Abstract
Background
Current guidelines advocate that all rectal cancer patients with American Joint Committee on Cancer (AJCC) stages II and III disease should be subjected to neoadjuvant therapy. However, improvements in surgical technique have resulted in single-digit local recurrence rates with surgery only.
Methods
Operative, postoperative, and oncological outcomes of patients with and without neoadjuvant therapy were compared between January 2002 and December 2013. For this purpose, all patients resected with low anterior rectal resection (LAR) and total mesorectal excision (TME) who had or had not been irradiated were identified from the authors’ prospectively maintained database. Patients who were excluded were those with high rectal cancer or AJCC stage IV disease; in the surgery-only group, patients with AJCC stage I disease or with pT4Nx rectal cancer; and in the irradiated patients, patients with ypT4Nx or cT4Nx rectal cancer.
Results
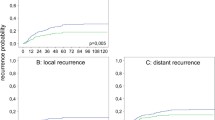
Overall, 454 consecutive patients were included. A total of 342 (75 %) patients were irradiated and 112 (25 %) were not irradiated. Median follow-up for all patients was 48 months. Among patients with and without irradiation, pathological circumferential resection margin positivity rates (2.9 vs. 1.8 %, p = 0.5) were not different. At 5 years, in irradiated patients compared with surgery-only patients, the incidence of local recurrence was decreased (4.5 vs. 3.8 %, p = 0.5); however, systemic recurrences occurred more frequently (10 vs. 17.8 %, p = 0.2). Irradiation did not affect overall or disease-free survival (neoadjuvant treatment vs. surgery-only: 84.9 vs. 88.2 %, p = 0.9; 76 vs. 79.1 %, p = 0.8).
Conclusions
The current study adds to the growing evidence that suggests a selective rather than generalized indication for neoadjuvant treatment in stages II and III rectal cancer.


Similar content being viewed by others
References
Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery: the clue to pelvic recurrence? Br J Surg. 1982;69:613–6.
Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1:1479–82.
Kapiteijn E, Marijnen CA, Nagtegaal ID, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345:638–46.
Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351:1731–40.
Sebag-Montefiore D, Stephens RJ, Steele R, et al. Preoperative radiotherapy versus selective postoperative chemoradiotherapy in patients with rectal cancer (MRC CR07 and NCIC-CTG C016): a multicentre, randomised trial. Lancet. 2009;373:811–20.
Peeters KC, Marijnen CA, Nagtegaal ID, et al. The TME trial after a median follow-up of 6 years: increased local control but no survival benefit in irradiated patients with resectable rectal carcinoma. Ann Surg. 2007;246:693–701.
van Gijn W, Marijnen CA, Nagtegaal ID, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011;12:575–82.
Bruheim K, Guren MG, Dahl AA, et al. Sexual function in males after radiotherapy for rectal cancer. Int J Radiat Oncol Biol Phys. 2010;76:1012–7.
Bruheim K, Guren MG, Skovlund E, et al. Late side effects and quality of life after radiotherapy for rectal cancer. Int J Radiat Oncol Biol Phys. 2010;76:1005–11.
Bruheim K, Svartberg J, Carlsen E, et al. Radiotherapy for rectal cancer is associated with reduced serum testosterone and increased FSH and LH. Int J Radiat Oncol Biol Phys. 2008;70:722–7.
Bruheim K, Tveit KM, Skovlund E, et al. Sexual function in females after radiotherapy for rectal cancer. Acta Oncol. 2010;49:826–32.
Peeters KC, van de Velde CJ, Leer JW, et al. Late side effects of short-course preoperative radiotherapy combined with total mesorectal excision for rectal cancer: increased bowel dysfunction in irradiated patients: a Dutch colorectal cancer group study. J Clin Oncol. 2005;23:6199–206.
Frasson M, Garcia-Granero E, Roda D, et al. Preoperative chemoradiation may not always be needed for patients with T3 and T2N+ rectal cancer. Cancer. 2011;117:3118–25.
