Abstract
Purpose
Patients with resectable thoracic esophageal squamous cell cancer (TESCC) and positron emission tomography (PET)-positive lymph nodes (PET-N positive) are likely to have ≥3 pathological lymph node metastases (pLNMs) and show a higher rate of postoperative recurrence despite curative resection than PET-N-negative TESCC patients. We examined the prognostic significance of 18F-fluorodeoxyglucose uptake into lymph node metastases after neoadjuvant chemotherapy (NAC) for PET-N positive TESCC and aimed to propose the optimal NAC response criteria for these patients.
Methods
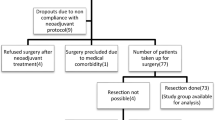
Fifty-one patients with PET-N positive TESCC underwent two courses of NAC followed by surgery. Metabolic responses of primary tumors and LNs were prospectively evaluated and associations with clinicopathological data and patient survival assessed by univariate and multivariate analyses.
Results
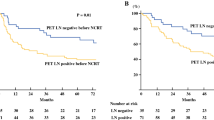
After NAC, 21 patients were post-treatment (post-) PET-N positive and 30 post-PET-N negative. A significantly (p < 0.001) high proportion of the post-PET-N-negative group had ≤2 pLNMs than the post-PET-N positive group (86.7 vs. 28.6 %). The PET-N negative group also had a significantly lower distant metastasis rate (23.3 vs. 75.0 %) and higher 5-year relapse-free survival (RFS) rate (69.0 vs. 20.0 %). Univariate and multivariate Cox’s proportional hazard regression analyses identified post-PET-N negative status as the only significant favorable predictive factor for low postoperative recurrence (p = 0.015) independent of the primary tumor response.
Conclusions
PET-N negative status predicts ≤2 pLNMs and longer RFS in resectable TESCC patients even after NAC. Therefore, post-PET-N status, not the effects on the primary tumor, is a critical NAC treatment response criterion for evaluating prognosis and guiding subsequent treatment.


Similar content being viewed by others
References
Dresner SM, Griffin SM. Pattern of recurrence following radical oesophagectomy with two-field lymphadenectomy. Br J Surg. 2000;87:1426–33.
Nakagawa S, Kanda T, Kosugi S, et al. Recurrence pattern of squamous cell carcinoma of the thoracic esophagus after extended radical esophagectomy with three-field lymphadenectomy. J Am Coll Surg. 2004;198:205–11.
Kunisaki C, Makino H, Kimura J, et al. Impact of lymph-node metastasis site in patients with thoracic esophageal cancer. J Surg Oncol. 2010;101:36–42.
Rice TW, Rusch VW, Apperson-Hansen C, et al. Worldwide esophageal cancer collaboration. Dis Esophagus. 2009;22:1–8.
Rice TW, Rusch VW, Ishwaran H, et al. Cancer of the esophagus and esophagogastric junction. Data-driven staging for the seventh edition of the American Joint Committee on Cancer/International Union against Cancer cancer staging manuals. Cancer. 2010;116:3763–73.
Yasuda T, Higuchi I, Yano M, et al. The impact of 18F-fluorodeoxyglucose positron emission tomography positive lymph nodes on postoperative recurrence and survival in resectable thoracic esophageal squamous cell carcinoma. Ann Surg Oncol. 2012;19:652–60.
Yasuda T, Yano M, Miyata H, et al. Systemic control and evaluation of the response to neoadjuvant chemotherapy in resectable thoracic esophageal squamous cell carcinoma with 18F-fluorodeoxyglucose positron emission tomography-positive lymph nodes. Surg Today. 2014 Jun 27; [Epub ahead of print]. Accessed 27 June 2014. doi:10.1007/s00595-014-0956-7.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumors: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
WHO handbook for reporting results of cancer treatment. Geneva (Switzerland): World Health Organization Offset Publication No. 48; 1979.
Miller AB, Hoogstraten B, Staquet M, et al. Reporting results of cancer treatment. Cancer. 1981;47:207–14.
Komori T, Doki Y, Kabuto T, et al. Prognostic significance of the size of cancer nests in metastatic lymph nodes in human esophageal cancers. J Surg Oncol. 2003;82:19–27.
