Abstract
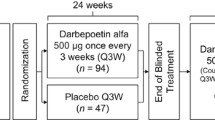
To investigate the efficacy of using recombinant human interleukin 11 (rhIL-11) to reduce the need for platelet transfusions, we performed a randomized, double-blind phase II/III study with 110 acute myelogenous leukemia (AML) patients in the first complete remission. Following chemotherapy patients were subcutaneously administered either placebo (n = 37) or rhIL-11 at a dose of 25 μg/kg (n = 37) or 50 μg/kg (n = 36). rhIL-11 administration was well tolerated. There was no difference between the rhIL-11 and placebo groups in the frequency and volume of platelet transfusions. In a perprotocol analysis set (101 patients), the platelet transfusion frequency in the 50-μg/kg group (3.0 ± 1.76 times) was significantly lower than in the placebo group (3.9 ±2.35 times; multiplicity-adjusted P = .049). We analyzed infection-related events retrospectively. The frequency of fever was significantly decreased in the 50-μg/kg, 25-μg/kg, and placebo groups (66.7%, 70.3%, and 89.2%, respectively; P = .018, Cochran-Armitage test). Stomatitis was less frequent in the 50-μg/kg and 25-μg/kg groups (2.8% and 0%, respectively) than in the placebo group (21.6%, P = .0012). These results show that rhIL-11 does not reduce the platelet transfusion requirement in AML patients, but the retrospective analysis confirms the previous finding that rhIL-11 reduces infection in patients undergoing chemotherapy.
Similar content being viewed by others
References
Anderlini P, Ghaddar HM, Smith TL, et al. Factors predicting complete remission and subsequent disease-free survival after a second course of induction therapy in patients with acute myelogenous leukemia resistant to the first. Leukemia. 1996;10:964–969.
Tornebohm E, Lockner D, Paul C. A retrospective analysis of bleeding complications in 438 patients with acute leukaemia during the years 1972—1991. Eur J Haematol. 1993;50:160–167.
Gmür J, Burger J, Schanz U, Fehr J, Schaffner A. Safety of stringent platelet transfusion policy for patients with acute leukaemia. Lancet. 1991;338:1223–1226.
Rebulla P, Finazzi G, Marangoni F, et al. The threshold for prophylactic platelet transfusions in adults with acute myeloid leukemia. N Engl J Med. 1997;337:1870–1875.
Schiffer CA, Anderson KC, Bennett CL, et al, for the American Society of Clinical Oncology. Platelet transfusion for patients with cancer: clinical practice guidelines of the American Society of Clinical Oncology. J Clin Oncol. 2001;19:1519–1538.
Surgenor DM, Wallace EL, Hao SH, Chapman RH. Collection and transfusion of blood in the United States, 1982—1988. N Engl J Med. 1990;322:1646–1651.
Wallace EL, Churchill WH, Surgenor DM, Cho GS, McGurk S. Collection and transfusion of blood and blood components in the United States, 1994. Transfusion. 1998;38:625–636.
Bishop JF, Matthews JP, Young GA, et al. A randomized study of high-dose cytarabine in induction in acute myeloid leukemia. Blood. 1996;87:1710–1717.
Zittoun RA, Mandelli F, Willemze R, et al. Autologous or allogeneic bone marrow transplantation compared with intensive chemotherapy in acute myelogenous leukemia. N Engl J Med. 1995;332:217–223.
Godwin JE, Kopecky KJ, Head DR, et al. A double-blind placebo-controlled trial of granulocyte colony-stimulating factor in elderly patients with previously untreated acute myeloid leukemia: a Southwest Oncology Group study (9031). Blood. 1998;91:3607–3615.
Heil G, Hoelzer D, Sanz MA, et al, for the International Acute Myeloid Leukemia Study Group. A randomized, double-blind, placebo-controlled, phase III study of filgrastim in remission induction and consolidation therapy for adults with de novo acute myeloid leukemia. Blood. 1997;90:4710–4718.
Usuki K, Urabe A, Masaoka T, et al. Efficacy of granulocyte colony-stimulating factor in the treatment of acute myelogenous leukaemia: a multicentre randomized study. Br J Haematol. 2002;116:103–112.
Maslak P, Nimer SD. The efficacy of IL-3, SCF, IL-6, and IL-11 in treating thrombocytopenia. Semin Hematol. 1998;35:253–260.
