Abstract
Background
Certain foods, such as grapefruit juice, are known to substantially alter the bioavailability of some drugs. These effects may be mediated by interactions with enzyme systems, such as cytochrome P450, or with active transporter systems, such as P-glycoprotein and organic anion transporting polypeptides.
Objective
To assess the effect of consumption of grapefruit juice on the oral bioavailability of two nonsedating antihistamines, fexofenadine and desloratadine.
Design
Non-blinded, randomised, single-dose, four-way crossover study.
Participants
Twenty-four healthy adult volunteers.
Interventions
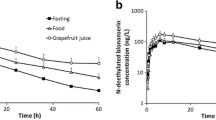
Single oral doses of desloratadine 5mg and fexofenadine 60mg taken without and with grapefruit juice (pretreatment with 240ml of double-strength juice three times daily for 2 days prior to administration of study drug, plus the same amount simultaneously with, and 2 hours after, the drug dose). Each treatment was separated by at least 10 days.
Main Outcome Measures
Log-transformed pharmacokinetic parameters [peak plasma concentration (Cmax) and area under the curve (AUC)], time to maximum concentration, elimination half-life and electrocardiographic (ECG) parameters.
Results
Comparing the ratio of the pharmacokinetic parameter means (Cmax and AUC) with and without grapefruit juice (expressed as a percentage), the rate (Cmax) and extent (AUC) of absorption of fexofenadine were reduced by 30% by consumption of grapefruit juice. In contrast, the bioavailability of desloratadine was unaffected by grapefruit juice. No clinically significant changes in ECG parameters were observed following coadministration of grapefruit juice with desloratadine or fexofenadine compared with either antihistamine given alone.
Conclusion
The bioavailability of drugs that do not undergo significant intestinal or hepatic metabolism, such as fexofenadine, may be altered when administered with agents that influence drug transport mechanisms.




Similar content being viewed by others
Notes
Use of tradenames is for product identification only and does not imply endorsement.
References
Bailey DG, Malcolm J, Arnold O, et al. Grapefruit juice-drag interactions. Br J Clin Pharmacol 1998; 46: 101–10
Schmiedlin-Ren P, Edwards DJ, Fitzsimmons ME, et al. Mechanisms of enhanced oral availability of CYP3A4 substrates by grapefruit constituents. Decreased enterocyte CYP3A4 concentration and mechanism-based inactivation by furanocoumarins. Drug Metab Dispos 1997; 25: 1228–33
Kim RB, Wandel C, Leake B, et al. Interrelationship between substrates and inhibitors of human CYP3A and P-glycoprotein. Pharm Res 1999; 16: 408–14
Kane GC, Lipsky JJ. Drag-grapefruit juice interactions. Mayo Clin Proc 2000; 75: 933–42
Cvetkovic M, Leake B, Fromm MF, et al. OATP and P-glycoprotein transporters mediate the cellular uptake and excretion of fexofenadine. Drag Metab Dispos 1999; 27: 866–71
Fromm MF. P-glycoprotein: a defense mechanism limiting oral bioavailability and CNS accumulation of drags. Int J Clin Pharmacol Ther 2000; 38: 69–74
Schinkel AH, Wagenaar E, Mol CA, et al. P-glycoprotein in the blood-brain barrier of mice influences the brain penetration and pharmacological activity of many drugs. J Clin Invest 1996; 97: 2517–24
Dresser GK, Scharwz U, Leake B, et al. Grapefruit juice selectively inhibits OATP not P-glycoprotein [abstract]. Drag Metab Rev 2000; 32 Suppl. 2: 193
Allegra (fexofenadine) prescribing information. Physicians’ Desk Reference. Montvale (NJ): Medical Economics, 2000: 1343–4
Schwarz UI, Gramatte T, Krappweis J, et al. P-glycoprotein inhibitor erythromycin increases oral bioavailability of talinolol in humans. Int J Clin Pharmacol Ther 2000; 38: 161–7
Takano M, Hasegawa R, Fukuda T, et al. Interaction with P-glycoprotein and transport of erythromycin, midazolam and ketoconazole in Caco-2 cells. Eur J Pharmacol 1998; 358: 289–94
Kreutner W, Hey JA, Anthes J, et al. Preclinical pharmacology of desloratadine, a selective and nonsedating histamine H1 receptor antagonist. 1st communication: receptor selectivity, antihistaminic activity, and antiallergenic effects. Arzneimittelforschung 2000; 50: 345–52
Kreutner W, Hey JA, Chiu P, et al. Preclinical pharmacology of desloratadine, a selective and nonsedating histamine H1 receptor antagonist: 2nd communication: Lack of central nervous system and cardiovascular effects. Arzneimittelforschung 2000; 50: 441–8
Meltzer E, Premier B, Nayak A, et al. Efficacy and tolerability of once-daily 5mg desloratadine, an H1-receptor antagonist, in patients with seasonal allergic rhinitis: assessment during the spring and fall allergy seasons. Clin Drag Invest 2001; 21: 25–32
Geha RS, Meltzer EO. Desloratadine: a new, nonsedating, oral antihistamine. J Allergy Clin Immunol 2001; 107: 751–62
Ring J, Hein R, Gauger A, et al. Once-daily desloratadine improves the signs and symptoms of chronic idiopathic urticaria: a randomized, double-blind, placebo-controlled study. Int J Dermatol 2001; 40: 1–5
Affrime MB, Banfield C, Glue P, et al. Lack of electrocardio-graphic effects when desloratadine and ketoconazole are coadministered [abstract]. Allergy 2000; 55 Suppl. 63: 277
Glue P, Banfield C, Affrime MB, et al. Lack of electrocardio-graphic interaction between desloratadine and erythromycin [abstract]. Allergy 2000; 55 Suppl. 63: 276
Banfield C. Lack of drag interactions between desloratadine and other common medications [abstract]. Allergy 2001; 56 Suppl. 68: 200
Barecki ME, Casciano CN, Johnson WW, et al. In vitro characterization of the inhibition profile of loratadine, desloratadine, and 3-OH-desloratadine for five human cytochrome P-450 enzymes. Drug Metab Dispos 2001; 29: 1173–5
Wang E-J, Casciano CN, Clement RP, et al. Evaluation of the interaction of loratadine and desloraladine with P-glycoprotein. Drug Metab Dispos 2001; 29: 1080–3
Barbey JT, Anderson M, Ciprandi G, et al. Cardiovascular safety of second-generation antihistamines. Am J Rhinol 1999; 13: 235–43
Dresser GK, Bailey DG, Leake BF, et al. Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin Pharmacol Ther 2002; 71(1): 11–20
Fukuda K, Guo L, Ohashi N, et al. Amounts and variation in grapefruit juice of the main components causing grapefruit-drug interaction. J Chromatogr B Biomed Sci Appl 2000; 741: 195–203
Dresser GK, Schwarz UI, Leake B, et al. Citrus juices are potent inhibitors of intestinal OATP but not P-glycoprotein [abstract]. Clin Pharmacol Ther 2001; 69: 23
Acknowledgements
This work was supported by a research grant from Schering-Plough Research Institute, Kenilworth, New Jersey, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banfield, C., Gupta, S., Marino, M. et al. Grapefruit Juice Reduces the Oral Bioavailability of Fexofenadine But Not Desloratadine. Clin Pharmacokinet 41, 311–318 (2002). https://doi.org/10.2165/00003088-200241040-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200241040-00004