al. A molecular theory of lipid-protein interactions in the plasma lipoproteins. FEBS Lett 1974; 38: 247–58
Cushley RJ, Okon M. NMR studies of lipoprotein structure. Annu Rev Biophys Biomol Struct 2002; 31: 177–206
Tricerri MA, Behling Agree AK, Sanchez SA, et al. Arrangement of apolipoprotein A-I in reconstituted high-density lipoprotein disks: an alternative model based on fluorescence resonance energy transfer experiments. Biochemistry 2001; 40: 5065–74
Segrest JP, Harvey SC, Zannis V. Detailed molecular model of apolipoprotein A-I on the surface of high-density lipoproteins and its functional implications. Trends Cardiovasc Med 2000; 10: 246–52
Jonas A. Lecithin cholesterol acyltransferase. Biochim Biophys Acta 2000; 1529: 245–56
Huuskonen J, Olkkonen VM, Jauhiainen M, et al. The impact of phospholipid transfer protein (PLTP) on HDL metabolism. Atherosclerosis 2001; 155: 269–81
Karkkainen M, Oka T, Olkkonen VM, et al. Isolation and partial characterization of the inactive and active forms of human plasma phospholipid transfer protein (PLTP). J Biol Chem 2002; 277: 15413–8
Jiang XC, Qin S, Qiao C, et al. Apolipoprotein B secretion and atherosclerosis are decreased in mice with phospholipid-transfer protein deficiency. Nat Med 2001; 7: 847–52
Yamashita S, Hirano K, Sakai N, et al. Molecular biology and pathophysiological aspects of plasma cholesteryl ester transfer protein. Biochim Biophys Acta 2000; 1529: 257–75
Wang X, Driscoll DM, Morton RE. Molecular cloning and expression of lipid transfer inhibitor protein reveals its identity with apolipoprotein F. J Biol Chem 1999; 274: 1814–20
Durrington PN, Mackness B, Mackness MI. Paraoxonase and atherosclerosis. Arterioscler Thromb Vasc Biol 2001; 21: 473–80
Nofer JR, Kehrel B, Fobker M, et al. HDL and arteriosclerosis: beyond reverse cholesterol transport. Atherosclerosis 2002; 161: 1–16
Gallagher AR, Hidaka S, Gretz N, et al. Molecular basis of autosomal-dominant polycystic kidney disease. Cell Mol Life Sci 2002; 59: 682–93
Hatters DM, Howlett GJ. The structural basis for amyloid formation by plasma apolipoproteins: a review. Eur Biophys J 2002; 31: 2–8
Van Lenten BJ, Hama SY, de Beer FC, et al. Anti-inflammatory HDL becomes pro-inflammatory during the acute phase response: loss of protective effect of HDL against LDL oxidation in aortic wall cell cocultures. J Clin Invest 1995; 96: 2758–67
von Eckardstein A, Huang Y, Assmann G. Physiological role and clinical relevance of high-density lipoprotein subclasses. Curr Opin Lipidol 1994; 5: 404–16
Barrans A, Jaspard B, Barbaras R, et al. Pre-beta HDL: structure and metabolism. Biochim Biophys Acta 1996; 1300: 73–85
Fielding CJ, Fielding PE. Molecular physiology of reverse cholesterol transport. J Lipid Res 1995; 36: 211–28
Huang Y, von Eckardstein A, Wu S, et al. A plasma lipoprotein containing only apolipoprotein E and with gamma mobility on electrophoresis releases cholesterol from cells. Proc Natl Acad Sci U S A 1994; 91: 1834–8
Duverger N, Ghalim N, Ailhaud G, et al. Characterization of apoA-IV-containing lipoprotein particles isolated from human plasma and interstitial fluid. Arterioscler Thromb 1993; 13: 126–32
von Eckardstein A, Huang Y, Wu S, et al. Lipoproteins containing apolipoprotein A-IV but not apolipoprotein A-I take up and esterify cell-derived cholesterol in plasma. Arterioscler Thromb Vasc Biol 1995; 15: 1755–63
Asztalos BF, Sloop CH, Wong L, et al. Comparison of apo A-I-containing subpopulations of dog plasma and prenodal peripheral lymph: evidence for alteration in subpopulations in the interstitial space. Biochim Biophys Acta 1993; 1169: 301–4
Nanjee MN, Cooke CJ, Olszewski WL, et al. Concentrations of electrophoretic and size subclasses of apolipoprotein A-I-containing particles in human peripheral lymph. Arterioscler Thromb Vasc Biol 2000; 20: 2148–55
Bjorkhem I, Diczfalusy U. Oxysterols: friends, foes, or just fellow passengers? Arterioscler Thromb Vasc Biol 2002; 22: 734–42
Langer C, Huang Y, Cullen P, et al. Endogenous apolipoprotein E modulates cholesterol efflux and cholesteryl ester hydrolysis mediated by high-density lipoprotein-3 and lipid-free apolipoproteins in mouse peritoneal macrophages. J Mol Med 2000; 78: 217–27
von Bahr S, Movin T, Papadogiannakis N, et al. Mechanism of accumulation of cholesterol and cholestanol in tendons and the role of sterol 27-hydroxylase (CYP27A1). Arterioscler Thromb Vasc Biol 2002; 22: 1129–35
Curtiss LK, Boisvert WA. Apolipoprotein E and atherosclerosis. Curr Opin Lipidol 2000; 11: 243–51
Rothblat GH, Llera-Moya M, Atger V, et al. Cell cholesterol efflux: integration of old and new observations provides new insights. J Lipid Res 1999; 40: 781–96
Mendez AJ, Anantharamaiah GM, Segrest JP, et al. Synthetic amphipathic helical peptides that mimic apolipoprotein A-I in clearing cellular cholesterol. J Clin Invest 1994; 94: 1698–705
Yancey PG, Bielicki JK, Johnson WJ, et al. Efflux of cellular cholesterol and phospholipid to lipid-free apolipoproteins and class A amphipathic peptides. Biochemistry 1995; 34: 7955–65
Gillotte KL, Zaiou M, Lund-Katz S, et al. Apolipoprotein-mediated plasma membrane microsolubilization: role of lipid affinity and membrane penetration in the efflux of cellular cholesterol and phospholipid. J Biol Chem 1999; 274: 2021–8
Chroni A, Liu T, Gorshkova I, et al. The central helices of APOA-I can promote ABCA1-mediated lipid efflux: amino acid residues 220–231 of the wild-type APOA-I are required for lipid efflux in vitro and HDL formation in vivo. J Biol Chem 2003; 278: 6719–30
Ji Y, Jian B, Wang N, et al. Scavenger receptor BI promotes high density lipoprotein-mediated cellular cholesterol efflux. J Biol Chem 1997; 272: 20982–5
Llera-Moya M, Rothblat GH, Connelly MA, et al. Scavenger receptor BI (SR-BI) mediates free cholesterol flux independently of HDL tethering to the cell surface. J Lipid Res 1999; 40: 575–80
Liu T, Krieger M, Kan HY, et al. The effects of mutations in helices 4 and 6 of ApoA-I on scavenger receptor class B type I (SR-BI)-mediated cholesterol efflux suggest that formation of a productive complex between reconstituted high density lipoprotein and SR-BI is required for efficient lipid transport. J Biol Chem 2002; 277: 21576–84
Silver DL, Tall AR. The cellular biology of scavenger receptor class B type I. Curr Opin Lipidol 2001; 12: 497–504
Trigatti B, Rayburn H, Vinals M, et al. Influence of the high density lipoprotein receptor SR-BI on reproductive and cardiovascular pathophysiology. Proc Natl Acad Sci U S A 1999; 96: 9322–7
Braun A, Trigatti BL, Post MJ, et al. Loss of SR-BI expression leads to the early onset of occlusive atherosclerotic coronary artery disease, spontaneous myocardial infarctions, severe cardiac dysfunction, and premature death in apolipoprotein E-deficient mice. Circ Res 2002; 90: 270–6
Arai T, Wang N, Bezouevski M, et al. Decreased atherosclerosis in heterozygous low density lipoprotein receptor-deficient mice expressing the scavenger receptor BI transgene. J Biol Chem 1999; 274: 2366–71
Kozarsky KF, Donahee MH, Glick JM, et al. Gene transfer and hepatic overexpression of the HDL receptor SR-BI reduces atherosclerosis in the cholesterol-fed LDL receptor-deficient mouse. Arterioscler Thromb Vasc Biol 2000; 20: 721–7
Oram JF. ATP-binding cassette transporter A1 and cholesterol trafficking. Curr Opin Lipidol 2002; 13: 373–81