MERCURY Study Group. Extramural depth of tumor invasion at thin-section MR in patients with rectal cancer: results of the MERCURY study. Radiology. 2007;243:132–9.
Simunovic M, Sexton R, Rempel E, et al. Optimal preoperative assessment and surgery for rectal cancer may greatly limit the need for radiotherapy. Br J Surg. 2003;90:999–1003.
Contin P, Kulu Y, Bruckner T, et al. Comparative analysis of late functional outcome following preoperative radiation therapy or chemoradiotherapy and surgery or surgery alone in rectal cancer. Int J Colorectal Dis. 2014;29:165–75.
Kulu Y, Ulrich A, Bruckner T, et al. Validation of the International Study Group of Rectal Cancer definition and severity grading of anastomotic leakage. Surgery. 2013;153:753–61.
Rahbari NN, Ulrich AB, Bruckner T, et al. Surgery for locally recurrent rectal cancer in the era of total mesorectal excision: is there still a chance for cure? Ann Surg. 2011;253:522–33.
Welsch T, Mategakis V, Contin P, et al. Results of extralevator abdominoperineal resection for low rectal cancer including quality of life and long-term wound complications. Int J Colorectal Dis. 2013;28:503–10.
Adam IJ, Mohamdee MO, Martin IG, et al. Role of circumferential margin involvement in the local recurrence of rectal cancer. Lancet. 1994;344:707–11.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Randomised trial of surgery alone versus surgery followed by radiotherapy for mobile cancer of the rectum. Medical Research Council Rectal Cancer Working Party. Lancet. 1996;348:1610–4.
Mrak K, Eberl T, Fritz J, Tschmelitsch J. Influence of body mass index on postoperative complications after rectal resection for carcinoma. South Med J. 2012;105:493–9.
Rodel C, Liersch T, Becker H, et al. Preoperative chemoradiotherapy and postoperative chemotherapy with fluorouracil and oxaliplatin versus fluorouracil alone in locally advanced rectal cancer: initial results of the German CAO/ARO/AIO-04 randomised phase 3 trial. Lancet Oncol. 2012;13:679–87.
Valenti V, Hernandez-Lizoain JL, Baixauli J, et al. Analysis of early postoperative morbidity among patients with rectal cancer treated with and without neoadjuvant chemoradiotherapy. Ann Surg Oncol. 2007;14:1744–51.
Laurent C, Leblanc F, Wutrich P, et al. Laparoscopic versus open surgery for rectal cancer: long-term oncologic results. Ann Surg. 2009;250:54–61.
Bennis M, Parc Y, Lefevre JH, et al. Morbidity risk factors after low anterior resection with total mesorectal excision and coloanal anastomosis: a retrospective series of 483 patients. Ann Surg. 2012;255:504–10.
Fujita S, Akasu T, Mizusawa J, et al. Postoperative morbidity and mortality after mesorectal excision with and without lateral lymph node dissection for clinical stage II or stage III lower rectal cancer (JCOG0212): results from a multicentre, randomised controlled, non-inferiority trial. Lancet Oncol. 2012;13:616–21.
Ulrich A, Weitz J, Slodczyk M, et al. Neoadjuvant treatment does not influence perioperative outcome in rectal cancer surgery. Int J Radiat Oncol Biol Phys. 2009;75:129–36.
S3-Leitlinie Kolorektales Karzinom. Langversion 1.0, AWMF Registrierungsnummer: 021-007OL. In: Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF).
Monson JR, Weiser MR, Buie WD, et al. Practice parameters for the management of rectal cancer (revised). Dis Colon Rectum. 2013;56:535–50.
Wong RK, Berry S, Spithoff K, et al. Preoperative or postoperative therapy for stage II or III rectal cancer: an updated practice guideline. Clin Oncol (R Coll Radiol). 2010;22:265–71.
Cawthorn SJ, Parums DV, Gibbs NM, et al. Extent of mesorectal spread and involvement of lateral resection margin as prognostic factors after surgery for rectal cancer. Lancet. 1990;335:1055–9.
