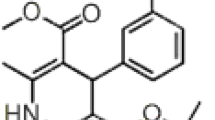
Abstract
A reversed-phase high-performance liquid chromatographic method has been developed and validated for quantitative determination of deflazacort in tablets and in compounded capsules. Isocratic chromatography was performed on a C18 column with acetonitrile–water 80:20 (v/v) as mobile phase at a flow rate of 1.0 mL min−1. UV detection was at 240 nm. The linearity of the method was good (r > 0.999), as also were intra-day and inter-day precision (RSD <2%) and accuracy (recovery >98%). The method was also validated for specificity and robustness. The results showed the proposed method is suitable for its intended use.


Similar content being viewed by others
References
Markham A, Bryson HM (1995) Drugs 50:317–333
Bernareggi A, Polett P, Zanolo G, Zerilli LF (1987) J Pharm Biomed Anal 5:177–181
Hirata H, Kasama T, Sawai Y, Fike RR (1994) J Chromatogr B 658:55–61
Reynolds DL, Burmaster SD, Eichmeier LS (1994) Biomed Chromatogr 8:230–235
Santos-Montes A, Gonzalo-Lumbreras R, Gasco-Lopes AI, Isquierdo-Hornillos R (1994) J Chromatogr B 657:248–253
Santos-Montes A, Izquierdo-Hornillos R (1999) J Chromatogr B 724:53–63
Ifa DR, Moraes ME, Moraes MO, Santagada V, Caliendo G, De Nucci G (2000) J Mass Spectrom 35:440–445
Özkan Y, Savaser A, Tas Ç, Uslu B, Özkan SA (2003) J Liq Chromatogr Relat Technol 26:2141–2156
Yapar D, Dal AG, Tuncel M, Uysal UD (2004) J Liq Chromatogr Relat Technol 27:2593–2601
ICH (2005) Harmonised tripartite guideline: validation of analytical procedures: text and methodology Q2 (R1)
Metha AC (1997) Analyst 122:83R–88R
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Corrêa, G.M., Bellé, L.P., Bajerski, L. et al. Development and Validation of a Reversed-Phase HPLC Method for the Determination of Deflazacort in Pharmaceutical Dosage Forms. Chroma 65, 591–594 (2007). https://doi.org/10.1365/s10337-007-0205-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-007-0205-y