Burgess JW, Frank PG, Franklin V, et al. Deletion of the C-terminal domain of apolipoprotein A-I impairs cell surface binding and lipid efflux in macrophage. Biochemistry 1999; 38: 14524–33
Hamon Y, Broccardo C, Chambenoit O, et al. ABC1 promotes engulfment of apoptotic cells and transbilayer redistribution of phosphatidylserine. Nat Cell Biol 2000; 2: 399–406
Chambenoit O, Hamon Y, Marguet D, et al. Specific docking of apolipoprotein A-I at the cell surface requires a functional ABCA1 transporter. J Biol Chem 2001; 276: 9955–60
Smith JD, Waelde C, Horwitz A, et al. Evaluation of the role of phosphatidylserine translocase activity in ABCA1-mediated lipid efflux. J Biol Chem 2002; 277: 17797–803
Neufeld EB, Remaley AT, Demosky SJ, et al. Cellular localization and trafficking of the human ABCA1 transporter. J Biol Chem 2001; 276: 27584–90
Chen W, Sun Y, Welch C, et al. Preferential ATP-binding cassette transporter A1-mediated cholesterol efflux from late endosomes/lysosomes. J Biol Chem 2001; 276: 43564–9
Takahashi Y, Smith JD. Cholesterol efflux to apolipoprotein AI involves endocytosis and resecretion in a calcium-dependent pathway. Proc Natl Acad Sci U S A 1999; 96: 11358–63
Schmitz G, Assmann G, Robenek H, et al. Tangier disease: a disorder of intracellular membrane traffic. Proc Natl Acad Sci U S A 1985; 82: 6305–9
Orso E, Broccardo C, Kaminski WE, et al. Transport of lipids from golgi to plasma membrane is defective in tangier disease patients and Abc1-deficient mice. Nat Genet 2000; 24: 192–6
von Eckardstein A, Langer C, Engel T, et al. ATP binding cassette transporter ABCA1 modulates the secretion of apolipoprotein E from human monocyte-derived macrophages. FASEB J 2001; 15: 1555–61
Zhou X, Engel T, Goepfert C, et al. The ATP binding cassette transporter A1 contributes to the secretion of interleukin 1beta from macrophages but not from monocytes. Biochem Biophys Res Commun 2002; 291: 598–604
Assmann G, von Eckardstein A, Brewer Jr HB. The metabolic and molecular bases of inherited disease. 8th ed. New York: McGraw-Hill, 2000: 2937–60
Santamarina-Fojo S, Remaley AT, Neufeld EB, et al. Regulation and intracellular trafficking of the ABCA1 transporter. J Lipid Res 2001; 42: 1339–45
Lawn RM, Wade DP, Garvin MR, et al. The Tangier disease gene product ABC1 controls the cellular apolipoprotein-mediated lipid removal pathway. J Clin Invest 1999; 104: R25–31
Langmann T, Klucken J, Reil M, et al. Molecular cloning of the human ATP-binding cassette transporter 1 (hABC1): evidence for sterol-dependent regulation in macrophages. Biochem Biophys Res Commun 1999; 257: 29–33
Costet P, Luo Y, Wang N, et al. Sterol-dependent transactivation of the ABC1 promoter by the liver X receptor/retinoid X receptor. J Biol Chem 2000; 275: 28240–5
Repa JJ, Turley SD, Lobaccaro JA, et al. Regulation of absorption and ABC1-mediated efflux of cholesterol by RXR heterodimers. Science 2000; 289: 1524–9
Uehara Y, Engel T, Li Z, et al. Polyunsaturated fatty acids and acetoacetate down-regulate the expression of the ATP binding cassette transporter Al. Diabetes 2002; 51: 2922–8
Wang Y, Oram JF. Unsaturated fatty acids inhibit cholesterol efflux from macrophages by increasing degradation of ATP-binding cassette transporter A 1. J Biol Chem 2002; 277: 5692–7
Baranova I, Vishnyakova T, Bocharov A, et al. Lipopolysaccharide down regulates both scavenger receptor B1 and ATP binding cassette transporter A1 in RAW cells. Infect Immun 2002; 70: 2995–3003
Wang XQ, Panousis CG, Alfaro ML, et al. Interferon-gamma-mediated downregulation of cholesterol efflux and ABC1 expression is by the Stat1 pathway. Arterioscler Thromb Vasc Biol 2002; 22: e5–9
Panousis CG, Evans G, Zuckerman SH. TGF-beta increases cholesterol efflux and ABC-1 expression in macrophage-derived foam cells: opposing the effects of IFN-gamma. J Lipid Res 2001; 42: 856–63
Navab M, Hama SY, Ready ST, et al. Oxidized lipids as mediators of coronary heart disease. Curr Opin Lipidol 2002; 13: 363–72
Mackness B, Durrington PN, Mackness MI. The paraoxonase gene family and coronary heart disease. Curr Opin Lipidol 2002; 13: 357–62
Aviram M, Hardak E, Vaya J, et al. Human serum paraoxonases (PON1) Q and R selectively decrease lipid peroxides in human coronary and carotid atherosclerotic lesions: PON1 esterase and peroxidase-like activities. Circulation 2000; 101: 2510–7
Shih DM, Gu L, Xia YR, et al. Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature 1998; 394: 284–7
Shih DM, Xia YR, Wang XP, et al. Combined serum paraoxonase knockout/apolipoprotein E knockout mice exhibit increased lipoprotein oxidation and atherosclerosis. J Biol Chem 2000; 275: 17527–35
Tward A, Xia YR, Wang XP, et al. Decreased atherosclerotic lesion formation in human serum paraoxonase transgenic mice. Circulation 2002; 106: 484–90
Gowri MS, Van der Westhuyzen DR, Bridges SR, et al. Decreased protection by HDL from poorly controlled type 2 diabetic subjects against LDL oxidation may Be due to the abnormal composition of HDL. Arterioscler Thromb Vasc Biol 1999; 19: 2226–33
Garner B, Waldeck AR, Witting PK, et al. Oxidation of high density lipoproteins: II, evidence for direct reduction of lipid hydroperoxides by methionine residues of apolipoproteins AI and AII. J Biol Chem 1998; 273: 6088–95
Biegelsen ES, Loscalzo J. Endothelial function and atherosclerosis. Coron Artery Dis 1999; 10: 241–56
Zhang X, Zhao SP, Li XP, et al. Endothelium-dependent and — independent functions are impaired in patients with coronary heart disease. Atherosclerosis 2000; 149: 19–24
Li XP, Zhao SP, Zhang XY, et al. Protective effect of high density lipoprotein on endothelium-dependent vasodilatation. Int J Cardiol 2000; 73: 231–6
Kaufmann PA, Gnecchi-Ruscone T, Schafers KP, et al. Low density lipoprotein cholesterol and coronary microvascular dysfunction in hypercholesterolemia. J Am Coll Cardiol 2000; 36: 103–9
Kuhn FE, Mohler ER, Satler LF, et al. Effects of high-density lipoprotein on acetylcholine-induced coronary vasoreactivity. Am J Cardiol 1991; 68: 1425–30
Zeiher AM, Schachlinger V, Hohnloser SH, et al. Coronary atherosclerotic wall thickening and vascular reactivity in humans: elevated high-density lipoprotein levels ameliorate abnormal vasoconstriction in early atherosclerosis. Circulation 1994; 89: 2525–32
Spieker LE, Sudano I, Hurlimann D, et al. High-density lipoprotein restores endothelial function in hypercholesterolemic men. Circulation 2002; 105: 1399–402
Matsuda Y, Hirata K, Inoue N, et al. High density lipoprotein reverses inhibitory effect of oxidized low density lipoprotein on endothelium-dependent arterial relaxation. Circ Res 1993; 72: 1103–9
Galle J, Ochslen M, Schollmeyer P, et al. Oxidized lipoproteins inhibit endothelium-dependent vasodilation: effects of pressure and high-density lipoprotein. Hypertension 1994; 23: 556–64
Yuhanna IS, Zhu Y, Cox BE, et al. High-density lipoprotein binding to scavenger receptor-BI activates endothelial nitric oxide synthase. Nat Med 2001; 7: 853–7
Fleisher LN, Tall AR, Witte LD, et al. Stimulation of arterial endothelial cell prostacyclin synthesis by high density lipoproteins. J Biol Chem 1982; 257: 6653–5
Myers DE, Huang WN, Larkins RG. Lipoprotein-induced prostacyclin production in endothelial cells and effects of lipoprotein modification. Am J Physiol 1996; 271: C1504–11
