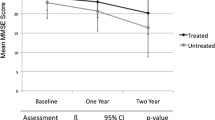
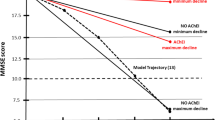
Abstract
Cholinesterase inhibitors have been available for the treatment of Alzheimer’s disease since 1993. They have significantly positive effects on cognitive functioning and other domains of functional capacity, such as activities of daily life in terms of efficacy, but the clinical value of these effects are under discussion. Cholinesterase inhibitors may also influence behavioural and psychological symptoms in Alzheimer’s disease. Cholinesterase inhibitors are also regarded as rather expensive and, therefore, the question of cost effectiveness is essential. Pharmacoeconomic evaluations of cholinesterase inhibitors have so far been conducted in retrospect on efficacy data from prospective randomised clinical trials combined with economic data from other sources. There are no published specific cost-effectiveness studies of cholinesterase inhibitors which prospectively collected empirical data on costs and outcomes. There is only one published randomised clinical trial with such empirical data with a cost consequence analysis design, indicating cost neutrality. Several types of models to describe the long-term effects have been published, indicating cost effectiveness. However, due to methodological considerations, the validity of these models is difficult to judge. A research agenda for the cost effectiveness of cholinesterase inhibitors is proposed, including long-term studies with empirical data on resource use, costs and outcomes, studies on quality of life, informal care and behavioural and psychological symptoms, combination and comparative studies on mild cognitive impairment.




Similar content being viewed by others
References
Wimo A, Winblad B, Aguero Torres H, et al. The magnitude of dementia occurrence in the world. Alzheimer Dis Assoc Disord 2003; 17: 63–7
Wimo A, Winblad B, Grafstrom M. The social consequences for families with Alzheimer’s disease patients: potential impact of new drug treatment. Int J Geriatr Psychiatry 1999; 14: 338–47
OECD. Policy issues in dementia care in OECD countries. Paris: Organisation for Economic Cooperation and Development, 2002
WHO. World health report, 1999. Geneva: World Health Organization, 1999
Nordberg A, Nilsson-Hakansson L, Adem A, et al. Multiple actions of THA on cholinergic neurotransmission in Alzheimer brains. Prog Clin Biol Res 1989; 317: 1169–78
Knopman DS. Current pharmacotherapies for Alzheimer’s disease. Geriatrics 1998; 53Suppl. 1: S31–4
Knopman D, Schneider L, Davis K, et al. Long-term tacrine (Cognex) treatment: effects on nursing home placement and mortality. Tacrine Study Group. Neurology 1996; 47: 166–77
Lubeck DP, Mazonson PD, Bowe T. Potential effect of tacrine on expenditures for Alzheimer’s disease. Med Interface 1994; 7: 130–8
Henke CJ, Burchmore MJ. The economic impact of the tacrine in the treatment of Alzheimer’s disease. Clin Ther 1997; 19: 330–45
Wimo A, Karlsson G, Nordberg A, et al. Treatment of Alzheimer’s disease with tacrine: a cost-analysis model. Alzheimer Dis Assoc Disord 1997; 11: 191–200
Jonsson B, Jonsson L, Wimo A. Cost of dementia. In: May M, Sartorius N, editors. Dementia: WPA series evidence and experience in psychiatry. London: John Wiley & Son, 2000: 335–63
Whitehouse PJ, Winblad B, Shostak D, et al. First International Pharmacoeconomic Conference on Alzheimer’s Disease: report and summary. Alzheimer Dis Assoc Disord 1998; 12: 266–80
Jonsson L, Jonsson B, Wimo A, et al. Second International Pharmacoeconomic Conference on Alzheimer’s Disease. Alzheimer Dis Assoc Disord 2000; 14: 137–40
Siegel JE, Torrance GW, Russell LB, et al. Guidelines for pharmacoeconomic studies: recommendations from the panel on cost effectiveness in health and medicine: panel on cost effectiveness in health and medicine. Pharmacoeconomics 1997; 11: 159–68
Torrance G. Designing and conducting cost-utility analysis. In: Spilker B, editor. Quality of life and pharmacoeconomics in clinical trials. Philadelphia (PA): Lippincott-Raven, 1996: 1105–21