Aguirre ACR, Ramos-Font C, Portero RV, et al. 18F-fluorodeoxyglucose positron emission tomography for the evaluation of neoadjuvant therapy response in esophageal cancer. systematic review of the literature. Ann Surg. 2009;250:247–54.
Omloo JMT, van Heijl M, Hoekstra OS, et al. FDG-PET parameters as prognostic factor in esophageal cancer patients: a review. Ann Surg Oncol. 2011;18:3338–352.
Wong R, Walker-Dilks C, Raifu A. Evidence-based guideline recommendations on the use of positron emission tomography imaging in oesophageal cancer. Clin Oncol. 2012;24:86–104.
Akutsu Y, Shuto K, Kono T, et al. The number of pathologic lymph nodes involved is still a significant prognostic factor even after neoadjuvant chemoradiotherapy in esophageal squamous cell carcinoma. J Surg Oncol. 2012;105:756–60.
Castoro C, Scarpa M, Cagol M, et al. Nodal metastasis from locally advanced esophageal cancer: how neoadjuvant therapy modifies their frequency and distribution. Ann Surg Oncol. 2011;18:3743–54.
Sobin LH, Wittekind C, eds. TNM classification of malignant tumors, 6th edn. New York: Wiley; 2002.
Greene FL, Page DL, Fleming ID, et al, eds. AJCC cancer staging manual, 6th edn. New York: Springer; 2002.
Fukunaga H, Sekimoto M, Tatsumi M, et al. Clinical relevance of fusion images using (18)F-2-fluoro-2-deoxy-D-glucose positron emission tomography in local recurrence of rectal cancer. Int J Oncol. 2002;20:691–5.
Paul AK, Tatsumi M, Higuchi I, et al. Gamma camera coincidence imaging with [18F]fluorodeoxyglucose in the pretreatment evaluation of patients with oesophageal cancer. Nucl Med Commun. 2003;24:963–70.
Higuchi I, Yasuda T, Yano M, et al. Lack of fluorodeoxyglucose F18 uptake in posttreatment positron emission tomography as a significant predictor of survival after subsequent surgery in multimodality treatment for patients with locally advanced esophageal squamous cell carcinoma. J Thorac Cardiovasc Surg. 2008;136:205–12.
Akita H, Doki Y, Miyata H, et al. Clinical significance of the second cycle response to cisplatin-based chemotherapy as preoperative treatment for esophageal squamous cell carcinoma. J Surg Oncol. 2006;93:401–9.
Shiozaki H, Yano M, Tsujinaka T, et al. Lymph node metastasis along the recurrent nerve chain is an indication for cervical lymph node dissection in thoracic esophageal cancer. Dis Esophagus. 2001;14:191–6.
Miyata H, Yano M, Doki Y, at al. A prospective trial for avoiding cervical lymph node dissection for thoracic esophageal cancers, based on intra-operative genetic diagnosis of micrometastasis in recurrent laryngeal nerve chain nodes. J Surg Oncol. 2006;93:477–84.
The Japan Esophageal Society. Japanese classification of esophageal cancer, tenth edition: part I. Esophagus. 2009;6:1–25.
Dhar DK, Hattori S, Tonomoto Y, et al. Appraisal of a revised lymph node classification system for esophageal squamous cell cancer. Ann Thorac Surg. 2007;83:1265–72.
Gillies RS, Middleton MR, Han C, et al. Role of positron emission tomography-computed tomography in predicting survival after neoadjuvant chemotherapy and surgery for oesophageal adenocarcinoma. Br J Surg. 2012;99:239–45.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2014_4299_MOESM1_ESM.ppt
Supplemental Fig. E1 Relapse-free survival according to the number of pathologically metastatic lymph nodes. Each percent value shows 5-year survival rate (PPT 171 kb)
Rights and permissions
About this article
Cite this article
Yasuda, T., Yano, M., Miyata, H. et al. Prognostic Significance of 18F-fluorodeoxyglucose Positron Emission Tomography (FDG-PET)-Positive Lymph Nodes Following Neoadjuvant Chemotherapy and Surgery for Resectable Thoracic Esophageal Squamous Cell Carcinoma. Ann Surg Oncol 22, 2599–2607 (2015). https://doi.org/10.1245/s10434-014-4299-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-4299-9