Cockerill GW, Saklatvala J, Ridley SH, et al. High-density lipoproteins differentially modulate cytokine-induced expression of E-selectin and cyclooxygenase-2. Arterioscler Thromb Vasc Biol 1999; 19: 910–7
Vinals M, Martinez-Gonzalez J, Badimon JJ, et al. HDL-induced prostacyclin release in smooth muscle cells is dependent on cyclooxygenase-2 (Cox-2). Arterioscler Thromb Vasc Biol 1997; 17: 3481–8
Jambou D, Dejour N, Bayer P, et al. Effect of human native low-density and high-density lipoproteins on prostaglandin production by mouse macrophage cell line P388 D1: possible implications in pathogenesis of atherosclerosis. Biochim Biophys Acta 1993; 1168: 115–21
Oravec S, Demuth K, Myara I, et al. The effect of high density lipoprotein subfractions on endothelial eicosanoid secretion. Thromb Res 1998; 92: 65–71
Navab M, Imes SS, Hama SY, et al. Monocyte transmigration induced by modification of low density lipoprotein in cocultures of human aortic wall cells is due to induction of monocyte chemotactic protein 1 synthesis and is abolished by high density lipoprotein. J Clin Invest 1991; 88: 2039–46
Cockerill GW, Rye KA, Gamble JR, et al. High-density lipoproteins inhibit cytokine-induced expression of endothelial cell adhesion molecules. Arterioscler Thromb Vasc Biol 1995; 15: 1987–94
Calabresi L, Franceschini G, Sirtori CR, et al. Inhibition of VCAM-1 expression in endothelial cells by reconstituted high density lipoproteins. Biochem Biophys Res Commun 1997; 238: 61–5
Xia P, Vadas MA, Rye KA, et al. High density lipoproteins (HDL) interrupt the sphingosine kinase signaling pathway: a possible mechanism for protection against atherosclerosis by HDL. J Biol Chem 1999; 274: 33143–7
Ashby DT, Rye KA, Clay MA, et al. Factors influencing the ability of HDL to inhibit expression of vascular cell adhesion molecule-1 in endothelial cells. Arterioscler Thromb Vasc Biol 1998; 18: 1450–5
Baker PW, Rye KA, Gamble JR, et al. Phospholipid composition of reconstituted high density lipoproteins influences their ability to inhibit endothelial cell adhesion molecule expression. J Lipid Res 2000; 41: 1261–7
Tamagaki T, Sawada S, Imamura H, et al. Effects of high-density lipoproteins on intracellular pH and proliferation of human vascular endothelial cells. Atherosclerosis 1996; 123: 73–82
Suc I, Escargueil-Blanc I, Troly M, et al. HDL and ApoA prevent cell death of endothelial cells induced by oxidized LDL. Arterioscler Thromb Vasc Biol 1997; 17: 2158–66
Sugano M, Tsuchida K, Makino N. High-density lipoproteins protect endothelial cells from tumor necrosis factor-alpha-induced apoptosis. Biochem Biophys Res Commun 2000; 272: 872–6
Speidel MT, Booyse FM, Abrams A, et al. Lipolyzed hypertriglyceridemic serum and triglyceride-rich lipoprotein cause lipid accumulation in and are cytotoxic to cultured human endothelial cells: high density lipoproteins inhibit this cytotoxicity. Thromb Res 1990; 58: 251–64
Rosenfeld SI, Packman CH, Leddy JP. Inhibition of the lytic action of cell-bound terminal complement components by human high density lipoproteins and apoproteins. J Clin Invest 1983; 71: 795–808
Packman CH, Rosenfeld SI, Leddy JP. High-density lipoprotein and its apolipoproteins inhibit cytolytic activity of complement: studies on the nature of inhibitory moiety. Biochim Biophys Acta 1985; 812: 107–15
Hamilton KK, Sims PJ. The terminal complement proteins C5b-9 augment binding of high density lipoprotein and its apolipoproteins A-I and A-II to human endothelial cells. J Clin Invest 1991; 88: 1833–40
Hamilton KK, Zhao J, Sims PJ. Interaction between apolipoproteins A-I and A-II and the membrane attack complex of complement: affinity of the apoproteins for polymeric C 9. J Biol Chem 1993; 268: 3632–8
Vakeva A, Jauhiainen M, Ehnholm C, et al. High-density lipoproteins can act as carriers of glycophosphoinositol lipid-anchored CD59 in human plasma. Immunology 1994; 82: 28–33
Schmiedt W, Kinscherf R, Deigner HP, et al. Complement C6 deficiency protects against diet-induced atherosclerosis in rabbits. Arterioscler Thromb Vasc Biol 1998; 18: 1790–5
Naqvi TZ, Shah PK, Ivey PA, et al. Evidence that high-density lipoprotein cholesterol is an independent predictor of acute platelet-dependent thrombus formation. Am J Cardiol 1999; 84: 1011–7
Aviram M, Brook JG. Platelet interaction with high and low density lipoproteins. Atherosclerosis 1983; 46: 259–68
Aviram M, Brook JG. Characterization of the effect of plasma lipoproteins on platelet function in vitro. Haemostasis 1983; 13: 344–50
Hassall DG, Owen JS, Bruckdorfer KR. The aggregation of isolated human platelets in the presence of lipoproteins and prostacyclin. Biochem J 1983; 216: 43–9
Nofer JR, Walter M, Kehrel B, et al. HDL3-mediated inhibition of thrombin-induced platelet aggregation and fibrinogen binding occurs via decreased production of phosphoinositide-derived second messengers 1,2-diacylglycerol and inositol 1,4,5-tris-phosphate. Arterioscler Thromb Vasc Biol 1998; 18: 861–9
Desai K, Bruckdorfer KR, Hutton RA, et al. Binding of apoE-rich high density lipoprotein particles by saturable sites on human blood platelets inhibits agonist-induced platelet aggregation. J Lipid Res 1989; 30: 831–40
Riddell DR, Graham A, Owen JS. Apolipoprotein E inhibits platelet aggregation through the L-arginine: nitric oxide pathway: implications for vascular disease. J Biol Chem 1997; 272: 89–95
Chen LY, Mehta JL. Inhibitory effect of high-density lipoprotein on platelet function is mediated by increase in nitric oxide synthase activity in platelets. Life Sci 1994; 55: 1815–21
Riddell DR, Vinogradov DV, Stannard AK, et al. Identification and characterization of LRP8 (apoER2) in human blood platelets. J Lipid Res 1999; 40: 1925–30
Nofer JR, Tepel M, Kehrel B, et al. High density lipoproteins enhance the Na+/H+ antiport in human platelets. Thromb Haemost 1996; 75: 635–41
Kaneko T, Wada H, Wakita Y, et al. Enhanced tissue factor activity and plasminogen activator inhibitor-1 antigen in human umbilical vein endothelial cells incubated with lipoproteins. Blood Coagul Fibrinolysis 1994; 5: 385–92
Rosenson RS, Lowe GD. Effects of lipids and lipoproteins on thrombosis and rheology. Atherosclerosis 1998; 140: 271–80
Lesnik P, Vonica A, Guerin M, et al. Anticoagulant activity of tissue factor pathway inhibitor in human plasma is preferentially associated with dense subspecies of LDL and HDL and with Lp(a). Arterioscler. Thromb 1993; 13: 1066–75
Epand RM, Stafford A, Leon B, et al. HDL and apolipoprotein A-I protect erythrocytes against the generation of procoagulant activity. Arterioscler Thromb 1994; 14: 1775–83
Griffin JH, Kojima K, Banka CL, et al. High-density lipoprotein enhancement of anticoagulant activities of plasma protein S and activated protein C. J Clin Invest 1999; 103: 219–27
Ko Y, Haring R, Stiebler H, et al. High-density lipoprotein reduces epidermal growth factor-induced DNA synthesis in vascular smooth muscle cells. Atherosclerosis 1993; 99: 253–9
Ishigami M, Swertfeger DK, Granholm NA, et al. Apolipoprotein E inhibits platelet-derived growth factor-induced vascular smooth muscle cell migration and proliferation by suppressing signal transduction and preventing cell entry to G1 phase. J Biol Chem 1998; 273: 20156–61
Rye KA, Clay MA, Barter PJ. Remodelling of high density lipoproteins by plasma factors. Atherosclerosis 1999; 145: 227–38