Wimo A, Wetterholm AL, Mastey V, et al. Evaluation of the resource utilization and caregiver time in anti-dementia drug trials: a quantitative battery. In: Wimo A, Jonsson B, Karlsson G, et al., editors. The health economics of dementia. London: John Wiley & Sons, 1998: 465–99
Wimo A, Winblad B. Resource utilisation in dementia: RUD Lite. Brain Aging 2003; 3: 48–59
Wimo A, Winblad B, Stöffler A, et al. Resource utilization and cost analysis of memantine in patients with moderate to severe Alzheimer’s disease. Pharmacoeconomics 2003; 21: 327–40
Wimo A, Winblad B, Engedal K, et al. An economic evaluation of donepezil in mild to moderate Alzheimer’s disease: results of a 1-year, double-blind, randomized trial. Dement Geriatr Cogn Disord 2003; 15: 44–54
Drummond MF, O’Brien B, Stoddart GL, et al. Methods for the economic evaluation of health care programmes. Oxford: Oxford University Press, 1997
Winblad B, Hill S, Beermann B, et al. Issues in the economic evaluation of treatment for dementia: position paper from the International Working Group on Harmonization of Dementia Drug Guidelines. Alzheimer Dis Assoc Disord 1997; 11: 39–45
Freund DA, Dittus RS. Principles of pharmacoeconomic analysis of drug therapy. Pharmacoeconomics 1992; 1: 20–31
Busschbach JJ, Brouwer WB, van der Donk A, et al. An outline for a cost-effectiveness analysis of a drug for patients with Alzheimer’s disease. Pharmacoeconomics 1998; 13: 21–34
Wimo A, von Strauss E, Nordberg G, et al. Time spent on informal and formal care giving for persons with dementia in Sweden. Health Policy 2002; 61: 255–68
McDaid D. Estimating the costs of informal care for people with Alzheimer’s disease: methodological and practical challenges. Int J Geriatr Psychiatry 2001; 16: 400–5
Moore MJ, Zhu CW, Clipp EC. Informal costs of dementia care: estimates from the National Longitudinal Caregiver Study. J Gerontol B Psychol Sci Soc Sci 2001; 56: S219–28
Langa KM, Chernew ME, Kabeto MU, et al. National estimates of the quantity and cost of informal caregiving for the elderly with dementia. J Gen Intern Med 2001; 16: 770–8
Max W. The cost of Alzheimer’s disease: will drug treatment ease the burden? Pharmacoeconomics 1996; 9: 5–10
Clipp EC, Moore MJ. Caregiver time use: an outcome measure in clinical trial research on Alzheimer’s disease. Clin Pharmacol Ther 1995; 58: 228–36
Davis KL, Marin DB, Kane R, et al. The Caregiver Activity Survey (CAS): development and validation of a new measure for caregivers of persons with Alzheimer’s disease. Int J Geriatr Psychiatry 1997; 12: 978–88
Werner P, Schnaider-Beeri M, Aharon J, et al. Family caregivers’ willingness to pay for drugs indicated for the treatment of Alzheimer’s disease: an economic or psychological model? Dementia 2002; 1: 59–75
Sano M, Ernesto C, Thomas RG, et al. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. N Engl J Med 1997; 336: 1216–22
Whitehouse PJ, Kittner B, Roessner M, et al. Clinical trial designs for demonstrating disease-course-altering effects in dementia. Alzheimer Dis Assoc Disord 1998; 12: 281–94
Selai C. Assessing quality of life in dementia. Med Care 2001; 39: 753–5
Salek SS, Walker MD, Bayer AJ. A review of quality of life in Alzheimer’s disease (Pt 2): issues in assessing drug effects. Pharmacoeconomics 1998; 14: 613–27
Walker MD, Salek SS, Bayer AJ. A review of quality of life in Alzheimer’s disease (Pt 1): issues in assessing disease impact. Pharmacoeconomics 1998; 14: 499–530
Coucill W, Bryan S, Bentham P, et al. EQ-5D in patients with dementia: an investigation of inter-rater agreement. Med Care 2001; 39: 760–71
Neumann PJ, Sandberg EA, Araki SS, et al. A comparison of HUI2 and HUI3 utility scores in Alzheimer’s disease. Med Decis Making 2000; 20: 413–22
Kerner DN, Patterson TL, Grant I, et al. Validity of the quality of well-being scale for patients with Alzheimer’s disease. J Aging Health 1998; 10: 44–61
