Abstract
Background
The variable incidence of gallbladder cancer (GBCA) suggests regional pathogenetic differences. This study compares cell cycle-regulatory, angiogenesis-related, and PI3K pathway protein expression in GBCAs from three continents.
Methods
Immunohistochemical expression of several proteins was assessed, correlated with clinicopathologic variables, and compared among centers from Chile (Fundación Arturo López Pérez [FALP]), Japan (Yokohama City University [YCU]), and the United States (Memorial Sloan-Kettering Cancer Center [MSKCC]). Hierarchical clustering was used to partition the data based on protein-expression and treatment center.
Results
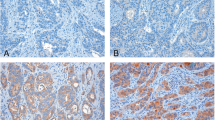
Tissue from 117 patients (MSKCC = 76; FALP = 22; YCU = 19) was analyzed. Mdm2 overexpression was seen only at MSKCC (p < 0.0001). Absence of p21 (p = 0.03) and VEGFR2 (p = 0.018) were more common and p27 expression was less frequent (p = 0.047) in tumors from YCU. Ki-67 labeling index in YCU tumors (median = 10) was two-thirds lower than at other centers. On hierarchical clustering analysis, all YCU patients (p = 0.017) and those with early tumors (p = 0.017) clustered separately from MSKCC. Median disease-specific survival after curative intent (R0) resection was 27 months and was similar among centers (p = 0.9). Median disease-specific survival of patients with early tumors was 28.4 months and was higher at YCU (not reached, p = 0.06).
Conclusions
Cell cycle-regulatory protein expression patterns of YCU tumors differed from those treated at FALP and MSKCC. The differential clustering of protein expression and survival in patients with early tumors suggest regional differences in pathogenesis and disease biology.


Similar content being viewed by others
References
Andia ME, Hsing AW, Andreotti G, Ferreccio C. Geographic variation of gallbladder cancer mortality and risk factors in Chile: a population-based ecologic study. Int J Cancer. 2008;123(6):1411–6.
Hariharan D, Saied A, Kocher HM. Analysis of mortality rates for gallbladder cancer across the world. HPB. 2008;10(5):327–31.
Kumar JR, Tewari M, Rai A, Sinha R, Mohapatra SC, Shukla HS. An objective assessment of demography of gallbladder cancer. J Surg Oncol. 2006;93(8):610–4.
Lazcano-Ponce EC, Miquel JF, Munoz N, et al. Epidemiology and molecular pathology of gallbladder cancer. CA Cancer J Clin. 2001;51(6):349–64.
Dutta U, Nagi B, Garg PK, Sinha SK, Singh K, Tandon RK. Patients with gallstones develop gallbladder cancer at an earlier age. Eur J Cancer Prev. 2005;14(4):381–5.
Elnemr A, Ohta T, Kayahara M, et al. Anomalous pancreaticobiliary ductal junction without bile duct dilatation in gallbladder cancer. Hepatogastroenterology. 2001;48(38):382–6.
Kayahara M, Nagakawa T. Recent trends of gallbladder cancer in Japan: an analysis of 4,770 patients. Cancer. 2007;110(3):572–80.
Tewari M, Kumar V, Mishra RR, Kumar M, Shukla HS. Is there a role for cholecystectomy in gallbladder carcinoma discovered to be unresectable for cure at laparotomy? World J Surg. 2008;32(12):2683–7.
Wistuba II, Miquel JF, Gazdar AF, Albores-Saavedra J. Gallbladder adenomas have molecular abnormalities different from those present in gallbladder carcinomas. Hum Pathol. 1999;30(1):21–5.
Moreno M, Pimentel F, Gazdar AF, Wistuba, II, Miquel JF. TP53 abnormalities are frequent and early events in the sequential pathogenesis of gallbladder carcinoma. Ann Hepatol. 2005;4(3):192–9.
Pai RK, Mojtahed K. Mutations in the RAS/RAF/MAP kinase pathway commonly occur in gallbladder adenomas but are uncommon in gallbladder adenocarcinomas. Appl Immunohistochem Mol Morphol. 2011;19(2):133–40.
Butte JM, Matsuo K, Gonen M, et al. Gallbladder cancer: differences in presentation, surgical treatment, and survival in patients treated at centers in three countries. J Am Coll Surg. 2011;212(1):50–61.
Nakayama K, Konno M, Kanzaki A, et al. Allelotype analysis of gallbladder carcinoma associated with anomalous junction of pancreaticobiliary duct. Cancer Lett. 2001;166(2):135–41.
Hanada K, Tsuchida A, Iwao T, et al. Gene mutations of K-ras in gallbladder mucosae and gallbladder carcinoma with an anomalous junction of the pancreaticobiliary duct. Am J Gastroenterol. 1999;94(6):1638–42.
Hanada K, Tsuchida A, Kajiyama G. Cellular kinetics and gene mutations in gallbladder mucosa with an anomalous junction of pancreaticobiliary duct. J Hepatobiliary Pancreat Surg. 1999;6(3):223–8.
Wistuba II, Gazdar AF, Roa I, Albores-Saavedra J. p53 protein overexpression in gallbladder carcinoma and its precursor lesions: an immunohistochemical study. Hum Pathol. 1996;27(4):360–5.