Castle CK, Pape ME, Marotti KR, et al. Secretion of pre-beta-migrating apoA-I by cynomolgus monkey hepatocytes in culture. J Lipid Res 1991; 32: 439–47
Danielsen EM, Hansen GH, Poulsen MD. Apical secretion of apolipoproteins from enterocytes. J Cell Biol 1993; 120: 1347–56
Musliner TA, Long MD, Forte TM, et al. Dissociation of high density lipoprotein precursors from apolipoprotein B-containing lipoproteins in the presence of unesterified fatty acids and a source of apolipoprotein A-I. J Lipid Res 1991; 32: 917–33
Strauss JG, Frank S, Kratky D, et al. Adenovirus-mediated rescue of lipoprotein lipase-deficient mice: lipolysis of triglyceride-rich lipoproteins is essential for high density lipoprotein maturation in mice. J Biol Chem 2001; 276: 36083–90
Liang HQ, Rye KA, Barter PJ. Dissociation of lipid-free apolipoprotein A-I from high density lipoproteins. J Lipid Res 1994; 35: 1187–99
Francone OL, Royer L, Haghpassand M. Increased prebeta-HDL levels, cholesterol efflux, and LCAT-mediated esterification in mice expressing the human cholesteryl ester transfer protein (CETP) and human apolipoprotein A-I (apoA-I) transgenes. J Lipid Res 1996; 37: 1268–77
von Eckardstein A, Jauhiainen M, Huang Y, et al. Phospholipid transfer protein mediated conversion of high density lipoproteins generates pre beta 1-HDL. Biochim Biophys Acta 1996; 1301: 255–62
Jiang X, Francone OL, Bruce C, et al. Increased prebeta-high density lipoprotein, apolipoprotein AI, and phospholipid in mice expressing the human phospholipid transfer protein and human apolipoprotein AI transgenes. J Clin Invest 1996; 98: 2373–80
Silver DL, Wang N, Xiao X, et al. High density lipoprotein (HDL) particle uptake mediated by scavenger receptor class B type 1 results in selective sorting of HDL cholesterol from protein and polarized cholesterol secretion. J Biol Chem 2001; 276: 25287–93
Barrans A, Collet X, Barbaras R, et al. Hepatic lipase induces the formation of pre-beta 1 high density lipoprotein (HDL) from triacylglycerol-rich HDL2: a study comparing liver perfusion to in vitro incubation with lipases. J Biol Chem 1994; 269: 11572–7
Oram JF, Yokoyama S. Apolipoprotein-mediated removal of cellular cholesterol and phospholipids. J Lipid Res 1996; 37: 2473–91
Klucken J, Buchler C, Orso E, et al. ABCG1 (ABC8), the human homolog of the Drosophila white gene, is a regulator of macrophage cholesterol and phospholipid transport. Proc Natl Acad Sci U S A 2000; 97: 817–22
Miida T, Kawano M, Fielding CJ, et al. Regulation of the concentration of pre beta high-density lipoprotein in normal plasma by cell membranes and lecithin-cholesterol acyltransferase activity. Biochemistry 1992; 31: 11112–7
Liang HQ, Rye KA, Barter PJ. Remodelling of reconstituted high density lipoproteins by lecithin: cholesterol acyltransferase. J Lipid Res 1996; 37: 1962–70
von Eckardstein A, Huang Y, Kastelein JJ, et al. Lipid-free apolipoprotein (apo) A-I is converted into alpha-migrating high density lipoproteins by lipoprotein-depleted plasma of normolipidemic donors and apo A-I-deficient patients but not of Tangier disease patients. Atherosclerosis 1998; 138: 25–34
Dieplinger H, Zechner R, Kostner GM. The in vitro formation of HDL2 during the action of LCAT: the role of triglyceride-rich lipoproteins. J Lipid Res 1985; 26: 273–82
Lusa S, Jauhiainen M, Metso J, et al. The mechanism of human plasma phospholipid transfer protein-induced enlargement of high-density lipoprotein particles: evidence for particle fusion. Biochem J 1996; 313 (Pt 1): 275–82
Jiang XC, Bruce C, Mar J, et al. Targeted mutation of plasma phospholipid transfer protein gene markedly reduces high-density lipoprotein levels. J Clin Invest 1999; 103: 907–14
Krieger M. Charting the fate of the ‘good cholesterol’: identification and characterization of the high-density lipoprotein receptor SR-BI. Annu Rev Biochem 1999; 68: 523–58
Trigatti B, Rigotti A, Krieger M. The role of the high-density lipoprotein receptor SR-BI in cholesterol metabolism. Curr Opin Lipidol 2000; 11: 123–31
Tall AR, Jiang X, Luo Y, et al. 1999 George Lyman Duff memorial lecture: lipid transfer proteins, HDL metabolism, and atherogenesis. Arterioscler Thromb Vasc Biol 2000; 20: 1185–8
Cohen JC, Vega GL, Grundy SM. Hepatic lipase: new insights from genetic and metabolic studies. Curr Opin Lipidol 1999; 10: 259–67
Thuren T. Hepatic lipase and HDL metabolism. Curr Opin Lipidol 2000; 11: 277–83
Rader DJ, Jaye M. Endothelial lipase: a new member of the triglyceride lipase gene family. Curr Opin Lipidol 2000; 11: 141–7
Martinez LO, Jacquet S, Esteve JP, et al. Ectopic beta-chain of ATP synthase is an apolipoprotein A-I receptor in hepatic HDL endocytosis. Nature 2003; 421(6918): 75–9
Christensen EI, Birn H. Megalin and cubilin: multifunctional endocytic receptors. Nat Rev Mol Cell Biol 2002; 3: 256–66
Kozyraki R. Cubilin, a multifunctional epithelial receptor: an overview. J Mol Med 2001; 79: 161–7
Bolibar I, von Eckardstein A, Assmann G, et al. Short-term prognostic value of lipid measurements in patients with angina pectoris. The ECAT Angina Pectoris Study Group: European Concerted Action on Thrombosis and Disabilities. Thromb Haemost 2000; 84: 955–60
Miller M, Seidler A, Moalemi A, et al. Normal triglyceride levels and coronary artery disease events: the Baltimore Coronary Observational Long-Term Study. J Am Coll Cardiol 1998; 31: 1252–7
Cullen P, Schulte H, Assmann G. The Munster Heart Study (PROCAM): total mortality in middle-aged men is increased at low total and LDL cholesterol concentrations in smokers but not in nonsmokers. Circulation 1997; 96: 2128–36
Jeppesen J, Hein HO, Suadicani P, et al. Triglyceride concentration and ischemic heart disease: an eight-year follow-up in the Copenhagen Male Study. Circulation 1998; 97: 1029–36
von Eckardstein A, Schulte H, Assmann G. Increased risk of myocardial infarction in men with both hypertriglyceridemia and elevated HDL cholesterol [letter]. Circulation 1999; 99: 1925
De Backer G, De Bacquer D, Kornitzer M. Epidemiological aspects of high density lipoprotein cholesterol. Atherosclerosis 1998; 137 Suppl.: S1–6
National Cholesterol Education Program (NCEP). Detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel II). Circulation 1994; 89: 1329–445
Harper CR, Jacobson TA. New perspectives on the management of low levels of high-density lipoprotein cholesterol. Arch Intern Med 1999; 159: 1049–57
von Eckardstein A, Assmann G. Prevention of coronary heart disease by raising high-density lipoprotein cholesterol? Curr Opin Lipidol 2000; 11: 627–37
Stern MP, Williams K, Haffner SM. Identification of persons at high risk for type 2 diabetes mellitus: do we need the oral glucose tolerance test? Ann Intern Med 2002; 136: 575–81
von Eckardstein A, Schulte H, Assmann G. Risk for diabetes mellitus in middle-aged Caucasian male participants of the PROCAM study: implications for the definition of impaired fasting glucose by the American Diabetes Association: Prospective Cardiovascular Munster. J Clin Endocrinol. Metab 2000; 85: 3101–8
Erren M, Reinecke H, Junker R, et al. Systemic inflammatory parameters in patients with atherosclerosis of the coronary and peripheral arteries. Arterioscler Thromb Vasc Biol 1999; 19: 2355–63
Stampfer MJ, Sacks FM, Salvini S, et al. A prospective study of cholesterol, apolipoproteins, and the risk of myocardial infarction. N Engl J Med 1991; 325: 373–81