Brod M, Stewart AL, Sands L, et al. Conceptualization and measurement of quality of life in dementia: the dementia quality of life instrument (DQoL). Gerontologist 1999; 39: 25–35
Logsdon RG, Gibbons LE, McCurry SM, et al. Assessing quality of life in older adults with cognitive impairment. Psychosom Med 2002; 64: 510–9
Selai C, Vaughan A, Harvey RJ, et al. Using the QOL-AD in the UK. Int J Geriatr Psychiatry 2001; 16: 537–8
Elstner K, Selai CE, Trimble MR, et al. Quality of Life (QOL) of patients with Gilles de la Tourette’s syndrome. Acta Psychiatr Scand 2001; 103: 52–9
Selai CE, Elstner K, Trimble MR. Quality of life pre and post epilepsy surgery. Epilepsy Res 2000; 38: 67–74
Berg L. Clinical dementia rating (CDR). Psychopharmacol Bull 1988; 24: 637–9
Reisberg B, Ferris SH, de Leon MJ, et al. Global deterioration scale (GDS). Psychopharmacol Bull 1988; 24: 661–3
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12: 189–98
Palmer K, Fratiglioni L, Winblad B. What is mild cognitive impairment: variations in definitions and evolution of nondemented persons with cognitive impairment. Acta Neurol Scand Suppl 2003; 179: 14–20
Fratiglioni L, Viitanen M, Backman L, et al. Occurrence of dementia in advanced age: the study design of the Kungsholmen Project. Neuroepidemiology 1992; 11Suppl. 1: 29–36
Aguero-Torres H, Fratiglioni L, Winblad B. Natural history of Alzheimer’s disease and other dementias: review of the literature in the light of the findings from the Kungsholmen Project. Int J Geriatr Psychiatry 1998; 13: 755–66
Ott BR, Lapane KL. Tacrine therapy is associated with reduced mortality in nursing home residents with dementia. J Am Geriatr Soc 2002; 50: 35–40
Geldmacher DS, Provenzano G, McRae T, et al. Donepezil is associated with delayed nursing home placement in patients with Alzheimer’s disease. J Am Geriatr Soc 2003; 51: 937–44
Lopez OL, Becker JT, Wisniewski S, et al. Cholinesterase inhibitor treatment alters the natural history of Alzheimer’s disease. J Neurol Neurosurg Psychiatry 2002; 72: 310–4
Small GW, Donohue JA, Brooks RL. An economic evaluation of donepezil in the treatment of Alzheimer’s disease. Clin Ther 1998; 20: 838–50
Hill JW, Futterman R, Mastey V, et al. The effect of donepezil therapy on health costs in a Medicare managed care plan. Manag Care Interface 2002; 15: 63–70
Fillit H, Gutterman EM, Lewis B. Donepezil use in managed Medicare: effect on health care costs and utilization. Clin Ther 1999; 21: 2173–85
Winblad B, Engedal K, Soininen H, et al. A 1-year, randomized, placebo-controlled study of donepezil in patients with mild to moderate AD. Neurology 2001; 57: 489–95
Sonnenberg FA, Leventhal EA. Modeling disease progression with Markov models. In: Wimo A, Jonsson B, Karlsson G, et al., editors. Health economics of dementia. London: John Wiley and Sons, 1998: 171–96
Fenn P, Gray A. Estimating long-term cost savings from treatment of Alzheimer’s disease: a modelling approach. Pharmacoeconomics 1999; 16: 165–74
Caro JJ, Getsios D, Migliaccio-Walle K, et al. Assessment of health economics in Alzheimer’s disease (AHEAD) based on need for full-time care. Neurology 2001; 57: 964–71
Stern Y, Tang MX, Albert MS, et al. Predicting time to nursing home care and death in individuals with Alzheimer disease. JAMA 1997; 277: 806–12
Hauber AB, Gnanasakthy A, Snyder EH, et al. Potential savings in the cost of caring for Alzheimer’s disease: treatment with rivastigmine. Pharmacoeconomics 2000; 17: 351–60
Ernst RL, Hay JW, Fenn C, et al. Cognitive function and the costs of Alzheimer disease: an exploratory study. Arch Neurol 1997; 54: 687–93
Hauber AB, Gnanasakthy A, Mauskopf JA. Savings in the cost of caring for patients with Alzheimer’s disease in Canada: an analysis of treatment with rivastigmine. Clin Ther 2000; 22: 439–51
Garfield FB, Getsios D, Caro JJ, et al. Assessment of Health Economics in Alzheimer’s Disease (AHEAD): treatment with galantamine in Sweden. Pharmacoeconomics 2002; 20: 629–37
