Abstract
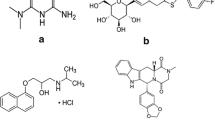
A simple, precise and reproducible liquid chromatography–tandem mass spectrometry method has been developed and validated according to the Food and Drug Administration guidelines for the simultaneous quantitation of antidiabetic drugs metformin, glimiperide and pioglitazone in human plasma using glipizide as an internal standard. Quantitation was performed on a triple quadrupole mass spectrometer employing electrospray ionization technique, operating in multiple reaction monitoring and positive ion mode. Inter-batch and intra-batch coefficient of variation across four validation runs for the quality control samples was less than 7%. The accuracy determined at quality control levels was within 92.81–105.13%. The method was applied to a bioequivalence study.





Similar content being viewed by others
References
DeFronzo RA (1997) Diabetes 4:177–269
Reaven GM (1988) Diabetes 37:595–607
Lillioja S, Mott DM, Howard BV, Bennett PH, Yki-Jarvinen H, Freymond D, Nyomba BL, Zurlo F, Swinburn B, Bogardus C (1988) N Engl J Med 318:1217–1225
Prospective Diabetes Study Group (1998) Lancet 352:837–853. doi:10.1016/S0140-6736(98)07019-6
Bennett PH (1989) Lancet 1:1356–1359
Bonora E, Targher G, Alberiche M, Formentini G, Calcaterra F, Lombardi S, Marini F, Poli M, Zenari L, Raffaelli A, Perbellini S, Zenere MB, Saggiani F, Bonadonna RC, Muggeo M (2002) Diabet Med 19:535–542. doi:10.1046/j.1464-5491.2002.00764.x
Turner RC, Cull CA, Frighi V, Holman RR (1999) JAMA 281:2005–2012. doi:10.1001/jama.281.21.2005
DeFronzo RA (1999) Ann Int Med 131:281–303
Spiegelman BM (1998) Diabetes 47:507–514. doi:10.2337/diabetes.47.4.507
Hofmann C, Lorenz K, Colca JR (1991) Endocrinology 129:1915–1925
Kemnitz JW, Elson DF, Roecker EB, Baum ST, Bergman RN, Meglasson MD (1994) Diabetes 43:204–211. doi:10.2337/diabetes.43.2.204
Spiegelman BM (1998) Diabetes 47:507–514. doi:10.2337/diabetes.47.4.507
Miyazaki Y, Mahankali A, Matsuda M, Glass L, Mahankali S, Ferrannini E, Cusi K, Mandarino LJ, DeFronzo RA (2001) Diabetes Care 24:710–719. doi:10.2337/diacare.24.4.710
Lenhard JM, Kliewer SA, Paulik MA, Plunket KD, Lehmann JM, Weiel JE (1997) Biochem Pharmacol 54:801–808. doi:10.1016/S0006-2952(97)00229-3
Rosenstock J, Einhorn D, Hershon K, Glazer NB, Yu S (2002) Int J Clin Pract 56:251–257
Aronoff S, Rosenblatt S, Braithwaite S, Egan JW, Mathisen AL, Schneider RL (2000) Diabetes Care 23:1605–1611. doi:10.2337/diacare.23.11.1605
Kipnes MS, Krosnick A, Rendell MS, Egan JW, Mathisen AL, Schneider RL (2001) Am J Med 111:10–17. doi:10.1016/S0002-9343(01)00713-6
Einhorn D, Rendell M, Rosenzweig J, Egan JW, Mathisen AL, Schneider RL (2000) Clin Ther 22:1395–1409. doi:10.1016/S0149-2918(00)83039-8
Matthews DR, Cull CA, Stratton IM, Holman RR, Turner RC (1998) Diabet Med 15:297–303. doi:10.1002/(SICI)1096-9136(199804)15:4<297::AID-DIA572>3.3.CO;2-N
Kiayias JA, Vlachou ED, Theodosopoulou E, Lakka-Papadodima E (2002) Diabetes Care 25:1251–1252. doi:10.2337/diacare.25.7.1251
Ramachandran A, Snehalatha C, Salini J, Vijay V (2004) J Assoc Physicians India 52:459–463
Dams R, Huestis MA, Lambert WE, Murphy CM (2003) J Am Soc Mass Spectrom 14:1290–1294. doi:10.1016/S1044-0305(03)00574-9
Food and Drug Administration of the United States (2001) Guidance for Industry-Bioanalytical Method Validation, US Department of Health and Human Services, Center for Drug Evaluation and Research (CDER), Center for Veterinary Medicine (CVM). http://www/fda.gov/cder/guidance/index.htm
Jain D, Jain S, Jain D, Amin M (2008) J Chromatogr Sci 46:501–504
Wang M, Miksa IR (2007) J Chromatogr B 856:318–327. doi:10.1016/j.jchromb.2007.06.037
Mistri HN, Jangid AG, Shrivastav PS (2007) J Pharm Biomed Anal 45(1):97–106. doi:10.1016/j.jpba.2007.06.003
Acknowledgments
One of the authors’ Pinaki Sengupta, is grateful to the Department of Science and Technology (DST), Govt. of India, New Delhi, for providing necessary instrumental facilities to carry out this work through their project No. VI/D&P/186/06-07/TDT under DPRP programme.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sengupta, P., Bhaumik, U., Ghosh, A. et al. LC–MS–MS Development and Validation for Simultaneous Quantitation of Metformin, Glimepiride and Pioglitazone in Human Plasma and Its Application to a Bioequivalence Study. Chroma 69, 1243–1250 (2009). https://doi.org/10.1365/s10337-009-1056-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-009-1056-5