Sharrett AR, Ballantyne CM, Coady SA, et al. Coronary heart disease prediction from lipoprotein cholesterol levels, triglycerides, lipoprotein(a), apolipoproteins A-I and B, and HDL density subfractions. The Atherosclerosis Risk in Communities (ARIC) Study. Circulation 2001; 104: 1108–13
Luc G, Bard JM, Ferneres J, et al. Value of HDL cholesterol, apolipoprotein A-I, lipoprotein A-I, and lipoprotein A-I/A-II in prediction of coronary heart disease: the PRIME Study. Prospective Epidemiological Study of Myocardial Infarction. Arterioscler Thromb Vasc Biol 2002; 22: 1155–61
Genest Jr J. Genetics and prevention: a new look at high-density lipoprotein cholesterol. Cardiol Rev 2002; 10: 61–71
Sirtori CR, Calabresi L, Franceschini G, et al. Cardiovascular status of carriers of the apolipoprotein A-I(Milano) mutant: the Limone sul Garda study. Circulation 2001; 103: 1949–54
Clee SM, Kastelein JJ, van Dam M, et al. Age and residual cholesterol efflux affect HDL cholesterol levels and coronary artery disease in ABCA1 heterozygotes. J Clin Invest 2000; 106: 1263–70
van Dam MJ, de Groot E, Clee SM, et al. Association between increased arterial-wall thickness and impairment in ABCA1-driven cholesterol efflux: an observational study. Lancet 2002; 359: 37–42
Zwarts KY, Clee SM, Zwinderman AH, et al. ABCA1 regulatory variants influence coronary artery disease independent of effects on plasma lipid levels. Clin Genet 2002; 61: 115–25
Lutucuta S, Ballantyne CM, Elghannam H, et al. Novel polymorphisms in promoter region of atp binding cassette transporter gene and plasma lipids, severity, progression, and regression of coronary atherosclerosis and response to therapy. Circ Res 2001; 88: 969–73
Clee SM, Zwinderman AH, Engert JC, et al. Common genetic variation in ABCA1 is associated with altered lipoprotein levels and a modified risk for coronary artery disease. Circulation 2001; 103: 1198–205
Hirano K, Yamashita S, Kuga Y, et al. Atherosclerotic disease in marked hyperalphalipoproteinemia: combined reduction of cholesteryl ester transfer protein and hepatic triglyceride lipase. Arterioscler Thromb Vasc Biol 1995; 15: 1849–56
Bruce C, Sharp DS, Tall AR. Relationship of HDL and coronary heart disease to a common amino acid polymorphism in the cholesteryl ester transfer protein in men with and without hypertriglyceridemia. J Lipid Res 1998; 39: 1071–8
Agerholm-Larsen B, Nordestgaard BG, Steffensen R, et al. Elevated HDL cholesterol is a risk factor for ischemic heart disease in white women when caused by a common mutation in the cholesteryl ester transfer protein gene. Circulation 2000; 101: 1907–12
Brousseau ME, O’Connor Jr JJ, Ordovas JM, et al. Cholesteryl ester transfer protein TaqI B2B2 genotype is associated with higher HDL cholesterol levels and lower risk of coronary heart disease end points in men with HDL deficiency: Veterans Affairs HDL Cholesterol Intervention Trial. Arterioscler Thromb Vasc Biol 2002; 22: 1148–54
Liu S, Schmitz C, Stampfer MJ, et al. A prospective study of TaqIB polymorphism in the gene coding for cholesteryl ester transfer protein and risk of myocardial infarction in middle-aged men. Atherosclerosis 2002; 161: 469–74
Li H, Reddick RL, Maeda N. Lack of apoA-I is not associated with increased susceptibility to atherosclerosis in mice. Arterioscler Thromb 1993; 13: 1814–21
Voyiaziakis E, Goldberg IJ, Plump AS, et al. ApoA-I deficiency causes both hypertriglyceridemia and increased atherosclerosis in human apoB transgenic mice. J Lipid Res 1998; 39: 313–21
Rubin EM, Krauss RM, Spangler EA, et al. Inhibition of early atherogenesis in transgenic mice by human apolipoprotein AI. Nature 1991; 353: 265–7
Duverger N, Kruth H, Emmanuel F, et al. Inhibition of atherosclerosis development in cholesterol-fed human apolipoprotein A-I-transgenic rabbits. Circulation 1996; 94: 713–7
Plump AS, Scott CJ, Breslow JL. Human apolipoprotein A-I gene expression increases high density lipoprotein and suppresses atherosclerosis in the apolipoprotein E-deficient mouse. Proc Natl Acad Sci U S A 1994; 91: 9607–11
Dansky HM, Charlten SA, Barlow CB, et al. Apo A-I inhibits foam cell formation in Apo E-deficient mice after monocyte adherence to endothelium. J Clin Invest 1999; 104: 31–9
Benoit P, Emmanuel F, Caillaud JM, et al. Somatic gene transfer of human ApoA-I inhibits atherosclerosis progression in mouse models. Circulation 1999; 99: 105–10
Tangirala RK, Tsukamoto K, Chun SH, et al. Regression of atherosclerosis induced by liver-directed gene transfer of apolipoprotein A-I in mice. Circulation 1999; 100: 1816–22
Atger V, de la Liera MM, Bamberger M, et al. Cholesterol efflux potential of sera from mice expressing human cholesteryl ester transfer protein and/or human apolipoprotein AI. J Clin Invest 1995; 96: 2613–22
Duverger N, Tremp G, Caillaud JM, et al. Protection against atherogenesis in mice mediated by human apolipoprotein A-IV. Science 1996; 273: 966–8
Cohen RD, Castellani LW, Qiao JH, et al. Reduced aortic lesions and elevated high density lipoprotein levels in transgenic mice overexpressing mouse apolipoprotein A-IV. J Clin Invest 1997; 99: 1906–16
McNeish J, Aiello RJ, Guyot D, et al. High density lipoprotein deficiency and foam cell accumulation in mice with targeted disruption of ATP-binding cassette transporter-1. Proc Natl Acad Sci U S A 2000; 97: 4245–50
Groen AK, Bloks VW, Bandsma RH, et al. Hepatobiliary cholesterol transport is not impaired in Abca1-null mice lacking HDL. J Clin Invest 2001; 108: 843–50
van Eck M, Bos IS, Kaminski WE, et al. Leukocyte ABCA1 controls susceptibility to atherosclerosis and macrophage recruitment into tissues. Proc Natl Acad Sci U S A 2002; 99: 6298–303
Aiello RJ, Brees D, Bourassa PA, et al. Increased atherosclerosis in hyperlipidemic mice with inactivation of ABCA1 in macrophages. Arterioscler Thromb Vasc Biol 2002; 22: 630–7
Singaraja RR, Bocher V, James ER, et al. Human ABCA1 BAC transgenic mice show increased high density lipoprotein cholesterol and ApoAI-dependent efflux stimulated by an internal promoter containing liver X receptor response elements in intron 1. J Biol Chem 2001; 276: 33969–79
Cavelier LB, Qiu Y, Bielicki JK, et al. Regulation and activity of the human ABCA1 gene in transgenic mice. J Biol Chem 2001; 276: 18046–51
Singaraja RR, Fievet C, Castro G, et al. Increased ABCA1 activity protects against atherosclerosis. J Clin Invest 2002; 110: 35–42
Joyce CW, Amar MJ, Lambert G, et al. The ATP binding cassette transporter A1 (ABCA1) modulates the development of aortic atherosclerosis in C57BL/6 and apoE-knockout mice. Proc Natl Acad Sci U S A 2002; 99: 407–12
Berard AM, Foger B, Remaley A, et al. High plasma HDL concentrations associated with enhanced atherosclerosis in transgenic mice overexpressing lecithin-cholesteryl acyltransferase. Nat Med 1997; 3: 744–9
Hoeg JM, Santamarina-Fojo S, Berard AM, et al. Over-expression of lecithin: cholesterol acyltransferase in transgenic rabbits prevents diet-induced atherosclerosis. Proc Natl Acad Sci U S A 1996; 93: 11448–53
Foger B, Chase M, Amar MJ, et al. Cholesteryl ester transfer protein corrects dysfunctional high density lipoproteins and reduces aortic atherosclerosis in lecithin cholesterol acyltransferase transgenic mice. J Biol Chem 1999; 274: 36912–20