Stewart A, Phillips R, Dempsey G. Pharmacotherapy for people with Alzheimer’s disease: a Markov-cycle evaluation of five years’ therapy using donepezil. Int J Geriatr Psychiatry 1998; 13: 445–53
O’Brien BJ, Goeree R, Hux M, et al. Economic evaluation of donepezil for the treatment of Alzheimer’s disease in Canada. J Am Geriatr Soc 1999; 47: 570–8
Jonsson L, Lindgren P, Wimo A, et al. The cost-effectiveness of donepezil therapy in Swedish patients with Alzheimer’s disease: a Markov model. Clin Ther 1999; 21: 1230–40
Neumann PJ, Hermann RC, Kuntz KM, et al. Cost-effectiveness of donepezil in the treatment of mild or moderate Alzheimer’s disease. Neurology 1999; 52: 1138–45
Ikeda S, Yamada Y, Ikegami N. Economic evaluation of donepezil treatment for Alzheimer’s disease in Japan. Dement Geriatr Cogn Disord 2002; 13: 33–9
Getsios D, Caro JJ, Caro G, et al. Assessment of Health Economics in Alzheimer’s Disease (AHEAD): galantamine treatment in Canada. Neurology 2001; 57: 972–8
Caro JJ, Salas M, Ward A, et al. Economic analysis of galantamine, a cholinesterase inhibitor, in the treatment of patients with mild to moderate Alzheimer’s disease in The Netherlands. Dement Geriatr Cogn Disord 2002; 14: 84–9
Migliaccio-Walle K, Getsios D, Caro JJ, et al. Economic evaluation of galantamine in the treatment of mild to moderate Alzheimer’s disease in the United States. Clin Ther 2003; 25: 1806–25
Ward A, Caro JJ, Getsios D, et al. Assessment of Health Economics in Alzheimer’s Disease (AHEAD): treatment with galantamine in the UK. Int J Geriatr Psychiatry 2003; 18: 740–7
Thompson SG, Barber JA. How should cost data in pragmatic randomised trials be analysed? BMJ 2000; 320: 1197–200
Buxton MJ, Drummond MF, Van Hout BA, et al. Modelling in economic evaluation: an unavoidable fact of life. Health Econ 1997; 6: 217–27
Jonsson L. Pharmacoeconomics of cholinesterase inhibitors in the treatment of Alzheimer’s disease. Pharmacoeconomics 2003; 21: 1025–37
Hux MJ, O’Brien BJ, Iskedjian M, et al. Relation between severity of Alzheimer’s disease and costs of caring. CMAJ 1998; 159: 457–65
Jonsson L, Lindgren P, Wimo A, et al. Costs of mini mental state examination-related cognitive impairment. Pharmacoeconomics 1999; 16: 409–16
Schneider LS, Olin JT, Lyness SA, et al. Eligibility of Alzheimer’s disease clinic patients for clinical trials. J Am Geriatr Soc 1997; 45: 923–8
Schafer JL, Grahan JW. Missing data: our view of the state of the art. Psychol Methods 2002; 7: 147–77
Knopman D. Tacrine for Alzheimer’s disease: costs and benefits. Pharmacoeconomics 1995; 7: 275–9
Ernst RL, Hay JW. Economic research on Alzheimer disease: a review of the literature. Alzheimer Dis Assoc Disord 1997; 11: 135–45
Whitehouse P. Cholinesterase inhibitors in Alzheimer’s disease: are they worth the cost? CNS Drugs 1999; 11: 167–73
Stein K. Donepezil in the treatment of mild to moderate senile dementia of the Alzheimer type (SDAT). Southampton: Wes-sex Institute for Health Research and Development (NHS Executive South and West, Development and Evaluation Committee), 1997
Glennie J. The efficacy of tacrine and the measurement of outcomes in Alzheimer’s disease. Ottawa (ON): The Canadian Coordinating Office for Health Technology Assessment (CCOHTA), 1997
Molnar FJ, Dalziel WB. The pharmacoeconomics of dementia therapies: bringing the clinical, research and economic perspectives together. Drugs Aging 1997; 10: 219–33
Foster RH, Plosker GL. Donepezil: pharmacoeconomic implications of therapy. Pharmacoeconomics 1999; 16: 99–114
Shukla VK, Otten N, Coyle D. Drug treatments for Alzheimer’s disease (III): a review of published pharmacoeconomic evaluations. Ottawa (ON): Canadian Coordinating Office for Health Technology Assessment (CCOHTA), 2000: 37
Wolfson C, Oremus M, Shukla V, et al. Donepezil and rivastigmine in the treatment of Alzheimer’s disease: a best-evidence synthesis of the published data on their efficacy and cost-effectiveness. Clin Ther 2002; 24: 862–86