Du X, Williams DA. Interleukin-11: review of molecular, cell biology, and clinical use. Blood. 1997;89:3897–3908.
Vij R, DiPersio J. The effects of multilineage cytokines on platelet recovery. In: Wingard JR, Demetri GD, eds. Clinical Applications of Cytokines and Growth Factors. Norwell, Mass: Kluwer Academic Publishers; 1999:237–268.
Paul SR, Bennett F, Calvetti JA, et al. Molecular cloning of a cDNA encoding interleukin 11, a stromal cell-derived lymphopoietic and hematopoietic cytokine. Proc Natl Acad Sci U S A. 1990;87:7512–7516.
Musashi M,Yang YC, Paul SR, Clark SC, Sudo T, Ogawa M. Direct and synergistic effects of interleukin 11 on murine hemopoiesis in culture. Proc Natl Acad Sci U S A. 1991;88:765–769.
Musashi M, Clark SC, Sudo T, Urdal DL, Ogawa M. Synergistic interactions between interleukin-11 and interleukin-4 in support of proliferation of primitive hematopoietic progenitors of mice. Blood. 1991;78:1448–1451.
Teramura M, Kobayashi S, Hoshino S, Oshimi K, Mizoguchi H. Interleukin-11 enhances human megakaryocytopoiesis in vitro. Blood. 1992;79:327–331.
Du XX, Neben T, Goldman S,Williams DA. Effects of recombinant human interleukin-11 on hematopoietic reconstitution in transplant mice: acceleration of recovery of peripheral blood neutrophils and platelets. Blood. 1993;81:27–34.
Leonard JP, Quinto CM, Kozitza MK, Neben TY, Goldman SJ. Recombinant human interleukin-11 stimulates multilineage hematopoietic recovery in mice after a myelosuppressive regimen of sublethal irradiation and carboplatin. Blood. 1994;83:1499–1506.
Neben TY, Loebelenz J, Hayes L, et al. Recombinant human interleukin-11 stimulates megakaryocytopoiesis and increases peripheral platelets in normal and splenectomized mice. Blood. 1993;81:901–908.
Gordon MS, McCaskill-Stevens WJ, Battiato LA, et al. A phase I trial of recombinant human interleukin-11 (Neumega rhIL-11 growth factor) in women with breast cancer receiving chemotherapy. Blood. 1996;87:3615–3624.
Isaacs C, Robert NJ, Bailey FA, et al. Randomized placebo-controlled study of recombinant human interleukin-11 to prevent chemotherapy-induced thrombocytopenia in patients with breast cancer receiving dose-intensive cyclophosphamide and doxorubicin. J Clin Oncol. 1997;15:3368–3377.
Tepler I, Elias L, Smith JW 2nd, et al. A randomized placebo-controlled trial of recombinant human interleukin-11 in cancer patients with severe thrombocytopenia due to chemotherapy. Blood. 1996;87:3607–3614.
Cairo MS, Davenport V, Bessmertny O, et al. Phase I/II dose escalation study of recombinant human interleukin-11 following ifosfamide, carboplatin and etoposide in children, adolescents and young adults with solid tumours or lymphoma: a clinical, haematological and biological study. Br J Haematol. 2005;128:49–58.
Ellis M, Zwaan F, Hedstrom U, et al. Recombinant human interleukin 11 and bacterial infection in patients with haematological malignant disease undergoing chemotherapy: a double-blind placebo-controlled randomised trial. Lancet. 2003;361:275–280.
Ohno R, Miyawaki S, Hatake K, et al. Human urinary macrophage colony-stimulating factor reduces the incidence and duration of febrile neutropenia and shortens the period required to finish three courses of intensive consolidation therapy in acute myeloid leukemia: a double-blind controlled study. J Clin Oncol. 1997;15:2954–2965.
Berridge MV, Fraser JK, Carter JM, Lin FK. Effects of recombinant human erythropoietin on megakaryocytes and on platelet production in the rat. Blood. 1988;72:970–977.
Dessypris EN, Gleaton JH, Armstrong OL. Effect of human recombinant erythropoietin on human marrow megakaryocyte colony formation in vitro. Br J Haematol. 1987;65:265–269.
Urabe A. Clinical features of the neutropenic host: definitions and initial evaluation. Clin Infect Dis. 2004;15(suppl 1):S53-S55.
Bussel JB, Kuter DJ, Geroge JN, et al. Long-term dosing of AMG531 is effective and well tolerated in thrombocytopenic patients with immune thrombocytopenic purpura [abstract]. Blood. 2005;106:220a.