Schultz JR, Verstuyft JG, Gong EL, et al. Protein composition determines the anti-atherogenic properties of HDL in transgenic mice. Nature 1993; 365: 762–4
Warden CH, Hedrick CC, Qiao JH, et al. Atherosclerosis in transgenic mice overexpressing apolipoprotein A-II. Science 1993; 261: 469–72
Escola-Gil JC, Marzal-Casacuberta A, Julve-Gil J, et al. Human apolipoprotein A-II is a pro-atherogenic molecule when it is expressed in transgenic mice at a level similar to that in humans: evidence of a potentially relevant species-specific interaction with diet. J Lipid Res 1998; 39: 457–62
Tailleux A, Bouly M, Luc G, et al. Decreased susceptibility to diet-induced atherosclerosis in human apolipoprotein A-II transgenic mice. Arterioscler Thromb Vasc Biol 2000; 20: 2453–8
Marotti KR, Castle CK, Boyle TP, et al. Severe atherosclerosis in transgenic mice expressing simian cholesteryl ester transfer protein. Nature 1993; 364: 73–5
Hayek T, Masucci-Magoulas L, Jiang X, et al. Decreased early atherosclerotic lesions in hypertriglyceridemic mice expressing cholesteryl ester transfer protein transgene. J Clin Invest 1995; 96: 2071–4
Plump AS, Masucci-Magoulas L, Bruce C, et al. Increased atherosclerosis in ApoE and LDL receptor gene knock-out mice as a result of human cholesteryl ester transfer protein transgene expression. Arterioscler Thromb Vasc Biol 1999; 19: 1105–10
Mezdour H, Jones R, Dengremont C, et al. Hepatic lipase deficiency increases plasma cholesterol but reduces susceptibility to atherosclerosis in apolipoprotein E-deficient mice. J Biol Chem 1997; 272: 13570–5
Busch SJ, Barnhart RL, Martin GA, et al. Human hepatic triglyceride lipase expression reduces high density lipoprotein and aortic cholesterol in cholesterol-fed transgenic mice. J Biol Chem 1994; 269: 16376–82
Ji Y, Wang N, Ramakrishnan R, et al. Hepatic scavenger receptor BI promotes rapid clearance of high density lipoprotein free cholesterol and its transport into bile. J Biol Chem 1999; 274: 33398–402
Mardones P, Quinones V, Amigo L, et al. Hepatic cholesterol and bile acid metabolism and intestinal cholesterol absorption in scavenger receptor class B type I-deficient mice. J Lipid Res 2001; 42: 170–80
Landschulz KT, Pathak RK, Rigotti A, et al. Regulation of scavenger receptor, class B, type I, a high density lipoprotein receptor, in liver and steroidogenic tissues of the rat. J Clin Invest 1996; 98: 984–95
Graf GA, Roswell KL, Smart EJ. 17beta-Estradiol promotes the up-regulation of SR-BII in HepG2 cells and in rat livers. J Lipid Res 2001; 42: 1444–9
Langer C, Gansz B, Goepfert C, et al. Testosterone up-regulates scavenger receptor B1 and stimulates cholesterol efflux from macrophages. Biochem Biophys Res Commun 2002; 296: 1051–7
Influence of pravastatin and plasma lipids on clinical events in the West of Scotland Coronary Prevention Study (WOSCOPS). Circulation 1998; 97: 1440-5
Sacks FM, Tonkin AM, Shepherd J, et al. Effect of pravastatin on coronary disease events in subgroups defined by coronary risk factors: the Prospective Pravastatin Pooling Project. Circulation 2000; 102: 1893–900
Ballantyne CM, Olsson AG, Cook TJ, et al. Influence of low high-density lipoprotein cholesterol and elevated triglyceride on coronary heart disease events and response to simvastatin therapy in 4S. Circulation 2001; 104: 3046–51
Robins SJ, Collins D, Wittes JT, et al. Relation of gemfibrozil treatment and lipid levels with major coronary events. VAHIT: a randomized controlled trial. JAMA 2001; 285: 1585–91
Secondary prevention by raising HDL cholesterol and reducing triglycerides in patients with coronary artery disease: the Bezafibrate Infarction Prevention (BIP) study. Circulation 2000; 102: 21-7
Hulley S, Grady D, Bush T, et al. Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women. Heart and Estrogen/progestin Replacement Study (HERS) Research Group. JAMA 1998; 280: 605–13
Ruotolo G, Ericsson CG, Tettamanti C, et al. Treatment effects on serum lipoprotein lipids, apolipoproteins and low density lipoprotein particle size and relationships of lipoprotein variables to progression of coronary artery disease in the Bezafibrate Coronary Atherosclerosis Intervention Trial (BECAIT). J Am Coll Cardiol 1998; 32: 1648–56
Ballantyne CM, Herd JA, Ferlic LL, et al. Influence of low HDL on progression of coronary artery disease and response to fluvastatin therapy. Circulation 1999; 99: 736–43
Frick MH, Syvanne M, Nieminen MS, et al. Prevention of the angiographic progression of coronary and vein-graft atherosclerosis by gemfibrozil after coronary bypass surgery in men with low levels of HDL cholesterol. Lopid Coronary Angiography Trial (LOCAT) Study Group. Circulation 1997; 96: 2137–43
Grady D, Herrington D, Bittner V, et al. Cardiovascular disease outcomes during 6.8 years of hormone therapy. Heart and Estrogen/progestin Replacement Study follow-up (HERS II). JAMA 2002; 288: 49–57
Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 2002; 288: 321-33
Gotto Jr AM. Management of dyslipidemia. Am J Med 2002; 112 Suppl. 8A: 10S–8S
Maron DJ, Fazio S, Linton MF. Current perspectives on statins. Circulation 2000; 101: 207–13
Jones P, Kafonek S, Laurora I, et al. Comparative dose efficacy study of atorvastatin versus simvastatin, pravastatin, lovastatin, and fluvastatin in patients with hypercholesterolemia (the CURVES study). Am J Cardiol 1998; 81: 582–7
Davignon J, Hanefeld M, Nakaya N, et al. Clinical efficacy and safety of cerivastatin: summary of pivotal phase IIb/III studies. Am J Cardiol 1998; 82: 32J-9J
Wierzbicki AS, Mikhailidis DP. Dose-response effects of atorvastatin and simvastatin on high-density lipoprotein cholesterol in hypercholesterolaemic patients: a review of five comparative studies. Int J Cardiol 2002; 84: 53–7
Watts GF, Barrett PH, Ji J, et al. Differential regulation of lipoprotein kinetics by atorvastatin and fenofibrate in subjects with the metabolic syndrome. Diabetes 2003; 52(3): 803–11
Martin G, Duez H, Blanquart C, et al. Statin-induced inhibition of the Rho-signaling pathway activates PPARalpha and induces HDL apoA-I. J Clin Invest 2001; 107: 1423–32
Bonn V, Cheung RC, Chen B, et al. Simvastatin, an HMG-CoA reductase inhibitor, induces the synthesis and secretion of apolipoprotein AI in HepG2 cells and primary hamster hepatocytes. Atherosclerosis 2002; 163: 59–68
Laufs U, Marra D, Node K, et al. 3-Hydroxy-3-methylglutaryl-CoA reductase inhibitors attenuate vascular smooth muscle proliferation by preventing rho GTPase-induced down-regulation of p27(Kipl). J Biol Chem 1999; 274: 21926–31
Guerin M, Lassel TS, Le Goff W, et al. Action of atorvastatin in combined hyperlipidemia: preferential reduction of cholesteryl ester transfer from HDL to VLDL1 particles. Arterioscler Thromb Vasc Biol 2000; 20: 189–97
Guerin M, Egger P, Soudant C, et al. Dose-dependent action of atorvastatin in type IIB hyperlipidemia. preferential and progressive reduction of atherogenic apoB-containing lipoprotein subclasses (VLDL-2, IDL, small dense LDL) and stimulation of cellular cholesterol efflux. Atherosclerosis 2002; 163: 287–96
Fruchart JC, Duriez P, Staels B. Peroxisome proliferator-activated receptor-alpha activators regulate genes governing lipoprotein metabolism, vascular inflammation and atherosclerosis. Curr Opin Lipidol 1999; 10: 245–57
Chinetti G, Gbaguidi FG, Griglio S, et al. CLA-1/SR-BI is expressed in atherosclerotic lesion macrophages and regulated by activators of peroxisome proliferator-activated receptors. Circulation 2000; 101: 2411–7
