Abstract
Background
Recent adjuvant chemotherapy trials after resection of stage II and III non-small cell lung cancer (NSCLC) have identified important survival differences among patients with immunohistochemical evidence suggesting platinum resistance. No clinical information exists regarding the impact upon survival of patients treated with platinum agents who exhibit cellular evidence of their tumors’ resistance to platinum. We evaluated the utility of the extreme drug resistance (EDR) assay to predict mortality among a consecutive group of stage II through IV NSCLC patients receiving adjuvant or definitive platinum-based chemotherapy after resection or surgical biopsy.
Methods
The Extreme Drug Resistance (EDR) Assay is a clinically validated cellular proliferation assay used to test tumors for chemotherapy drug resistance. Based on response in the EDR assay, tumor specimens from stage II through IV NSCLC patients were segregated into three groups: extreme drug resistant (EDR), intermediate drug resistant (IDR), and low drug resistant (LDR). Patient survival was evaluated after platinum-based chemotherapy.
Results
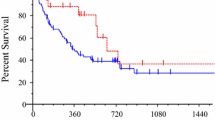
Platinum IDR/EDR was statistically significant in predicting shorter overall survival (29.8 months vs. 15.6 months) among platinum IDR/EDR-resistant patients compared with LDR patients (P = 0.047). Median survival was 16.6 months for patients with IDR/EDR to platinum and any other second agent of doublet therapy compared with patients with LDR to any platinum-based doublet where median survival was not achieved (P = 0.0268).
Conclusions
This is the first study to demonstrate the utility of the EDR assay to predict poor clinical outcome when platinum-based therapy is used to treat patients with biological evidence of tumor resistance to platinum. These data corroborate the finding of recent studies evaluating possible molecular correlates to poor response to specific chemotherapeutic agents.




Similar content being viewed by others
References
American Cancer Society. Cancer facts & figures 2008. American Cancer Society, Atlanta, 2008.
Mountain CF. Revisions in the international system for staging lung cancer. Chest. 1997;111:1710–7.
Winton TL, Livingston R, Johnson D, et al. A prospective randomized trial of adjuvant vinorelbine (VIN) and cisplatin (CIS) in completely resected stage 1B and II non small cell lung cancer (NSCLC) Intergroup JBR.10. N Engl J Med. 2005;352:2289–97.
Douillard J, Rosell R, Delena M, et al. ANITA: phase III adjuvant vinorelbine (N) and cisplatin (P) versus observation (OBS) in completely resected (stage I–III) non-small-cell lung cancer (NSCLC) patients (pts): final results after 70-month median follow-up. On behalf of the Adjuvant Navelbine International Trialist Association. ASCO annual meeting proceedings, Orlando, Florida, USA. J Clin Oncol. 2005;23:7013.
Olaussen KA, Dunant A, Fouret P, Brambilla E, et al. DNA Repair by ERCC1 in non-small-cell lung cancer and cisplatin-based adjuvant chemotherapy. N Engl J Med. 2006;355:983–91.
Mehta R, Bomstein R, Yu I-R, et al. Breast cancer survival and in vitro tumor response in the extreme drug resistance assay. Breast Cancer Res Treatment. 2001;66:225–37.
Holloway R, Mehta R, Finkler N, et al. Association between in vitro platinum resistance in the EDR assay and clinical outcomes for ovarian cancer patients. Gynecol Oncol. 2002;87:8–16.
Parker RJ, Fruehauf JP, Mehta R, et al. A prospective blinded study of predictive value of extreme drug resistance assay in patients receiving CPT-11 for recurrent glioma. J Neuro-Oncol. 2004;66:365–75.
d’Amato TA, Landreneau RJ, McKenna RJ, et al. Prevalence of in vitro extreme chemotherapy resistance in resected non-small cell lung cancer. Ann Thor Surg. 2006;81:440–7.
d’Amato TA, Landreneau RJ, Ricketts W, Huang W, et al. Chemotherapy resistance and oncogene expression in non-small cell lung cancer. J Thorac Cardiovasc Surg. 2007;133:352–63.
Greene FL, Page DL, Fleming ID, et. al. AJCC cancer staging manual, 6th ed. Springer-Verlag, New York, 2002.
Schiller J, Harrington D, Belani C, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002;346:92–8.
Kern D, Weisenthal L. Highly specific prediction of antineoplastic drug resistance with an in vitro assay using suprapharacologic drug exposures. J Natl Cancer Inst. 1990;82:582–8.
Wilbur DW, Camacho ES, Hilliard DA, et al. Chemotherapy of non-small cell lung carcinoma guided by an in vitro drug resistance assay measuring total tumour cell kill. Br J Cancer. 1992;65:27–32.
Moon YW, Choi SH, Kim YT, Sohn JH, et al. Adenosine triphosphate-based chemotherapy response assay (ATP-CRA)-guided platinum-based 2-drug chemotherapy for unresectable non-small-cell lung cancer. Cancer. 2007;109:1829–35.
Langer CJ, Stephenson P, Thor A, et al. Trastuzumab in the treatment of advanced non–small-cell lung cancer: is there a role? Focus on Eastern Cooperative Oncology Group Study 2598 J Clin Oncol. 2004;22:1180–7.
Pignon JP, Tribodet H, Scagliotti GV, Douillard JY, et al. Lung adjuvant cisplatin evaluation: a pooled analysis by the lace collaborative group. J Clin Oncol. 2008;26:3552–9.
Tsao MS, Aviel-Ronen S, Ding K, Lau D, et al. Prognostic and predictive importance of p53 and ras for adjuvant chemotherapy in non small-cell lung cancer. J Clin Oncol. 2007;25:5240–7.
Riely GJ, Marks J, Pao W. KRAS mutations in non-small cell lung cancer. Proc Am Thorac Soc. 2009;6:201–5.
Potti A, Mukherjee S, Petersen R, et al. A genomic strategy to refine prognosis in early-stage non-small cell lung cancer. N Engl J Med. 2006;355:570–80.
Chen HY, Yu SL, Chen CH, Chang GC, et al. A five-gene signature and clinical outcome in non-small-cell lung cancer. N Engl J Med. 2007;356:11–20.
Boutros PC, Lau SK, Pintilie M, Liu N, et al. Prognostic gene signatures for non-small-cell lung cancer. Proc Natl Acad Sci. 2009;106:2824–8.
Schwartz AM, Henson DE. Diagnostic surgical pathology in lung cancer: ACCP evidence-based clinical practice guidelines, 2nd ed. Chest. 2007;132(3 Suppl):78S–93S.
d’Amato TA. Adjuvant chemotherapy and the role of chemoresistance testing for stage I non-small cell lung cancer. Thorac Surg Clin. 2007;17:287–99.
Strauss G, Herndon J, Maddaus M, et al. Adjuvant chemotherapy in stage IB non-small cell lung cancer (NSCLC): Update of Cancer and Leukemia Group B (CALGB) protocol 9633. ASCO Annual Meeting Proceedings Part I. J Clin Oncol. 2006;24(18S):7007.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
d’Amato, T.A., Pettiford, B.L., Schuchert, M.J. et al. Survival Among Patients with Platinum Resistant, Locally Advanced Non-Small Cell Lung Cancer Treated with Platinum-Based Systemic Therapy. Ann Surg Oncol 16, 2848–2855 (2009). https://doi.org/10.1245/s10434-009-0608-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-009-0608-0