Vredenburgh JJ, Hussein A, Fisher D, et al. A randomized trial of recombinant human interleukin-11 following autologous bone marrow transplantation with peripheral blood progenitor cell support in patients with breast cancer. Biol Blood Marrow Transplant. 1998;4:134–141.
Fanucchi M, Glaspy J, Crawford J, et al. Effects of polyethylene glycol-conjugated recombinant human megakaryocyte growth and development factor on platelet counts after chemotherapy for lung cancer. N Engl J Med. 1997;336:404–409.
Vadhan-Raj S, Murray LJ, Bueso-Ramos C, et al. Stimulation of megakaryocyte and platelet production by a single dose of recombinant human thrombopoietin in patients with cancer. Ann Intern Med. 1997;26:673–681.
Archimbauddagger E, Ottmann OG, Liu Yin JA, et al. A randomized, double-blind, placebo-controlled study with pegylated recombinant human megakaryocyte growth and development factor (PEG-rHuMGDF) as an adjunct to chemotherapy for adults with de novo acute myeloid leukemia. Blood. 1999;94:3694–3701.
Schiffer CA, Miller K, Larson RA, et al. A double-blind, placebo-controlled trial of pegylated recombinant human megakaryocyte growth and development factor as an adjunct to induction and consolidation therapy for patients with acute myeloid leukemia. Blood. 2000;95:2530–2535.
Geissler K, Yin JA, Ganser A, et al. Prior and concurrent administration of recombinant human megakaryocyte growth and development factor in patients receiving consolidation chemotherapy for de novo acute myeloid leukemia: a randomized, placebo-controlled, double-blind safety and efficacy study. Ann Hematol. 2003;82:677–683.
Stepan DE, Sergis-Deavenport E, Kelly R, Christal J, Chen C-F, Nicol JL. Safety profile of AMG531 in healthy volunteers and in thrombocytopenic patients with immune thrombocytopenic purpura [abstract]. Blood. 2005;106:1240a.
Du XX, Doerschuk CM, Orazi A, Williams DA. A bone marrow stromal-derived growth factor, interleukin-11, stimulates recovery of small intestinal mucosal cells after cytoablative therapy. Blood. 1994;83:33–38.
Sonis ST, Peterson RL, Edwards LJ, et al. Defining mechanisms of action of interleukin-11 on the progression of radiation-induced oral mucositis in hamsters. Oral Oncol. 2000;36:373–381.
Opal SM, Jhung JW, Keith JC Jr, Goldman SJ, Palardy JE, Parejo NA. Additive effects of human recombinant interleukin-11 and granulocyte colony-stimulating factor in experimental gram-negative sepsis. Blood. 1999;93:3467–3472.
Hu JP, Cesano A, Santoli D, Clark SC, Hoang T. Effects of interleukin-11 on the proliferation and cell cycle status of myeloid leukemic cells. Blood. 1993;81:1586–1592.
Kimura T, Sakabe H, Minamiguchi H, et al. Interleukin-11 (IL-11) enhances clonal proliferation of acute myelogenous leukemia cells with strong expression of the IL-11 receptor alpha chain and signal transducing gp130. Leukemia. 1999;13:1018–1027.
Lemoli RM, Fogli M, Fortuna A, et al. Interleukin-11 (IL-11) acts as a synergistic factor for the proliferation of human myeloid leukaemic cells. Br J Haematol. 1991;91:319–326.
Estey EH, Thall PF, Giles FJ, et al. Gemtuzumab ozogamicin with or without interleukin 11 in patients 65 years of age or older with untreated acute myeloid leukemia and high-risk myelodysplastic syndrome: comparison with idarubicin plus continuous-infusion, high-dose cytosine arabinoside. Blood. 2002;99:4343–4349.
Kurzrock R, Cortes J, Thomas DA, Jeha S, Pilat S, Talpaz M. Pilot study of low-dose interleukin-11 in patients with bone marrow failure. J Clin Oncol. 2001;19:4165–4172.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Usuki, K., Urabe, A., Ikeda, Y. et al. A Multicenter Randomized, Double-Blind, Placebo-Controlled Late-Phase II/III Study of Recombinant Human Interleukin 11 in Acute Myelogenous Leukemia. Int J Hematol 85, 59–69 (2007). https://doi.org/10.1532/IJH97.06027
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1532/IJH97.06027