Fruchart JC. Peroxisome proliferator-activated receptor-alpha activation and high-density lipoprotein metabolism. Am J Cardiol 2001; 88: 24N-9N
Manninen V, Elo MO, Frick MH, et al. Lipid alterations and decline in the incidence of coronary heart disease in the Helsinki Heart Study. JAMA 1988; 260: 641–51
Despres JP. Increasing high-density lipoprotein cholesterol: an update on fenofibrate. Am J Cardiol 2001; 88: 30N-6N
Vega GL, Grundy SM. Lipoprotein responses to treatment with lovastatin, gemfibrozil, and nicotinic acid in normolipidemic patients with hypoalphalipoproteinemia. Arch Intern Med 1994; 154: 73–82
Tavintharan S, Kashyap ML. The benefits of niacin in atherosclerosis. Curr Atheroscler Rep 2001; 3: 74–82
Shepherd J, Packard CJ, Patsch JR, et al. Effects of nicotinic acid therapy on plasma high density lipoprotein subfraction distribution and composition and on apolipoprotein A metabolism. J Clin Invest 1979; 63: 858–67
Jin FY, Kamanna VS, Kashyap ML. Niacin decreases removal of high-density lipoprotein apolipoprotein A-I but not cholesterol ester by Hep G2 cells: implication for reverse cholesterol transport. Arterioscler Thromb Vasc Biol 1997; 17: 2020–8
Kamanna VS, Kashyap ML. Mechanism of action of niacin on lipoprotein metabolism. Curr Atheroscler Rep 2000; 2: 36–46
Elam MB, Hunninghake DB, Davis KB, et al. Effect of niacin on lipid and lipoprotein levels and glycemic control in patients with diabetes and peripheral arterial disease: the ADMIT study: a randomized trial. Arterial Disease Multiple Intervention Trial. JAMA 2000; 284: 1263–70
Sakai T, Kamanna VS, Kashyap ML. Niacin, but not gemfibrozil, selectively increases LP-AI, a cardioprotective subfraction of HDL, in patients with low HDL cholesterol. Arterioscler Thromb Vasc Biol 2001; 21: 1783–9
Canner PL, Berge KG, Wenger NK, etal. Fifteen year mortality in Coronary Drug Project patients: long-term benefit with niacin. J Am Coll Cardiol 1986; 8: 1245–55
Brown BG, Zhao XQ, Chait A, et al. Simvastatin and niacin, antioxidant vitamins, or the combination for the prevention of coronary disease. N Engl J Med 2001; 345: 1583–92
Kashyap ML, McGovern ME, Berra K, et al. Long-term safety and efficacy of a once-daily niacin/lovastatin formulation for patients with dyslipidemia. Am J Cardiol 2002; 89: 672–8
Wink J, Giacoppe G, King J. Effect of very-low-dose niacin on high-density lipoprotein in patients undergoing long-term statin therapy. Am Heart J 2002; 143: 514–8
Sposito AC, Caramelli B, Serrano Jr CV, et al. Effect of niacin and etofibrate association on subjects with coronary artery disease and serum high-density lipoprotein cholesterol <35 mg/dl. Am J Cardiol 1999; 83: 98–100
Zema MJ. Gemfibrozil, nicotinic acid and combination therapy in patients with isolated hypoalphalipoproteinemia: a randomized, open-label, crossover study. J Am Coll Cardiol 2000; 35: 640–6
Evans M, Rees A. The myotoxicity of statins. Curr Opin Lipidol 2002; 13: 415–20
Shek A, Ferrill MJ. Statin-fibrate combination therapy. Ann Pharmacother 2001; 35: 908–17
Athyros VG, Papageorgiou AA, Athyrou VV, et al. Atorvastatin versus four statin-fibrate combinations in patients with familial combined hyperlipidaemia. J Cardiovasc Risk 2002; 9: 33–9
Athyros VG, Papageorgiou AA, Hatzikonstandinou HA, et al. Safety and efficacy of long-term statin-fibrate combinations in patients with refractory familial combined hyperlipidemia. Am J Cardiol 1997; 80: 608–13
Kiortsis DN, Millionis H, Bairaktari E, et al. Efficacy of combination of atorvastatin and micronised fenofibrate in the treatment of severe mixed hyperlipidemia. Eur J Clin Pharmacol 2000; 56: 631–5
Gavish D, Leibovitz E, Shapira I, et al. Bezafibrate and simvastatin combination therapy for diabetic dyslipidaemia: efficacy and safety. J Intern Med 2000; 247: 563–9
Lopez D, Sanchez MD, Shea-Eaton W, et al. Estrogen activates the high-density lipoprotein receptor gene via binding to estrogen response elements and interaction with sterol regulatory element binding protein-1A. Endocrinology 2002; 143: 2155–68
Jones DR, Schmidt RJ, Pickard RT, et al. Estrogen receptor-mediated repression of human hepatic lipase gene transcription. J Lipid Res 2002; 43: 383–91
Brinton EA. Oral estrogen replacement therapy in postmenopausal women selectively raises levels and production rates of lipoprotein A-I and lowers hepatic lipase activity without lowering the fractional catabolic rate. Arterioscler Thromb Vasc Biol 1996; 16: 431–40
Fluiter K, van der Westhuijzen DR, van Berkel TJ. In vivo regulation of scavenger receptor BI and the selective uptake of high density lipoprotein cholesteryl esters in rat liver parenchymal and Kupffer cells. J Biol Chem 1998; 273: 8434–8
Binder EF, Williams DB, Schechtman KB, et al. Effects of hormone replacement therapy on serum lipids in elderly women: a randomized, placebo-controlled trial. Ann Intern Med 2001; 134: 754–60
Komesaroff PA, Fullerton M, Esler MD, et al. Low-dose estrogen supplementation improves vascular function in hypogonadal men. Hypertension 2001; 38: 1011–6
Knopp RH, Broyles FE, Cheung M, et al. Comparison of the lipoprotein, carbohydrate, and hemostatic effects of phasic oral contraceptives containing desogestrel or levonorgestrel. Contraception 2001; 63: 1–11
Wiegratz I, Lee JH, Kutschera E, et al. Effect of dienogest-containing oral contraceptives on lipid metabolism. Contraception 2002; 65: 223–9
Fletcher SW, Colditz GA. Failure of estrogen plus progestin therapy for prevention. JAMA 2002; 288: 366–8
Johnston Jr CC, Bjarnason NH, Cohen FJ, et al. Long-term effects of raloxifene on bone mineral density, bone turnover, and serum lipid levels in early postmenopausal women: three-year data from 2 double-blind, randomized, placebo-controlled trials. Arch Intern Med 2000; 160: 3444–50
Cummings SR, Eckert S, Krueger KA, et al. The effect of raloxifene on risk of breast cancer in postmenopausal women: results from the MORE randomized trial. Multiple Outcomes of Raloxifene Evaluation. JAMA 1999; 281: 2189–97
von Eckardstein A, Crook D, Elbers J, et al. Tibolone lowers HDL cholesterol by increasing hepatic lipase activity but does not impair cholesterol efflux. Clin Endocrinol 2003; 58: 49–58
von Eckardstein A, Schmiddem K, Hovels A, et al. Lowering of HDL cholesterol in post-menopausal women by tibolone is not associated with changes in cholesterol efflux capacity or paraoxonase activity. Atherosclerosis 2001; 159: 433–9
Bhasin S, Woodhouse L, Casaburi R, et al. Testosterone dose-response relationships in healthy young men. Am J Physiol Endocrinol Metab 2001; 281: E1172–81
Wu FCW, von Eckardstein A. Androgens and coronary artery disease. Endocrine Rev 2003; 24: 183–217
Connor WE. n-3 fatty acids from fish and fish oil: panacea or nostrum? Am J Clin Nutr 2001; 74: 415–6
Harris WS. n-3 fatty acids and lipoproteins: comparison of results from human and animal studies. Lipids 1996; 31: 243–52
Farmer A, Montori V, Dinneen S, et al. Fish oil in people with type 2 diabetes mellitus. Available in The Cochrane Library [database on disk and CD ROM]. Updated quarterly. The Cochrane Collaboration; issue 1. Oxford: Update Software, 2001, CD003205
Marchioli R, Barzi F, Bomba E, et al. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)-Prevenzione. Circulation 2002; 105: 1897–903