Merkel S, Mansmann U, Siassi M, et al. The prognostic inhomogeneity in pT3 rectal carcinomas. Int J Colorectal Dis. 2001;16:2983–04.
Willett CG, Badizadegan K, Ancukiewicz M, Shellito PC. Prognostic factors in stage T3N0 rectal cancer: do all patients require postoperative pelvic irradiation and chemotherapy? Dis Colon Rectum. 1999;42:167–73.
Muthusamy VR, Chang KJ. Optimal methods for staging rectal cancer. Clin Cancer Res. 2007;13:6877s–84s.
Hermanek P, Merkel S, Fietkau R, et al. Regional lymph node metastasis and locoregional recurrence of rectal carcinoma in the era of TME [corrected] surgery. Implications for treatment decisions. Int J Colorectal Dis. 2010;25:359–68.
Kosinski L, Habr-Gama A, Ludwig K, Perez R. Shifting concepts in rectal cancer management: a review of contemporary primary rectal cancer treatment strategies. CA Cancer J Clin. 2012;62:173–202.
Park JS, Jang YJ, Choi GS, et al. Accuracy of preoperative MRI in predicting pathology stage in rectal cancers: node-for-node matched histopathology validation of MRI features. Dis Colon Rectum. 2014;57:32–8.
Nagtegaal ID, Marijnen CA, Kranenbarg EK, et al. Circumferential margin involvement is still an important predictor of local recurrence in rectal carcinoma: not one millimeter but two millimeters is the limit. Am J Surg Pathol. 2002;26:350–7.
Nagtegaal ID, Quirke P. What is the role for the circumferential margin in the modern treatment of rectal cancer? J Clin Oncol. 2008;26:303–12.
Quirke P, Steele R, Monson J, et al. Effect of the plane of surgery achieved on local recurrence in patients with operable rectal cancer: a prospective study using data from the MRC CR07 and NCIC-CTG CO16 randomised clinical trial. Lancet. 2009;373:821–8.
Taylor FG, Quirke P, Heald RJ, et al. Preoperative high-resolution magnetic resonance imaging can identify good prognosis stage I, II, and III rectal cancer best managed by surgery alone: a prospective, multicenter, European study. Ann Surg. 2011;253:711–9.
Taylor FG, Quirke P, Heald RJ, et al. One millimetre is the safe cut-off for magnetic resonance imaging prediction of surgical margin status in rectal cancer. Br J Surg. 2011;98:872-879.
Nagtegaal ID, van de Velde CJ, van der Worp E, et al. Macroscopic evaluation of rectal cancer resection specimen: clinical significance of the pathologist in quality control. J Clin Oncol. 2002;20:1729–34.
Strassburg J, Ruppert R, Ptok H, et al. MRI-based indications for neoadjuvant radiochemotherapy in rectal carcinoma: interim results of a prospective multicenter observational study. Ann Surg Oncol. 2011;18:2790–9.
Bokey EL, Ojerskog B, Chapuis PH, et al. Local recurrence after curative excision of the rectum for cancer without adjuvant therapy: role of total anatomical dissection. Br J Surg. 1999;86:1164–70.
Merkel S, Meyer T, Gohl J, Hohenberger W. Late locoregional recurrence in rectal carcinoma. Eur J Surg Oncol. 2002;28:716–22.
Merkel S, Mansmann U, Hohenberger W, Hermanek P. Time to locoregional recurrence after curative resection of rectal carcinoma is prolonged after neoadjuvant treatment: a systematic review and meta-analysis. Colorectal Dis. 2011;13:123–31.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kulu, Y., Tarantino, I., Billeter, A.T. et al. Comparative Outcomes of Neoadjuvant Treatment Prior to Total Mesorectal Excision and Total Mesorectal Excision Alone in Selected Stage II/III Low and Mid Rectal Cancer. Ann Surg Oncol 23, 106–113 (2016). https://doi.org/10.1245/s10434-015-4832-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4832-5