Yokoyama N, Hitomi J, Watanabe H, et al. Mutations of p53 in gallbladder carcinomas in high-incidence areas of Japan and Chile. Cancer Epidemiol Biomarkers Prev. 1998;7(4):297–301.
Hanada K, Itoh M, Fujii K, et al. TP53 mutations in stage I gallbladder carcinoma with special attention to growth patterns. Eur J Cancer. 1997;33(7):1136–40.
Goldin RD, Roa JC. Gallbladder cancer: a morphological and molecular update. Histopathology. 2009;55(2):218–29.
Harino Y, Imura S, Kanemura H, et al. Role of tumor angiogenesis in gallbladder carcinoma: with special reference to thymidine phosphorylase. Int J Clin Oncol. 2008;13(5):452–7.
Jarnagin WR, Klimstra DS, Hezel M, et al. Differential cell cycle-regulatory protein expression in biliary tract adenocarcinoma: correlation with anatomic site, pathologic variables, and clinical outcome. J Clin Oncol. 2006;24(7):1152–60.
Deshpande V, Nduaguba A, Zimmerman SM, et al. Mutational profiling reveals PIK3CA mutations in gallbladder carcinoma. BMC Cancer. 2011;11:60.
Butte JM, Gonen M, Allen PJ, et al. The role of laparoscopic staging in patients with incidental gallbladder cancer. HPB. 2011;13(7):463–72.
D’Angelica M, Dalal KM, DeMatteo RP, Fong Y, Blumgart LH, Jarnagin WR. Analysis of the extent of resection for adenocarcinoma of the gallbladder. Ann Surg Oncol. 2009;16(4):806–16.
AJCC Cancer Staging Handbook. 7 ed. Chicago: American Joint Committee on Cancer; 2010.
Sakr RA, Barbashina V, Morrogh M, et al. Protocol for PTEN expression by immunohistochemistry in formalin-fixed paraffin-embedded human breast carcinoma. Appl Immunohistochem Mol Morphol. 2010;18(4):371–4.
Della Torre G, Pasquini G, Pilotti S, et al. TP53 mutations and mdm2 protein overexpression in cholangiocarcinomas. Diagn Mol Pathol. 2000;9(1):41–6.
Furubo S, Harada K, Shimonishi T, Katayanagi K, Tsui W, Nakanuma Y. Protein expression and genetic alterations of p53 and ras in intrahepatic cholangiocarcinoma. Histopathology. 1999;35(3):230–40.
Ito Y, Takeda T, Sasaki Y, et al. Bcl-2 expression in cholangiocellular carcinoma is inversely correlated with biologically aggressive phenotypes. Oncology. 2000;59(1):63–7.
Ito Y, Takeda T, Sasaki Y, et al. Expression and clinical significance of the G1-S modulators in intrahepatic cholangiocellular carcinoma. Oncology. 2001;60(3):242–51.
Tenjo T, Toyoda M, Okuda J, et al. Prognostic significance of p27(kip1) protein expression and spontaneous apoptosis in patients with colorectal adenocarcinomas. Oncology. 2000;58(1):45–51.
Wiksten JP, Lundin J, Nordling S, Kokkola A, von Boguslawski K, Haglund C. The prognostic value of p27 in gastric cancer. Oncology. 2002;63(2):180–4.
Randi G, Franceschi S, La Vecchia C. Gallbladder cancer worldwide: geographical distribution and risk factors. Int J Cancer. 2006;118(7):1591–602.
Lemrow SM, Perdue DG, Stewart SL, et al. Gallbladder cancer incidence among American Indians and Alaska Natives, US, 1999–2004. Cancer. 2008;113(5 Suppl):1266–73.
Mori K, Nagakawa T, Ohta T, et al. Association between gallbladder cancer and anomalous union of the pancreaticobiliary ductal system. Hepatogastroenterology. 1993;40(1):56–60.
Schafer KA. The cell cycle: a review. Vet Pathol. 1998;35(6):461–78.
Vermeulen K, Van Bockstaele DR, Berneman ZN. The cell cycle: a review of regulation, deregulation and therapeutic targets in cancer. Cell Prolif. 2003;36(3):131–49.
da Rocha AO, Coutinho LM, Schall JG. The prognostic value of angiogenesis by Chalkley counting in gallbladder carcinoma. Hepatogastroenterology. 2009;56(89):34–8.
Conde E, Angulo B, Tang M, et al. Molecular context of the EGFR mutations: evidence for the activation of mTOR/S6K signaling. Clin Cancer Res. 2006;12(3 Pt 1):710–7.
Yin Y, Shen WH. PTEN: a new guardian of the genome. Oncogene. 2008;27(41):5443–53.
Baba Y, Nosho K, Shima K, et al. Phosphorylated AKT expression is associated with PIK3CA mutation, low stage, and favorable outcome in 717 colorectal cancers. Cancer. 2011;117(7):1399–408.
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Butte, J.M., Torres, J., Veras, E.F. et al. Regional Differences in Gallbladder Cancer Pathogenesis: Insights from a Comparison of Cell Cycle-Regulatory, PI3K, and Pro-Angiogenic Protein Expression. Ann Surg Oncol 20, 1470–1481 (2013). https://doi.org/10.1245/s10434-012-2761-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2761-0