Schoonjans K, Staels B, Auwerx J. Role of the peroxisome proliferator-activated receptor (PPAR) in mediating the effects of fibrates and fatty acids on gene expression. J Lipid Res 1996; 37: 907–25
Huggins KW, Colvin PL, Burleson ER, et al. Dietary n-3 polyunsaturated fat increases the fractional catabolic rate of medium-sized HDL particles in African green monkeys. J Lipid Res 2001; 42: 1457–66
Dallongeville J, Bauge E, Tailleux A, et al. Peroxisome proliferator-activated receptor alpha is not rate-limiting for the lipoprotein-lowering action of fish oil. J Biol Chem 2001; 276: 4634–9
Yoshikawa T, Shimano H, Yahagi N, et al. Polyunsaturated fatty acids suppress sterol regulatory element-binding protein 1c promoter activity by inhibition of liver X receptor (LXR) binding to LXR response elements. J Biol Chem 2002; 277: 1705–11
Laffitte BA, Tontonoz P. Orphan nuclear receptors find a home in the arterial wall. Curr Atheroscler Rep 2002; 4: 213–21
Edwards PA, Kast HR, Anisfeld AM. BAREing it all: the adoption of LXR and FXR and their roles in lipid homeostasis. J Lipid Res 2002; 43: 2–12
Ou J, Tu H, Shan B, et al. Unsaturated fatty acids inhibit transcription of the sterol regulatory element-binding protein-1c (SREBP-1c) gene by antagonizing ligand-dependent activation of the LXR. Proc Natl Acad Sci U S A 2001; 98: 6027–32
Okamoto H, Yonemori F, Wakitani K, et al. A cholesteryl ester transfer protein inhibitor attenuates atherosclerosis in rabbits. Nature 2000; 406: 203–7
Hirano K, Yamashita S, Matsuzawa Y. Pros and cons of inhibiting cholesteryl ester transfer protein. Curr Opin Lipidol 2000; 11: 589–96
de Grooth GJ, Kuivenhoven JA, Stalenhoef AF, et al. Efficacy and safety of a novel cholesteryl ester transfer protein inhibitor, JTT-705, in humans. a randomized phase II dose-response study. Circulation 2002; 105: 2159–65
Nanjee MN, Cooke CJ, Garvin R, et al. Intravenous apoA-I/ lecithin discs increase pre-beta-HDL concentration in tissue fluid and stimulate reverse cholesterol transport in humans. J Lipid Res 2001; 42: 1586–93
Eriksson M, Carlson LA, Miettinen TA, et al. Stimulation of fecal steroid excretion after infusion of recombinant proapolipoprotein A-I: potential reverse cholesterol transport in humans. Circulation 1999; 100: 594–8
Shah PK, Yano J, Reyes O, et al. High-dose recombinant apolipoprotein A-I(milano) mobilizes tissue cholesterol and rapidly reduces plaque lipid and macrophage content in apolipoprotein e-deficient mice: potential implications for acute plaque stabilization. Circulation 2001; 103: 3047–50
Shah PK, Nilsson J, Kaul S, et al. Effects of recombinant apolipoprotein A-I(Milano) on aortic atherosclerosis in apolipoprotein E-deficient mice. Circulation 1998; 97: 780–5
Navab M, Anantharamaiah GM, Hama S, et al. Oral administration of an Apo A-I mimetic Peptide synthesized from D-amino acids dramatically reduces atherosclerosis in mice independent of plasma cholesterol. Circulation 2002; 105: 290–2
Garber DW, Datta G, Chaddha M, et al. A new synthetic class A amphipathic peptide analogue protects mice from diet-induced atherosclerosis. J Lipid Res 2001; 42: 545–52
Van Lenten BJ, Wagner AC, Anantharamaiah GM, et al. Influenza infection promotes macrophage traffic into arteries of mice that is prevented by D-4F, an apolipoprotein A-I mimetic peptide. Circulation 2002; 106: 1127–32
Garber DW, Venkatachalapathi YV, Gupta KB, et al. Turnover of synthetic class A amphipathic peptide analogues of exchangeable apolipoproteins in rats: correlation with physical properties. Arterioscler. Thromb 1992; 12: 886–94
Davidson WS, Lund-Katz S, Johnson WJ, et al. The influence of apolipoprotein structure on the efflux of cellular free cholesterol to high density lipoprotein. J Biol Chem 1994; 269: 22975–82
Spuhler P, Anantharamaiah GM, Segrest JP, et al. Binding of apolipoprotein A-I model peptides to lipid bilayers: measurement of binding isotherms and peptide-lipid headgroup interactions. J Biol Chem 1994; 269: 23904–10
Anantharamaiah GM, Jones JL, Brouillette CG, et al. Studies of synthetic peptide analogs of the amphipathic helix: structure of complexes with dimyristoyl phosphatidylcholine. J Biol Chem 1985; 260: 10248–55
Owens BJ, Anantharamaiah GM, Kahlon JB, et al. Apolipoprotein A-I and its amphipathic helix peptide analogues inhibit human immunodeficiency virus-induced syncytium formation. J Clin Invest 1990; 86: 1142–50
Srinivas RV, Birkedal B, Owens RJ, et al. Antiviral effects of apolipoprotein A-I and its synthetic amphipathic peptide analogs. Virology 1990; 176: 48–57
Chawla A, Repa JJ, Evans RM, et al. Nuclear receptors and lipid physiology: opening the X-files. Science 2001; 294: 1866–70
Walczak R, Tontonoz P. PPARadigms and PPARadoxes: expanding roles for PPARgamma in the control of lipid metabolism. J Lipid Res 2002; 43: 177–86
Oliver Jr WR, Shenk JL, Snaith MR, et al. A selective peroxisome proliferator-activated receptor delta agonist promotes reverse cholesterol transport. Proc Natl Acad Sci U S A 2001; 98: 5306–11
Shih DQ, Bussen M, Sehayek E, et al. Hepatocyte nuclear factor-1alpha is an essential regulator of bile acid and plasma cholesterol metabolism. Nat Genet 2001; 27: 375–82
Porsch-Ozcurumez M, Langmann T, Heimerl S, et al. The zinc finger protein 202 (ZNF202) is a transcriptional repressor of ATP binding cassette transporter A1 (ABCA1) and ABCG1 gene expression and a modulator of cellular lipid efflux. J Biol Chem 2001; 276: 12427–33
Wagner S, Hess MA, Ormonde-Hanson P, et al. A broad role for the zinc finger protein ZNF202 in human lipid metabolism. J Biol Chem 2000; 275: 15685–90
Asztalos B, Horvath K, McNamara J, et al. Comparing the effects of five different statins on the HDL subpopulation profiles of coronary heart disease patients. Atherosclerosis 2002; 164: 361–9
Rothblat GH, Llera-Moya M, Favari E, et al. Cellular cholesterol flux studies: methodological considerations. Atherosclerosis 2002; 163: 1–8
Mikkola TS, Anthony MS, Clarkson TB, et al. Serum cholesterol efflux potential in postmenopausal monkeys treated with tibolone or conjugated estrogens. Metabolism 2002; 51: 523–30
Mackness MI, Mackness B, Durrington PN, et al. Paraoxonase and coronary heart disease. Curr Opin Lipidol 1998; 9: 319–24
Aviram M. Does paraoxonase play a role in susceptibility to cardiovascular disease? Mol Med Today 1999; 5: 381–6
Stein O, Dabach Y, Hollander G, et al. High levels of human apolipoprotein A-I and high density lipoproteins in transgenic mice do not enhance efflux of cholesterol from a depot of injected lipoproteins: relevance to regression of atherosclerosis? Atherosclerosis 1999; 144: 367–74
Marsh JB, Welty FK, Schaefer EJ. Stable isotope turnover of apolipoproteins of high-density lipoproteins in humans. Curr Opin Lipidol 2000; 11: 261–6
Acknowledgements
Dr Arnold von Eckardstein is supported by an International HDL Research Award sponsored by Pfizer Inc. and by a grant from the Swiss National Foundation. Dr Martin Hersberger is supported by the EMDO fund and by the Forschungskommission of the University of Zurich.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hersberger, M., Eckardstein, A.v. Low High-Density Lipoprotein Cholesterol. Drugs 63, 1907–1945 (2003). https://doi.org/10.2165/00003495-200363180-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200363180-00003