NICE. Guidance on the use of donepezil, rivastigmine and galantamine for the treatment of Alzheimer’s disease. London: National Institute for Clinical Excellence, 2001: 5–6
Grutzendler J, Morris JC. Cholinesterase inhibitors for Alzheimer’s disease. Drugs 2001; 61: 41–52
Lamb HM, Goa KL. Rivastigmine: a pharmacoeconomic review of its use in Alzheimer’s disease. Pharmacoeconomics 2001; 19: 303–18
Clegg A, Bryant J, Nicholson T, et al. Clinical and cost-effectiveness of donepezil, rivastigmine, and galantamine for Alzheimer’s disease: a systematic review. Int J Technol Assess Health Care 2002; 18: 497–507
Lyseng-Williamson KA, Plosker GL. Galantamine: a pharmacoeconomic review of its use in Alzheimer’s disease. Pharmacoeconomics 2002; 20: 919–42
Lyseng-Williamson KA, Plosker GL. Spotlight on galantamine in Alzheimer’s disease. Dis Manag Health Outcomes 2003; 11: 125–8
Gray A. Health economics. In: Qizilbash N, Schneider L, Chui H, et al., editors. Evidence-based dementia practice. Oxford: Blackwell Publishing, 2002: 844–54
Olin J, Schneider L. Galantamine for Alzheimer’s disease (Cochrane Review). Available in The Cochrane Library [database on disk and CD ROM]. Updated quarterly. The Cochrane Collaboration; issue 2. Oxford: Update Software, 2003
Leung GM, Yeung RY, Chi I, et al. The economics of Alzheimer disease. Dement Geriatr Cogn Disord 2003; 15: 34–43
Birks JS, Harvey R. Donepezil for dementia due to Alzheimer’s disease (Cochrane review). Available in The Cochrane Library [database on disk and CD ROM]. Updated quarterly. The Cochrane Collaboration; issue 3. Oxford: Update Software, 2003
Wimo A, Jonsson B, Karlsson G, et al. The health economics of dementia. London: John Wiley & Sons, 1998
Briggs A. Economic evaluation and clinical trials: size matters. BMJ 2000; 321: 1362–3
Olsen JA, Smith RD. Theory versus practice: a review of “willingness-to-pay” in health and health care. Health Econ 2001; 10: 39–52
Stewart A, Brod M. Measuring health related quality of life in older and demented people. In: Spilker B, editor. Quality of life and pharmacoeconomics in clinical trials. Philadelphia (PA): Lippincott-Raven, 1996: 819–30
Tariot PN, Farlow MR, Grossberg GT. Memantine treatment in patients with moderate to severe Alzheimer disease already receiving donepezil: a randomized controlled trial. JAMA 2004; 291: 317–24
Finkel S. The significance of the behavioural and psychological symptoms of dementia. Clinician 1998; 16: 33–42
Murman DL, Chen Q, Powell MC, et al. The incremental direct costs associated with behavioral symptoms in AD. Neurology 2002; 59: 1721–9
Beeri MS, Werner P, Davidson M, et al. The cost of behavioral and psychological symptoms of dementia (BPSD) in community dwelling Alzheimer’s disease patients. Int J Geriatr Psychiatry 2002; 17: 403–8
Hemels ME, Lanctot KL, Iskedjian M, et al. Clinical and economic factors in the treatment of behavioural and psychological symptoms of dementia. Drugs Aging 2001; 18: 527–50
Brodaty H, Clarke J, Ganguli M, et al. Screening for cognitive impairment in general practice: toward a consensus. Alzheimer Dis Assoc Disord 1998; 12: 1–13
Wimo A, Winblad B. Pharmacoeconomics of mild cognitive impairment. Acta Neurol Scand Suppl 2003; 179: 94–9
Knottnerus JA. Dementia. The Hague: Health Council of The Netherlands, 2002
Melzer D. New drug treatment for Alzheimer’s disease: lessons for healthcare policy. BMJ 1998; 316: 762–4
Shah A, Jenkins R. “Value for money” in treating Alzheimer’s disease with the new cholinesterase inhibitors. Int J Psychiatry Clin Pract 2003; 7: 45–7
Acknowledgements
Dr Wimo has no shares or employment in any pharmaceutical company. He has, or has been, acting as a consultant to Parke-Davis, HMR/Aventis, Pfizer, Novartis, Janssen-Cilag, and Merz, Lundbeck.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wimo, A. Cost Effectiveness of Cholinesterase Inhibitors in the Treatment of Alzheimer’s Disease. Drugs Aging 21, 279–295 (2004). https://doi.org/10.2165/00002512-200421050-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-200421050-00001